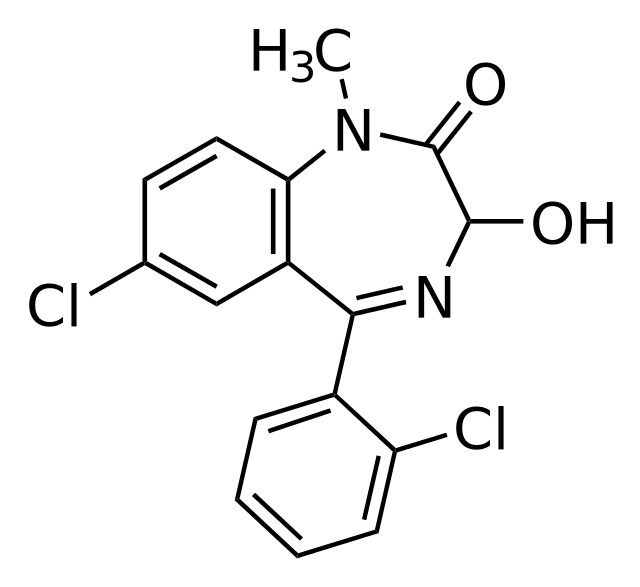
Lormetazepam
 | |
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 80% |
| Metabolism | Hepatic |
| Elimination half-life | 10–12 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C16H12Cl2N2O2 |
| Molar mass | 335.2 g/mol |
|
WikiDoc Resources for Lormetazepam |
|
Articles |
|---|
|
Most recent articles on Lormetazepam Most cited articles on Lormetazepam |
|
Media |
|
Powerpoint slides on Lormetazepam |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Lormetazepam at Clinical Trials.gov Clinical Trials on Lormetazepam at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Lormetazepam
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Lormetazepam Discussion groups on Lormetazepam Patient Handouts on Lormetazepam Directions to Hospitals Treating Lormetazepam Risk calculators and risk factors for Lormetazepam
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Lormetazepam |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Lormetazepam is available as a generic or available under the following trade or brand names (Noctamid, Ergocalm, Loramet, Dilamet, Sedaben, Stilaze, Nocton, Pronoctan, Noctamide, Loretam, Minias, Aldosomnil) and is also sometimes known as methyllorazepam, is a drug which is a benzodiazepine derivative. It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties.
Lormetazepam is not approved for sale in the United States or Canada, though it is licensed it the UK as 0.5 and 1 mg tablets for short term treatment (2-4 weeks) of severe or disabling insomnia. It is the most expensive hypnotic benzodiazepine on the UK market - costing upwards of £15 (trade price) for a 28 day supply of the 1mg variant. Recently however, prices have stabilised towards the £3 region. It's bioavailability was found to be 80%.[1]
Mechanism of Action
Lormetazepam and other benzodiazepine drugs act as positive modulators at the GABAa-benzodiazepine receptor complex. Lormetazepam binds to the benzodiazepine receptor which in turn enhances the effect of the GABAa receptor producing its therapeutic effects as well as adverse effects. When lormetazepam binds to the benzodiazepine receptor sites in sufficient quantities it produces sedation which is used clinically as a therapeutic treatment for insomnia. Lormetazepam alters the brain electrical activity which has been studied via EEG readings.[2] Changes in EEG can therefore be used to measure the sedative sleep promoting properties of lormetazepam.
Indications
Insomnia. Insomnia can be described as a difficulty falling asleep, frequent awakening, early awakenings or a combination of each. Lormetazepam is an intermediate acting benzodiazepine and is sometimes used in patients who have difficulty in maintaining sleep or falling asleep. Hypnotics should only be used on a short term basis or in those with chronic insomnia on an occasional basis.[3]
Side Effects
Side effects of lormetazepam are similar to other hypnotic benzodiazepines and can for the most part be regarded as a class effect.[4] Some side effects can be enhanced when lormetazepam is combined with other drugs with similar effects eg alcohol and nonbenzodiazepine drugs, with subsequent increased drowsiness, amnesia or respiratory depression. Lormetazepam has been associated with adversely affecting immediate and delayed recall memory functions.[5]
Side Effects List
- Somnolence
- paradoxical increase in aggression
- lightheadedness
- confusion
- muscle weakness
- ataxia (particularly in the elderly)
- anterograde amnesia
- headache
- vertigo
- hypotension
- salivation changes
- gastro-intestinal disturbances
- visual disturbances
- dysarthria
- tremor
- changes in libido
- incontinence
- urinary retention
- blood disorders and jaundice
- skin reactions
- dependence and withdrawal reactions
Residual 'hangover' effects after nighttime administration of lormetazepam such as sleepiness, impaired psychomotor and cognitive functions may persist into the next day which may impair the ability of users to drive safely and increase risks of falls and hip fractures.[6]
Tolerance, Dependence and Withdrawal
It should be noted that the risks of tolerance, dependence and withdrawal are very low when the drug is used for 2 - 4 weeks only and that lormetazepam is generally a safe and effective drug when used for no longer than 2-4 weeks. Some sleep disturbance in the form of rebound insomnia can however occur even after short term usage of 7 days.[7]Those with a history of addiction may be at increased risk of problems of tolerance and dependence especially those with a past history of dependency on sedative hypnotic drugs.
Tolerance
Lormetazepam as with other benzodiazepines is generally only recommended for short term use (2-4 weeks) due to tolerance and loss of efficacy. Tolerance to the and loss of the sedative effects of benzodiazepine hypnotics occurs within 14 days of regular use.[8]
Dependence
Dependence is the medical term for addiction. Dependence can either be psychological or physical. Psychological dependence can manifest itself as a reliance on a drug to cope with every day life or in the form of craving. Physical dependence occurs due to physiological adaptions occurring as the body attempts to overcome the drugs effects which is known as tolerance and the continuing need to take the drug to avoid or suppress withdrawal symptoms which can sometimes resemble the original condition being treated. When the dose or the drug is discontinued withdrawal symptoms typically occur. Lormetazepam as with all other benzodiazepines produces both physical and psychological dependence but the main problem of concern is physical dependence which appears in the form of the benzodiazepine withdrawal syndrome after the dosage is reduced or the drug is stopped completely. The dependence induced by lormetazepam is related to changes in the sensitivity of the GABA-BZD receptor complex.[9]
Withdrawal Symptoms
Withdrawal Symptoms which can occur from stopping benzodiazepines such as lormetazepam can include:[10]
- rebound insomnia and nightmares
- anxiety, panic attacks and agorophobia
- clinical depression
- malaise
- lack of concentration
- abdominal discomfort
- depersonalisation and feelings of unreality
- emotional liability
- cognitive impairment
- tinnitus
- paraesthesiae, tingling, numbness and pain
- muscle pain, weakness, tension, painful tremor, shaking attacks, jerks, blepharospasm
- excitability, jumpiness and restlessness
- stiffness
- sweats
Abrupt or over rapid withdrawal from high doses can provoke:
- epileptic seizures
- hallucinations (visual, auditory)
- misperceptions
Withdrawal symptoms typically subside after 4 - 8 weeks but in approximately 10 - 15% of individuals symptoms can perist for many months[11] and in rare cases years.[12] Some "Withdrawal Symptoms" can emerge despite a constant dosage with the body needing extra dosage in order to feel normal. This is sometimes associated with dosage escalation.
Lormetazepam has a short to intermediate half life of approximately 10-12 hours. Shorter acting benzodiazepine compounds are generally associated with a more intense and immediate withdrawal reaction compared to longer acting benzodiazepines. For this reason it is generally recommended to cross from lormetazepam to an equivalent dose of diazepam to gradually taper the dosage.[13]
References
- ↑ Hümpel1, M (1982). "Pharmacokinetics and biotransformation of the new benzodiazepine, lormetazepam, in man". Eur. J. Clin. Pharmaco. 21 (5): 421–425. doi:10.1007/BF00542330. Unknown parameter
|coauthors=ignored (help) - ↑ Kurowski M (1982). "Relationship between EEG dynamics and pharmacokinetics of the benzodiazepine lormetazepam". Pharmacopsychiatria. 15 (3): 77–83. Retrieved 16/03/07. Unknown parameter
|coauthors=ignored (help); Check date values in:|accessdate=(help) - ↑ Rickels K. (1986). "The clinical use of hypnotics: indications for use and the need for a variety of hypnotics". Acta Psychiatr Scand Suppl. 332: 132–41. PMID 2883820.
- ↑ British National Formulary Benzodiazepines Information
- ↑ Dorow, R (1987). "Amnestic effects of lormetazepam and their reversal by the benzodiazepine antagonist Ro 15-1788". Psychopharmacology (Berl). 93 (4): 507–514. Unknown parameter
|coauthors=ignored (help) - ↑ Vermeeren A. (2004). "Residual effects of hypnotics: epidemiology and clinical implications". CNS Drugs. 18 (5): 297–328. PMID 15089115.
- ↑ Kales, A (1982). "Dose-response studies of lormetazepam: efficacy, side effects, and rebound insomnia". J Clin Pharmacol. 22 (11–12): 520–530. Retrieved 16/03/07. Unknown parameter
|coauthors=ignored (help); Check date values in:|accessdate=(help) - ↑ Smith AE (1989) Benzodiazepines - Use & Abuse - A GUIDE FOR PRESCRIBERS
- ↑ Gerra, G (1996). "Intravenous flumazenil following prolonged exposure to lormetazepam in humans: lack of precipitated withdrawal". Int Clin Psychopharmacol. 11 (2): 81–88. Retrieved 16/03/07. Unknown parameter
|coauthors=ignored (help); Check date values in:|accessdate=(help) - ↑ Ashton CH PROTRACTED WITHDRAWAL SYMPTOMS FROM BENZODIAZEPINES
- ↑ Ashton CH BENZODIAZEPINES: HOW THEY WORK AND HOW TO WITHDRAW
- ↑ Lader M A pilot study of the effects of flumazenil on symptoms persisting after benzodiazepine withdrawal
- ↑ (Roche Products (UK) Ltd 1990) Benzodiazepines and Your Patients: A Management Programme
External links
- Pages with script errors
- Pages with citations using unsupported parameters
- CS1 errors: dates
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Benzodiazepines
- Sedatives
- Hypnotics
- Anticonvulsants
- Muscle relaxants
- Anxiolytics