Secobarbital sodium
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Stefano Giannoni [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Secobarbital sodium is a barbiturate that is FDA approved for the treatment of insomnia and for preanesthetic use.. Common adverse reactions include somnolence.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Dosages of barbiturates must be individualized with full knowledge of their particular characteristics. Factors of consideration are the patient's age, weight, and condition.
Insomnia
100 mg at bedtime.
- For the short-term treatment of insomnia, since it appears to lose its effectiveness for sleep induction and sleep maintenance after 2 weeks.
Preanesthetic
Preoperatively, 200 to 300 mg, 1 to 2 hours before surgery.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Secobarbital sodium in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Secobarbital sodium in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Preanesthetic
Preoperatively, 2 to 6 mg/kg, with a maximum dosage of 100 mg.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Secobarbital sodium in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Secobarbital sodium in pediatric patients.
Contraindications
- Hypersensitive to barbiturates.
- Patients with a history of manifest or latent porphyria,
- Marked impairment of liver function
- Marked impairment respiratory disease in which dyspnea or obstruction is evident.
Warnings
- Because sleep disturbances may be the presenting manifestation of a physical and/or psychiatric disorder, symptomatic treatment of insomnia should be initiated only after a careful evaluation of the patient.
- The failure of insomnia to remit after 7 to 10 days of treatment may indicate the presence of a primary psychiatric and/or medical illness that should be evaluated.
- Worsening of insomnia or the emergence of new thinking or behavior abnormalities may be the consequence of an unrecognized psychiatric or physical disorder. Such findings have emerged during the course of treatment with sedative-hypnotic drugs.
- Because some of the important adverse effects of sedative-hypnotics appear to be dose related, it is important to use the smallest possible effective dose, especially in the elderly.
- Complex behaviors such as "sleep-driving" (i.e., driving while not fully awake after ingestion of a sedative-hypnotic, with amnesia for the event) have been reported.
- These events can occur in sedative-hypnotic-naïve as well as in sedative-hypnotic-experienced persons. Although behaviors such as sleep-driving may occur with sedative-hypnotics alone at therapeutic doses, the use of alcohol and other CNS depressants with sedative-hypnotics appears to increase the risk of such behaviors, as does the use of sedative-hypnotics at doses exceeding the maximum recommended dose.
- Due to the risk to the patient and the community, discontinuation of sedative-hypnotics should be strongly considered for patients who report a "sleep-driving" episode.
- Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a sedative-hypnotic. As with sleep-driving, patients usually do not remember these events.
Habit-Forming
- Secobarbital may be habit-forming.
- Tolerance and psychological and physical dependence may occur with continued use.
- Patients who have psychological dependence on barbiturates may increase the dosage or decrease the dosage interval without consulting a physician and subsequently may develop a physical dependence on barbiturates. To minimize the possibility of overdosage or development of dependence, the prescribing and dispensing of sedative-hypnotic barbiturates should be limited to the amount required for the interval until the next appointment.
- The abrupt cessation after prolonged use in a person who is dependent on the drug may result in withdrawal symptoms, including delirium, convulsions, and possibly death. Barbiturates should be withdrawn gradually from any patient known to be taking excessive doses over long periods of time.
Acute or Chronic Pain
- Caution should be exercised when barbiturates are administered to patients with acute or chronic pain, because paradoxical excitement could be induced or important symptoms could be masked.
Usage in Pregnancy
- Barbiturates can cause fetal harm when administered to a pregnant woman. *Retrospective, case-controlled studies have suggested that there may be a connection between the maternal consumption of barbiturates and a higher than expected incidence of fetal abnormalities.
- Barbiturates readily cross the placental barrier and are distributed throughout fetal tissues; the highest concentrations are found in the placenta, fetal liver, and brain.
- Fetal blood levels approach maternal blood levels following parenteral administration.
- Withdrawal symptoms occur in infants born to women who receive barbiturates throughout the last trimester of pregnancy.
- If secobarbital sodium is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Synergistic Effects
- The concomitant use of alcohol or other CNS depressants may produce additive CNS-depressant effects.
Adverse Reactions
Clinical Trials Experience
The following adverse reactions and their incidences were compiled from surveillance of thousands of hospitalized patients who received barbiturates. Because such patients may be less aware of some of the milder adverse effects of barbiturates, the incidence of these reactions may be somewhat higher in fully ambulatory patients.
More than 1 in 100 Patients
The most common adverse reaction estimated to occur at a rate of 1 to 3 patients per 100 is the following:
Nervous System
Less than 1 in 100 Patients
Adverse reactions estimated to occur at a rate of less than 1 in 100 patients are listed below, grouped by organ system and by decreasing order of occurrence:
Nervous System
- Agitation
- Confusion
- Hyperkinesia
- Ataxia
- CNS depression
- Nightmares
- Nervousness
- Psychiatric disturbance
- Hallucinations
- Insomnia
- Anxiety
- Dizziness
- Abnormality in thinking
Respiratory System
Cardiovascular System
Digestive System
Other Reported Reactions
- Headache
- Injection site reactions
- Hypersensitivity reactions (angioedema, skin rashes, exfoliative dermatitis)
- Fever
- Liver damage
- Megaloblastic anemia following chronic phenobarbital use
Postmarketing Experience
There is limited information regarding Secobarbital sodium Postmarketing Experience in the drug label.
Drug Interactions
Most reports of clinically significant drug interactions occurring with the barbiturates have involved phenobarbital. However, the application of these data to other barbiturates appears valid and warrants serial blood level determinations of the relevant drugs when there are multiple therapies.
Anticoagulants
- Phenobarbital lowers the plasma levels of dicumarol and causes a decrease in anticoagulant activity as measured by the prothrombin time.
- Barbiturates can induce hepatic microsomal enzymes, resulting in increased metabolism and decreased anticoagulant response of oral anticoagulants (eg, warfarin, acenocoumarol, dicumarol, and phenprocoumon).
- Patients stabilized on anticoagulant therapy may require dosage adjustments if barbiturates are added to or withdrawn from their dosage regimen.
Corticosteroids
- Barbiturates appear to enhance the metabolism of exogenous corticosteroids, probably through the induction of hepatic microsomal enzymes.
- Patients stabilized on corticosteroid therapy may require dosage adjustments if barbiturates are added to or withdrawn from their dosage regimen.
Griseofulvin
- Phenobarbital appears to interfere with the absorption of orally administered griseofulvin, thus decreasing its blood level.
- The effect of the resultant decreased blood levels of griseofulvin on therapeutic response has not been established. However, it would be preferable to avoid concomitant administration of these drugs.
Doxycycline
- Phenobarbital has been shown to shorten the half-life of doxycycline for as long as 2 weeks after barbiturate therapy is discontinued.
- This mechanism is probably through the induction of hepatic microsomal enzymes that metabolize the antibiotic.
- If barbiturates and doxycycline are administered concurrently, the clinical response to doxycycline should be monitored closely.
Phenytoin, Sodium Valproate, Valproic Acid
- The effect of barbiturates on the metabolism of phenytoin appears to be variable.
- Some investigators report an accelerating effect, whereas others report no effect.
- Because the effect of barbiturates on the metabolism of phenytoin is not predictable, phenytoin and barbiturate blood levels should be monitored more frequently if these drugs are given concurrently.
- Sodium valproate and valproic acid increase the secobarbital sodium serum levels; therefore, secobarbital sodium blood levels should be monitored closely and appropriate dosage adjustments made as clinically indicated.
CNS Depressants
- The concomitant use of other CNS depressants, including other sedatives or hypnotics, antihistamines, tranquilizers, or alcohol, may produce additive depressant effects.
Monoamine Oxidase Inhibitors (MAOIs)
- MAOIs prolong the effects of barbiturates, probably because metabolism of the barbiturate is inhibited.
Estradiol, Estrone, Progesterone, and Other Steroidal Hormones
- Pretreatment with or concurrent administration of phenobarbital may decrease the effect of estradiol by increasing its metabolism.
- There have been reports of patients treated with antiepileptic drugs (eg, phenobarbital) who become pregnant while taking oral contraceptives.
- An alternate contraceptive method might be suggested to women taking barbiturates.
Use in Specific Populations
Pregnancy
- Barbiturates can cause fetal harm when administered to a pregnant woman. *Retrospective, case-controlled studies have suggested that there may be a connection between the maternal consumption of barbiturates and a higher than expected incidence of fetal abnormalities.
- Barbiturates readily cross the placental barrier and are distributed throughout fetal tissues; the highest concentrations are found in the placenta, fetal liver, and brain.
- Fetal blood levels approach maternal blood levels following parenteral administration.
- Withdrawal symptoms occur in infants born to women who receive barbiturates throughout the last trimester of pregnancy.
- If secobarbital sodium is used during pregnancy or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
- Teratogenic Effects.
- Nonteratogenic Effects. Reports of infants suffering from long-term barbiturate exposure in utero included the acute withdrawal syndrome of seizures and hyperirritability from birth to a delayed onset of up to 14 days
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Secobarbital sodium in women who are pregnant.
Labor and Delivery
- Hypnotic doses of barbiturates do not appear to impair uterine activity significantly during labor.
- Full anesthetic doses of barbiturates decrease the force and frequency of uterine contractions.
- Administration of sedative-hypnotic barbiturates to the mother during labor may result in respiratory depression in the newborn.
- Premature infants are particularly susceptible to the depressant effects of barbiturates.
- If barbiturates are used during labor and delivery, resuscitation equipment should be available.
- Data are not available to evaluate the effect of barbiturates when forceps delivery or other intervention is necessary or to determine the effect of barbiturates on the later growth, development, and functional maturity of the pediatric patient.
Nursing Mothers
- Caution should be exercised when secobarbital sodium is administered to a nursing woman, because small amounts of barbiturates are excreted in the milk.
Pediatric Use
- Barbiturates repeatedly produce excitement rather than depression.
Geriatic Use
- Dosage should be reduced in the elderly or debilitated because these patients may be more sensitive to barbiturates.
- Elderly or debilitated patients may react to barbiturates with marked excitement, depression, or confusion.
Gender
There is no FDA guidance on the use of Secobarbital sodium with respect to specific gender populations.
Race
There is no FDA guidance on the use of Secobarbital sodium with respect to specific racial populations.
Renal Impairment
- Dosage should be reduced for patients with impaired renal function.
Hepatic Impairment
- Dosage should be reduced for patients with hepatic disease.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Secobarbital sodium in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Secobarbital sodium in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
- Prolonged therapy with barbiturates should be accompanied by periodic laboratory evaluation of organic systems, including hematopoietic, renal, and hepatic systems
IV Compatibility
There is limited information regarding the compatibility of Secobarbital sodium and IV administrations.
Overdosage
- The toxic dose of barbiturates varies considerably.
- In general, an oral dose of 1 g of most barbiturates produces serious poisoning in an adult.
- Death commonly occurs after 2 to 10 g of ingested barbiturate.
- The sedated, therapeutic blood levels of secobarbital range between 0.5 to 5 mcg/mL; the usual lethal blood level ranges from 15 to 40 mcg/mL.
- Barbiturate intoxication may be confused with alcoholism, bromide intoxication, and various neurologic disorders.
- Potential tolerance must be considered when evaluating significance of dose and plasma concentration.
Signs and Symptoms
- Symptoms of oral overdose may occur within 15 minutes and begin with central nervous system depression, underventilation, hypotension, and hypothermia, which may progress to pulmonary edema and death.
- Hemorrhagic blisters may develop, especially at pressure points.
- In extreme overdose, all electrical activity in the brain may cease, in which case a "flat" EEG normally equated with clinical death cannot be accepted as indicative of brain death.
- This effect is fully reversible unless hypoxic damage occurs.
- Consideration should be given to the possibility of barbiturate intoxication even in situations that appear to involve trauma.
- Complications such as pneumonia, pulmonary edema, cardiac arrhythmias, congestive heart failure, and renal failure may occur.
- Uremia may increase CNS sensitivity to barbiturates if renal function is impaired.
- Differential diagnosis should include hypoglycemia, head trauma, cerebrovascular accidents, convulsive states, and diabetic coma.
Treatment
- To obtain up-to-date information about the treatment of overdose, a good resource is your certified Regional Poison Control Center.
- In managing overdosage, consider the possibility of multiple drug overdoses, interaction among drugs, and unusual drug kinetics in your patient.
- Protect the patient's airway and support ventilation and perfusion. *Meticulously monitor and maintain, within acceptable limits, the patient's vital signs, blood gases, serum electrolytes, etc.
- Absorption of drugs from the gastrointestinal tract may be decreased by giving activated charcoal, which, in many cases, is more effective than emesis or lavage; consider charcoal instead of or in addition to gastric emptying. *Repeated doses of charcoal over time may hasten elimination of some drugs that have been absorbed.
- Safeguard the patient's airway when employing gastric emptying or charcoal.
- Diuresis and peritoneal dialysis are of little value; hemodialysis and hemoperfusion enhance drug clearance and should be considered in serious poisoning.
- If the patient has chronically abused sedatives, withdrawal reactions may be manifest following acute overdose.
Pharmacology
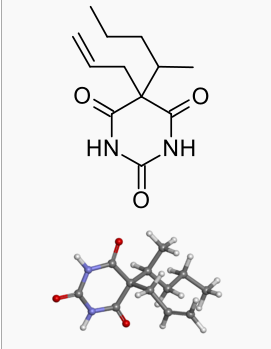 | |
| File:Secobarbital ball-and-stick.png | |
| Clinical data | |
|---|---|
| Trade names | secobarbital |
| AHFS/Drugs.com | Consumer Drug Information |
| MedlinePlus | a682386 |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ? |
| Protein binding | 45-60% |
| Metabolism | Hepatic |
| Elimination half-life | 15-40 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C12H18N2O3 |
| Molar mass | 238.283 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Mechanism of Action
- The barbiturates are nonselective central nervous system (CNS) depressants that are primarily used as sedative hypnotics. In subhypnotic doses, they are also used as anticonvulsants.
- Barbiturates are capable of producing all levels of CNS mood alteration, from excitation to mild sedation, hypnosis, and deep coma. Overdosage can produce death.
- In high enough therapeutic doses, barbiturates induce anesthesia.
- Barbiturates depress the sensory cortex, decrease motor activity, alter cerebellar function, and produce drowsiness, sedation, and hypnosis.
- Barbiturate-induced sleep differs from physiologic sleep.
- Sleep laboratory studies have demonstrated that barbiturates reduce the amount of time spent in the rapid eye movement (REM) phase, or dreaming stage of sleep. Also, Stages III and IV sleep are decreased. Following abrupt cessation of regularly used barbiturates, patients may experience markedly increased dreaming, nightmares, and/or insomnia. Therefore, withdrawal of a single therapeutic dose over 5 or 6 days has been recommended to lessen the REM rebound and disturbed sleep that contribute to drug withdrawal syndrome (for example, decreasing the dose from 3 to 2 doses a day for 1 week).
- In studies, secobarbital sodium and pentobarbital sodium have been found to lose most of their effectiveness for both inducing and maintaining sleep by the end of 2 weeks of continued drug administration, even with the use of multiple doses.
- As with secobarbital sodium and pentobarbital sodium, other barbiturates (including amobarbital) might be expected to lose their effectiveness for inducing and maintaining sleep after about 2 weeks.
- The short-, intermediate-, and to a lesser degree, long-acting barbiturates have been widely prescribed for treating insomnia.
- Although the clinical literature abounds with claims that the short-acting barbiturates are superior for producing sleep whereas the intermediate-acting compounds are more effective in maintaining sleep, controlled studies have failed to demonstrate these differential effects.
- Therefore, as sleep medications, the barbiturates are of limited value beyond short-term use.
- Barbiturates have little analgesic action at subanesthetic doses.
- Rather, in subanesthetic doses, these drugs may increase the reaction to painful stimuli.
- All barbiturates exhibit anticonvulsant activity in anesthetic doses. However, of the drugs in this class, only phenobarbital, mephobarbital, and metharbital are effective as oral anticonvulsants in subhypnotic doses.
- Barbiturates are respiratory depressants, and the degree of depression is dependent on the dose.
- With hypnotic doses, respiratory depression is similar to that which occurs during physiologic sleep accompanied by a slight decrease in blood pressure and heart rate.
- Studies in laboratory animals have shown that barbiturates cause reduction in the tone and contractility of the uterus, ureters, and urinary bladder. However, concentrations of the drugs required to produce this effect in humans are not reached with sedative-hypnotic doses.
- Barbiturates do not impair normal hepatic function, but have been shown to induce liver microsomal enzymes, thus increasing and/or altering the metabolism of barbiturates and other drugs.
Structure
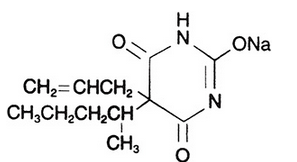
Secobarbital sodium (secobarbital sodium Capsules, USP) is a barbituric acid derivative and occurs as a white, odorless, bitter powder that is very soluble in water, soluble in alcohol, and practically insoluble in ether. Chemically, the drug is sodium 5-allyl-5-(1-methylbutyl) barbiturate, with the molecular formula C12H17N2NaO3. Its molecular weight is 260.27. The structural formula is as follows:
Pharmacodynamics
There is limited information regarding Secobarbital sodium Pharmacodynamics in the drug label.
Pharmacokinetics
- Barbiturates are absorbed in varying degrees following oral or parenteral administration.
- The salts are more rapidly absorbed than are the acids.
- The rate of absorption is increased if the sodium salt is ingested as a dilute solution or taken on an empty stomach.
- Duration of action, which is related to the rate at which the barbiturates are redistributed throughout the body, varies among persons and in the same person from time to time.
- Secobarbital sodium is classified as a short-acting barbiturate when taken orally. *Its onset of action is 10 to 15 minutes and its duration of action ranges from 3 to 4 hours.
- Barbiturates are weak acids that are absorbed and rapidly distributed to all tissues and fluids, with high concentrations in the brain, liver, and kidneys.
- Lipid solubility of the barbiturates is the dominant factor in their distribution within the body.
- The more lipid soluble the barbiturate, the more rapidly it penetrates all tissues of the body.
- Barbiturates are bound to plasma and tissue proteins to a varying degree, with the degree of binding increasing directly as a function of lipid solubility.
- Phenobarbital has the lowest lipid solubility, lowest plasma binding, lowest brain protein binding, the longest delay in onset of activity, and the longest duration of action.
- At the opposite extreme is secobarbital, which has the highest lipid solubility, highest plasma protein binding, highest brain protein binding, the shortest delay in onset of activity, and the shortest duration of action.
- The plasma half-life for secobarbital sodium in adults ranges between 15 to 40 hours, with a mean of 28 hours.
- No data are available for pediatric patients and newborns.
- Barbiturates are metabolized primarily by the hepatic microsomal enzyme system, and the metabolic products are excreted in the urine and, less commonly, in the feces.
- The excretion of unmetabolized barbiturate is 1 feature that distinguishes the long-acting category from those belonging to other categories, which are almost entirely metabolized.
- The inactive metabolites of the barbiturates are excreted as conjugates of glucuronic acid.
Nonclinical Toxicology
There is limited information regarding Secobarbital sodium Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Secobarbital sodium Clinical Studies in the drug label.
How Supplied
- Secobarbital sodium capsules are orange and imprinted with RX679 on both the cap and the body:
NDC 42998-679-01 100 mg Bottles of 100
Storage
- Store at 20° - 25° C (68° - 77° F).
- Dispense in a tight container.
Images
Drug Images
{{#ask: Page Name::Secobarbital sodium |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
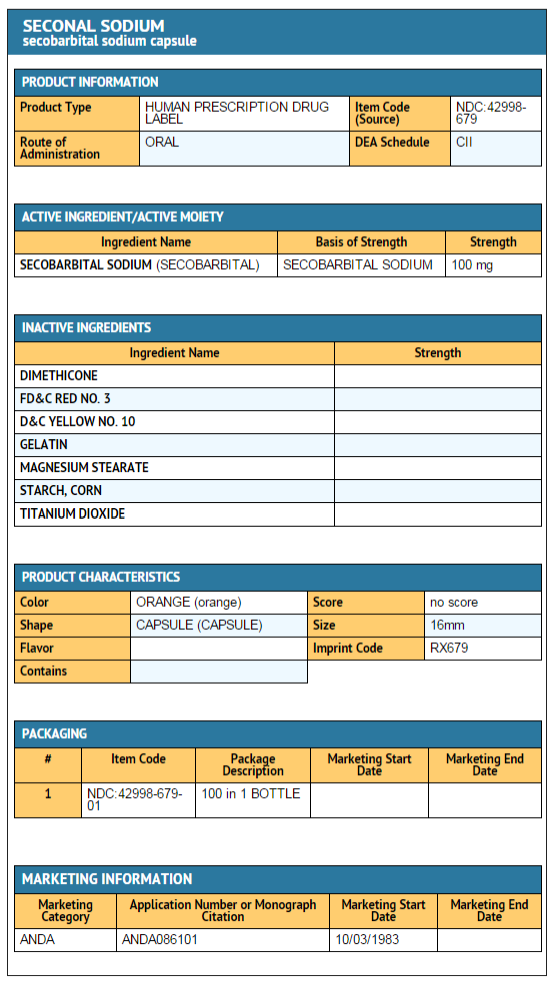
{{#ask: Label Page::Secobarbital sodium |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Sleep-Driving" and other complex behaviors
- There have been reports of people getting out of bed after taking a sedative-hypnotic and driving their cars while not fully awake, often with no memory of the event.
- If a patient experiences such an episode, it should be reported to his or her doctor immediately, since "sleep-driving" can be dangerous.
- This behavior is more likely to occur when sedative-hypnotics are taken with alcohol or other central nervous system depressants.
- Other complex behaviors (e.g., preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a sedative-hypnotic.
- As with sleep-driving, patients usually do not remember these events.
The following information should be given to patients receiving secobarbital sodium:
- The use of secobarbital sodium carries with it an associated risk of psychological and/or physical dependence.
- The patient should be warned against increasing the dose of the drug without consulting a physician.
- Secobarbital sodium may impair the mental and/or physical abilities required for the performance of potentially hazardous tasks, such as driving a car or operating machinery.
- The patient should be cautioned accordingly.
- Alcohol should not be consumed while taking secobarbital sodium.
- The concurrent use of secobarbital sodium with other CNS depressants (eg, alcohol, narcotics, tranquilizers, and antihistamines) may result in additional CNS-depressant effects.
Precautions with Alcohol
- Alcohol should not be consumed while taking secobarbital sodium.
- The concurrent use of secobarbital sodium with other CNS depressants (eg, alcohol, narcotics, tranquilizers, and antihistamines) may result in additional CNS-depressant effects.
Brand Names
- Seconal[1]
Look-Alike Drug Names
There is limited information regarding Secobarbital sodium Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Secobarbital sodium |Label Name=Serobarbital pack.png
}}