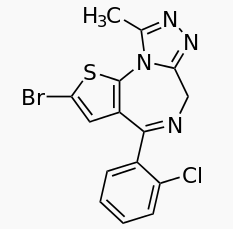
Brotizolam
 | |
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 48-95% |
| Metabolism | Hepatic |
| Elimination half-life | 4.4 hours (range, 2.6–6.9 h) |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C15H10BrClN4S |
| Molar mass | 393.7 g/mol |
|
WikiDoc Resources for Brotizolam |
|
Articles |
|---|
|
Most recent articles on Brotizolam |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Brotizolam at Clinical Trials.gov Clinical Trials on Brotizolam at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Brotizolam
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Brotizolam Discussion groups on Brotizolam Patient Handouts on Brotizolam Directions to Hospitals Treating Brotizolam Risk calculators and risk factors for Brotizolam
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Brotizolam |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Brotizolam (marketed under brand name Lendormin) is a drug which is thienobenzodiazepine (a benzodiazepine derivative). It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties, and is considered to be similar in effect to short-acting benzodiazepines such as triazolam. It is used in the short term treatment of insomnia although due to its short half life it is considered to have relatively high abuse potential and so would not be a first-line treatment. Brotizolam is a potent drug with a dosage of between 0.5 and 1.5 milligrams, but is rapidly eliminated with an average half life of 4.4 hours (range 3.6 - 7.9 hours).
Brotizolam is not approved for sale in the UK, United States or Canada.
Pharmacology
Brotizolam induces impairment of motor function and has hypnotic properties.[1] Brotizolam increases the slow wave light sleep (SWLS) in a dose-dependent manner whilst suppressing deep sleep stages. Less time is spent in stages 3 and 4 which are the deep sleep stages when benzodiazepines such as brotizolam are used. Benzodiazepines are therefore not good hypnotics in the treatment of insomnia. The suppression of deep sleep stages by benzodiazepines may be especially problematic to the elderly as they naturally spend less time in the deep sleep stage.[2]
Indications
Insomnia. Brotizolam is prescribed for the short term treatment, 2 - 4 weeks only of severe insomnia. Insomnia can be described as a difficulty falling asleep, frequent awakening, early awakenings or a combination of each. Brotizolam is a short-intermediate acting benzodiazepine and is sometimes used in patients who have difficulty in maintaining sleep or getting to sleep. Hypnotics should only be used on a short term basis or in those with chronic insomnia on an occasional basis.[3]
See also
Notes
- ↑ Yasui M (2005). "[Pharmacological profiles of benzodiazepinergic hypnotics and correlations with receptor subtypes]". 25 (3): 143–51. PMID 16045197. Text "Nihon Shinkei Seishin Yakurigaku Zasshi. " ignored (help); Unknown parameter
|coauthors=ignored (help); Unknown parameter|month=ignored (help) - ↑ Noguchi H (2004). "Electroencephalographic properties of zaleplon, a non-benzodiazepine sedative/hypnotic, in rats" (pdf). J Pharmacol Sci. 94 (3): 246–51. PMID 15037809. Unknown parameter
|coauthors=ignored (help); Unknown parameter|month=ignored (help) - ↑ Rickels K. (1986). "The clinical use of hypnotics: indications for use and the need for a variety of hypnotics". Acta Psychiatr Scand Suppl. 332: 132–41. PMID 2883820.
References
{{Reflist|2]]
- Greenblatt DJ, Locniskar A, Shader RI. Pilot pharmacokinetic study of brotizolam, a thienodiazepine hypnotic, using electron-capture gas-liquid chromatography. Sleep. 1983;6(1):72-6. PMID 6844800.
- Langley MS, Clissold SP. Brotizolam. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy as an hypnotic. Drugs. 1988 Feb;35(2):104-22.
- Bechtel WD. Pharmacokinetics and metabolism of brotizolam in humans. British Journal of Clinical Pharmacology. 1983;16 Suppl 2:279S-283S.
- Jochemsen R. Pharmacokinetics of brotizolam in healthy subjects following intravenous and oral administration. British Journal of Clinical Pharmacology. 1983;16 Suppl 2:285S-290S.
External links
- Pages with script errors
- Pages with citations using unnamed parameters
- Pages with citations using unsupported parameters
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Drug
- Benzodiazepines
- Hypnotics