Talbutal
 | |
| Clinical data | |
|---|---|
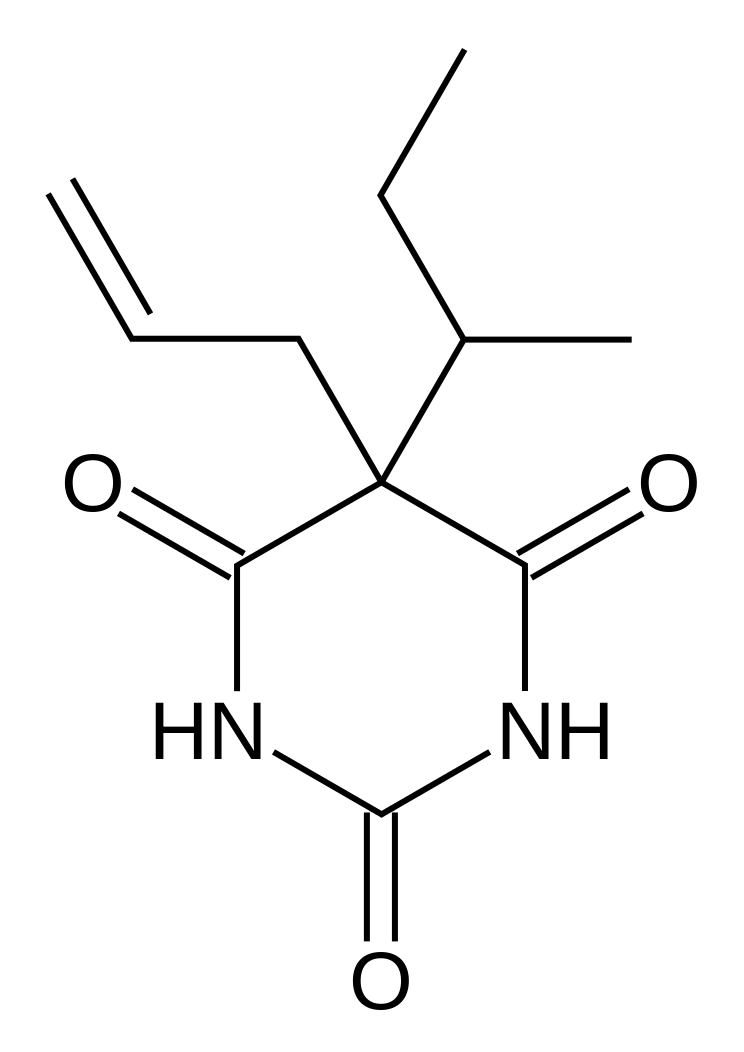
| Synonyms | 5-(1-methylpropyl)-5-(2-propenyl)-2,4,6(1H,3H,5H)-pyrimidinetrione |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C11H16N2O3 |
| Molar mass | 224.256 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Talbutal |
|
Articles |
|---|
|
Most recent articles on Talbutal |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Talbutal at Clinical Trials.gov Clinical Trials on Talbutal at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Talbutal
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Talbutal Risk calculators and risk factors for Talbutal
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Talbutal |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Talbutal (Lotusate) is a barbiturate with a short to intermediate duration of action. It is a structural isomer of butalbital. Talbutal is a schedule III drug in the U.S.
Pharmacology
Talbutal is a short to intermediate-acting barbiturate. Barbiturates act as nonselective depressants of the central nervous system (CNS), capable of producing all levels of CNS mood alteration from excitation to mild sedation, hypnosis, and deep coma. In sufficiently high therapeutic doses, barbiturates induce anesthesia.[1]
Mechanism of action
Talbutal binds at a distinct binding site associated with a Cl− ionopore at the GABAA receptor, increasing the duration of time for which the Cl− ionopore is open. The post-synaptic inhibitory effect of GABA in the thalamus is, therefore, prolonged.
Toxicity
Symptoms of acute barbiturate poisoning include drowsiness, confusion, coma, respiratory depression, hypotension,[1] and shock.
References
- ↑ 1.0 1.1 Mutschler, Ernst; Schäfer-Korting, Monika (2001). Arzneimittelwirkungen (in German) (8 ed.). Stuttgart: Wissenschaftliche Verlagsgesellschaft. pp. 280ff. ISBN 3-8047-1763-2.
- Pages with script errors
- CS1 maint: Unrecognized language
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without KEGG source
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Barbiturates
- Drug