G protein-coupled receptor
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
G protein-coupled receptors (GPCRs), also known as seven transmembrane domain receptors, 7TM receptors, heptahelical receptors, and G protein-linked receptors (GPLR), comprise a large protein family of transmembrane receptors that sense molecules outside the cell and activate inside signal transduction pathways and, ultimately, cellular responses. G protein-coupled receptors are found only in eukaryotes, including yeast, plants, choanoflagellates,[1] and animals. The ligands that bind and activate these receptors include light-sensitive compounds, odors, pheromones, hormones, and neurotransmitters, and vary in size from small molecules to peptides to large proteins. G protein-coupled receptors are involved in many diseases, but are also the target of around half of all modern medicinal drugs.[2]
Classification
GPCRs can be grouped into 6 classes based on sequence homology and functional similarity:[3][4][5][6]
- Class A (or 1) (Rhodopsin-like)
- Class B (or 2) (Secretin receptor family)
- Class C (or 3) (Metabotropic glutamate/pheromone)
- Class D (or 4) (Fungal mating pheromone receptors)
- Class E (or 5) (Cyclic AMP receptors)
- Class F (or 6) (Frizzled/Smoothened)
The very large rhodopsin A group has been further subdivided into 19 subgroups (A1-A19).[7] More recently, an alternative classification system called GRAFS (Glutamate, Rhodopsin, Adhesion, Frizzled/Taste2, Secretin) has been proposed.[8]
Physiological roles
GPCRs are involved in a wide variety of physiological processes. Some examples of their physiological roles include:
- the visual sense: the opsins use a photoisomerization reaction to translate electromagnetic radiation into cellular signals. Rhodopsin, for example, uses the conversion of 11-cis-retinal to all-trans-retinal for this purpose
- the sense of smell: receptors of the olfactory epithelium bind odorants (olfactory receptors) and pheromones (vomeronasal receptors)
- behavioral and mood regulation: receptors in the mammalian brain bind several different neurotransmitters, including serotonin, dopamine, GABA, and glutamate
- regulation of immune system activity and inflammation: chemokine receptors bind ligands that mediate intercellular communication between cells of the immune system; receptors such as histamine receptors bind inflammatory mediators and engage target cell types in the inflammatory response
- autonomic nervous system transmission: both the sympathetic and parasympathetic nervous systems are regulated by GPCR pathways, responsible for control of many automatic functions of the body such as blood pressure, heart rate, and digestive processes
- cell density sensing: A novel GPCR role in regulating cell density sensing.
Receptor structure
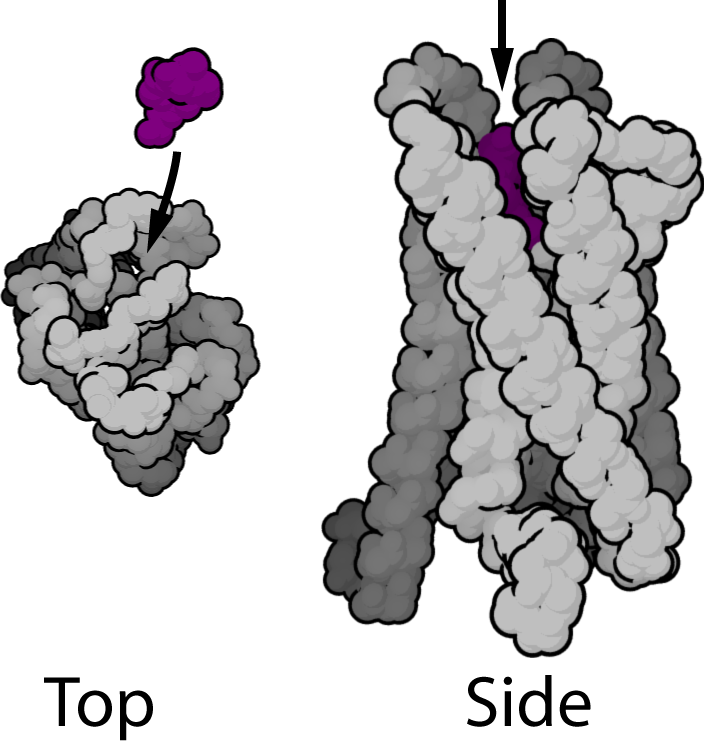
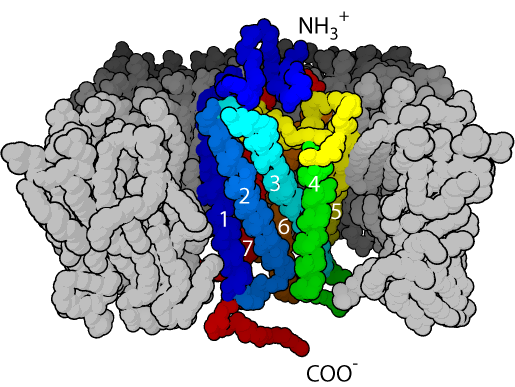
GPCRs are integral membrane proteins that possess seven membrane-spanning domains or transmembrane helices (Figure 1). The extracellular parts of the receptor can be glycosylated. These extracellular loops also contain two highly-conserved cysteine residues that form disulfide bonds to stabilize the receptor structure. Some seven transmembrane helix proteins (such as channelrhodopsin) that resemble GPCRs may contain different functional groups, such as entire ion channels, within their protein.
Early structural models for GPCRs were based on their weak analogy to bacteriorhodopsin for which a structure had been determined by both electron diffraction (PDB 2BRD, 1AT9) [9][10] and X ray-based crystallography (1AP9).[11] In 2000, the first crystal structure of a mammalian GPCR, that of bovine rhodopsin (1F88), was solved.[12] While the main feature, the seven transmembrane helices, is conserved, the relative orientation of the helices differ significantly from that of bacteriorhodopsin. In 2007, the first structure of a human GPCR was solved (2R4R, 2R4S).[13] This was followed immediately by a higher resolution structure of the same receptor (2RH1).[14][15] This human β2-adrenergic receptor GPCR structure, proved to be highly similar to the bovine rhodopsin in terms of the relative orientation of the seven transmembrane helices. However the conformation of the second extracellular loop is entirely different between the two structures. Since this loop constitutes the "lid" that covers the top of the ligand binding site, this conformational difference highlights the difficulties in constructing homology models of other GPCRs based only on the rhodopsin structure.
Mechanism
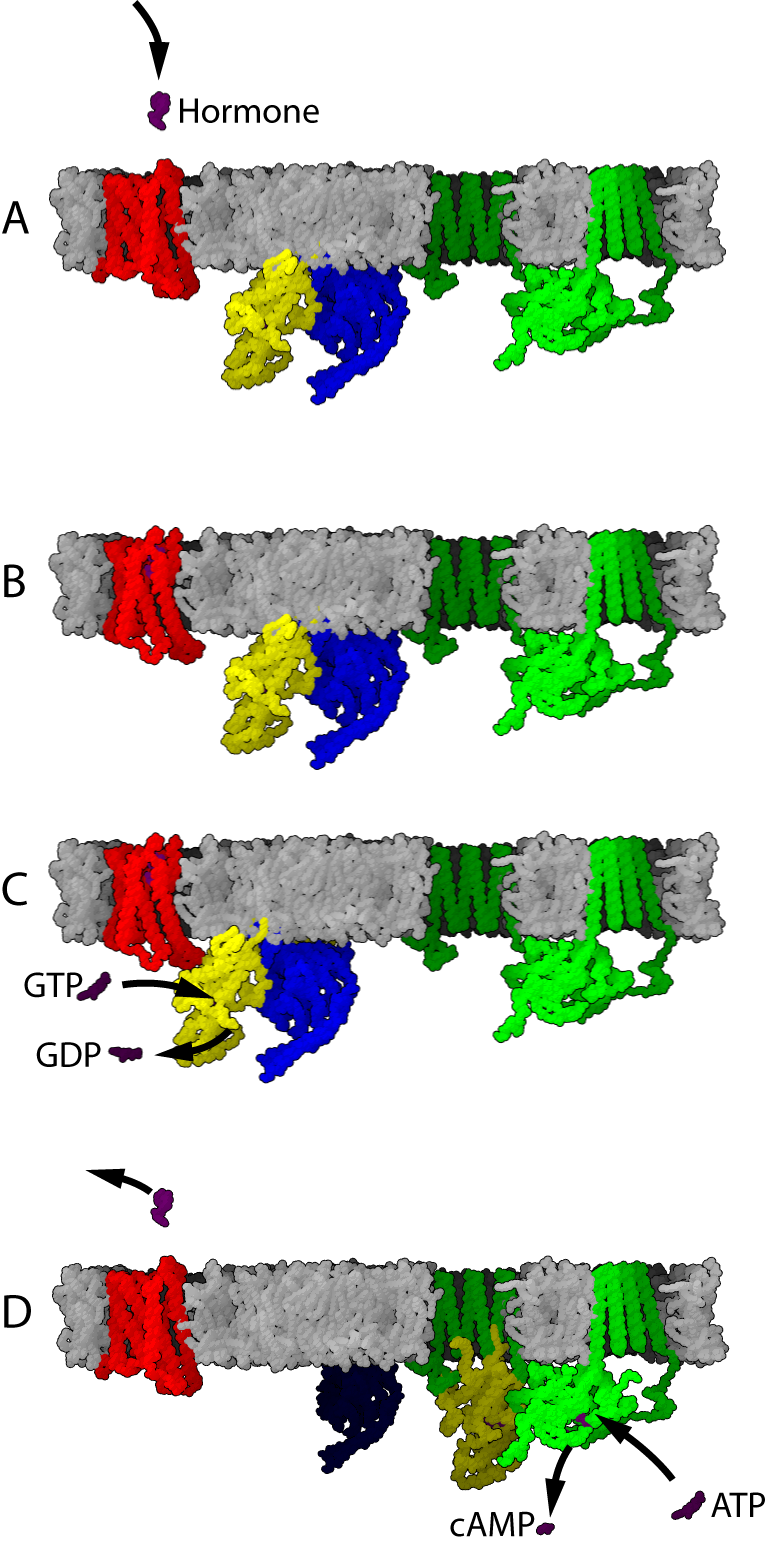
G protein-coupled receptor are activated by an external signal in the form of a ligand or other signal mediator. This creates a conformational change in the receptor, causing activation of a G protein. Further effect depends on the type of G protein.
Ligand binding
GPCRs include receptors for sensory signal mediators (e.g., light and olfactory stimulatory molecules); adenosine, bombesin, bradykinin, endothelin, γ-aminobutyric acid (GABA), hepatocyte growth factor, melanocortins, neuropeptide Y, opioid peptides, opsins, somatostatin, tachykinins, vasoactive intestinal polypeptide family, and vasopressin; biogenic amines (e.g., dopamine, epinephrine, norepinephrine, histamine, glutamate (metabotropic effect), glucagon, acetylcholine (muscarinic effect), and serotonin); chemokines; lipid mediators of inflammation (e.g., prostaglandins, prostanoids, platelet-activating factor, and leukotrienes); and peptide hormones (e.g., calcitonin, C5a anaphylatoxin, follicle-stimulating hormone (FSH), gonadotropic-releasing hormone (GnRH), neurokinin, thyrotropin-releasing hormone (TRH), and oxytocin). GPCRs that act as receptors for stimuli that have yet to be identified are known as orphan receptors.
Whereas, in other types of receptors that have been studied, ligands bind externally to the membrane, the ligands of GPCRs typically bind within the transmembrane domain.
Conformational change
The transduction of the signal through the membrane by the receptor is not completely understood. It is known that the inactive G protein is bound to the receptor in its inactive state. Once the ligand is recognized, the receptor shifts conformation and thus mechanically activates the G protein, which detaches from the receptor. The receptor can now either activate another G protein or switch back to its inactive state. This is an overly simplistic explanation, but suffices to convey the overall set of events.
It is believed that a receptor molecule exists in a conformational equilibrium between active and inactive biophysical states.[16] The binding of ligands to the receptor may shift the equilibrium toward the active receptor states.[17] Three types of ligands exist: agonists are ligands that shift the equilibrium in favour of active states; inverse agonists are ligands that shift the equilibrium in favour of inactive states; and neutral antagonists are ligands that do not affect the equilibrium. It is not yet known how exactly the active and inactive states differ from each other.
Activation of G protein
Template:Seealso If a receptor in an active state encounters a G protein, it may activate it (Figure 2, blue protein in part B). Some evidence suggests that receptors and G proteins are actually pre-coupled. For example, binding of G proteins to receptors affects the receptor's affinity for ligands. Activated G proteins are bound to GTP.
Further signal transduction depends on the type of G protein. The enzyme adenylate cyclase (Figure 2, green protein in panel C) is an example of a cellular protein that can be regulated by a G protein, in this case the G protein Gs. Adenylate cyclase activity is activated when it binds to a subunit of the activated G protein (Figure 2, Panel D). Activation of adenylate cyclase ends when the G protein returns to the GDP-bound state (Figure 2, panels E and A).
GPCR signaling without G proteins
In the late 1990s, evidence began accumulating to suggest that some GPCRs are able to signal without G proteins. The ERK2 mitogen-activated protein kinase, a key signal transduction mediator downstream of receptor activation in many pathways, has been shown to be activated in response to cAMP-mediated receptor activation in the slime mold D. discoideum despite the absence of the associated G protein α- and β-subunits.
In mammalian cells, the much-studied β2-adrenoceptor has been demonstrated to activate the ERK2 pathway after arrestin-mediated uncoupling of G-protein-mediated signaling. It therefore seems likely that some mechanisms previously believed to be purely related to receptor desensitisation are actually examples of receptors switching their signaling pathway rather than simply being switched off.
In kidney cells, the bradykinin receptor B2 has been shown to interact directly with a protein tyrosine phosphatase. The presence of a tyrosine-phosphorylated ITIM (immunoreceptor tyrosine-based inhibitory motif) sequence in the B2 receptor is necessary to mediate this interaction and subsequently the antiproliferative effect of bradykinin.[18]
Receptor regulation
GPCRs become desensitized when exposed to their ligand for a prolonged period of time. There are two recognized forms of desensitization: 1) homologous desensitization, in which the activated GPCR is downregulated; and 2) heterologous desensitization, wherein the activated GPCR causes downregulation of a different GPCR. The key reaction of this downregulation is the phosphorylation of the intracellular (or cytoplasmic) receptor domain by protein kinases.
Phosphorylation by cAMP-dependent protein kinases
Cyclic AMP-dependent protein kinases (protein kinase A) are activated by the signal chain coming from the G protein (that was activated by the receptor) via adenylate cyclase and cyclic AMP (cAMP). In a feedback mechanism, these activated kinases phosphorylate the receptor. The longer the receptor remains active, the more kinases are activated, the more receptors are phosphorylated. In β2-adrenoceptors, this phosphorylation results in the switching of the coupling from the Go class of G-protein to the Gi class. cAMP-dependent PKA mediated phosphorylation is also known as heterologous desensitisation, because it is not specific to ligand bound receptor. In fact any receptor causing in a increase in PKA activity will cause increased amounts of this type of desensitisation of other receptors coupled to Go (e.g., dopamine receptor D2 activation may lead to β2-adrenoceptor desensitisation of this type).[19]
Phosphorylation by GRKs
The G protein-coupled receptor kinases (GRKs) are protein kinases that phosphorylate only active GPCRs.
Phosphorylation of the receptor can have two consequences:
- Translocation: The receptor is, along with the part of the membrane it is embedded in, brought to the inside of the cell, where it is dephosphorylated within the acidic vesicular environment[20] and then brought back. This mechanism is used to regulate long-term exposure, for example, to a hormone, by allowing resensitisation to follow desensitisation. Alternatively, the receptor may undergo lysozomal degradation, or remain internalised, where it is thought to participate in the initiation of signalling events, the nature of which depend on the internalised vesicle's subcellular localisation.[19]
- Arrestin linking: The phosphorylated receptor can be linked to arrestin molecules that prevent it from binding (and activating) G proteins, effectively switching it off for a short period of time. This mechanism is used, for example, with rhodopsin in retina cells to compensate for exposure to bright light. In many cases, arrestin binding to the receptor is a prerequisite for translocation. For example, beta-arrestin bound to β2-adrenoreceptors acts as an adaptor for binding with clathrin, and with the beta-subunit of AP2 (clathrin adaptor molecules); thus the arrestin here acts as a scaffold assembling the componenets needed for clathrin-mediated endocytosis of β2-adrenoreceptors.[21][22]
Receptor oligomerization
It is generally accepted that G-protein-coupled receptors can form homo- and/or heterodimers and possibly more complex oligomeric structures, and indeed heterodimerization has been shown to be essential for the function of receptors such as the metabotropic GABA(B) receptors. However, it is presently unproven that true heterodimers exist. Present biochemical and physical techniques lack the resolution to differentiate between distinct homodimers assembled into an oligomer or true 1:1 heterodimers. It is also unclear what the functional significance of oligomerization might be, although it is thought that the phenomenon may contribute to the pharmacological heterogeneity of GPCRs in a manner not previously anticipated. This is an actively-studied area in GPCR research.
The best studied example of receptor oligomerisation are the metabotropic GABAB receptors. These receptors are formed by heterodimerization of GABABR1 and GABABR2 subunits. Expression of the GABABR1 without the GABABR2 in heterologous systems leads to retention of the subunit in the endoplasmic reticulum. Expression of the GABABR2 subunit alone, meanwhile, leads to surface expression of the subunit, although with no functional activity (i.e., the receptor does not bind agonist and cannot initiate a response following exposure to agonist). Expression of the two subunits together leads to plasma membrane expression of functional receptor. It has been shown that GABABR2 binding to GABABR1 causes masking of a retention signal[23] of functional receptors.[24]
Plants
GCR2 is a G-protein-coupled receptor for the plant hormone abscisic acid that has been identified in Arabidopsis thaliana. Another putative receptor is GCR1 for which no ligand has been identified yet.[25]
Dictyostelium
A novel GPCR containing a lipid kinase domain has recently been identified in Dictyostelium that regulates cell density sensing.[26]
References
- ↑ King N, Hittinger CT, Carroll SB (2003). "Evolution of key cell signaling and adhesion protein families predates animal origins". Science. 301 (5631): 361–3. doi:10.1126/science.1083853. PMID 12869759.
- ↑ Filmore, David (2004). "It's a GPCR world". Modern Drug Discovery. American Chemical Society. 2004 (November): 24–28.
- ↑ Attwood TK, Findlay JB (1994). "Fingerprinting G-protein-coupled receptors". Protein Eng. 7 (2): 195–203. PMID 8170923.
- ↑ Kolakowski LF Jr (1994). "GCRDb: a G-protein-coupled receptor database". Receptors Channels. 2 (1): 1–7. PMID 8081729.
- ↑ Foord SM, Bonner TI, Neubig RR, Rosser EM, Pin JP, Davenport AP, Spedding M, Harmar AJ (2005). "International Union of Pharmacology. XLVI. G protein-coupled receptor list". Pharmacol Rev. 57 (2): 279–88. doi:doi:10.1124/pr.57.2.5 Check
|doi=value (help). PMID 15914470. - ↑ InterPro
- ↑ Joost P, Methner A (2002). "Phylogenetic analysis of 277 human G-protein-coupled receptors as a tool for the prediction of orphan receptor ligands". Genome Biol. 3 (11): research0063.1–0063.16. doi:10.1186/gb-2002-3-11-research0063. PMID 12429062.
- ↑ Bjarnadottir TK, Gloriam DE, Hellstrand SH, Kristiansson H, Fredriksson R, Schioth HB (2006). "Comprehensive repertoire and phylogenetic analysis of the G protein-coupled receptors in human and mouse". Genomics. 88 (3): 263–73. doi:10.1016/j.ygeno.2006.04.001. PMID 16753280.
- ↑ Grigorieff N, Ceska TA, Downing KH, Baldwin JM, Henderson R (1996). "Electron-crystallographic refinement of the structure of bacteriorhodopsin". J. Mol. Biol. 259 (3): 393–421. doi:10.1006/jmbi.1996.0328. PMID 8676377.
- ↑ Kimura Y, Vassylyev DG, Miyazawa A, Kidera A, Matsushima M, Mitsuoka K, Murata K, Hirai T, Fujiyoshi Y (1997). "Surface of bacteriorhodopsin revealed by high-resolution electron crystallography". Nature. 389 (6647): 206–11. doi:10.1038/38323. PMID 9296502.
- ↑ Pebay-Peyroula E, Rummel G, Rosenbusch JP, Landau EM (1997). "X-ray structure of bacteriorhodopsin at 2.5 angstroms from microcrystals grown in lipidic cubic phases". Science. 277 (5332): 1676–81. doi:10.1126/science.277.5332.1676. PMID 9287223.
- ↑ Palczewski K, Kumasaka T, Hori T, Behnke CA, Motoshima H, Fox BA, Trong IL, Teller DC, Okada T, Stenkamp RE, Yamamoto M, Miyano M (2000). "Crystal structure of rhodopsin: A G protein-coupled receptor". Science. 289 (5480): 739–45. doi:10.1126/science.289.5480.739. PMID 10926528.
- ↑ Rasmussen SG, Choi HJ, Rosenbaum DM, Kobilka TS, Thian FS, Edwards PC, Burghammer M, Ratnala VR, Sanishvili R, Fischetti RF, Schertler GF, Weis WI, Kobilka BK (2007). "Crystal structure of the human β2-adrenergic G-protein-coupled receptor". Nature. 450 (7168): 383–7. doi:10.1038/nature06325. PMID 17952055.
- ↑ Cherezov V, Rosenbaum DM, Hanson MA, Rasmussen SG, Thian FS, Kobilka TS, Choi HJ, Kuhn P, Weis WI, Kobilka BK, Stevens RC (2007). "High-resolution crystal structure of an engineered human β2-adrenergic G protein-coupled receptor". Science. 318 (5854): 1258–65. doi:10.1126/science.1150577. PMID 17962520.
- ↑ Rosenbaum DM, Cherezov V, Hanson MA, Rasmussen SG, Thian FS, Kobilka TS, Choi HJ, Yao XJ, Weis WI, Stevens RC, Kobilka BK (2007). "GPCR engineering yields high-resolution structural insights into β2-adrenergic receptor function". Science. 318 (5854): 1266–73. doi:10.1126/science.1150609. PMID 17962519.
- ↑ Rubenstein, Lester A. and Lanzara, Richard G. (1998). "Activation of G protein-coupled receptors entails cysteine modulation of agonist binding". Journal of Molecular Structure (Theochem). 430: 57–71.
- ↑ http://www.bio-balance.com/
- ↑ Duchene J, Schanstra JP, Pecher C, Pizard A, Susini C, Esteve JP, Bascands JL, Girolami JP (2002). "A novel protein-protein interaction between a G protein-coupled receptor and the phosphatase SHP-2 is involved in bradykinin-induced inhibition of cell proliferation". J Biol Chem. 277 (43): 40375–83. doi:10.1074/jbc.M202744200. PMID 12177051.
- ↑ 19.0 19.1 Tan CM, Brady AE, Nickols HH, Wang Q, Limbird LE (2004). "Membrane trafficking of G protein-coupled receptors". Annu. Rev. Pharmacol. Toxicol. 44: 559–609. doi:10.1146/annurev.pharmtox.44.101802.121558. PMID 14744258.
- ↑ Krueger KM, Daaka Y, Pitcher JA, Lefkowitz RJ (1997). "The role of sequestration in G protein-coupled receptor resensitization. Regulation of β2-adrenergic receptor dephosphorylation by vesicular acidification". J. Biol. Chem. 272 (1): 5–8. doi:10.1074/jbc.272.1.5. PMID 8995214.
- ↑ Laporte SA, Oakley RH, Holt JA, Barak LS, Caron MG (2000). "The interaction of β-arrestin with the AP-2 adaptor is required for the clustering of β2-adrenergic receptor into clathrin-coated pits". J. Biol. Chem. 275 (30): 23120–6. doi:10.1074/jbc.M002581200. PMID 10770944.
- ↑ Laporte SA, Oakley RH, Zhang J, Holt JA, Ferguson SS, Caron MG, Barak LS (1999). "The beta2-adrenergic receptor/betaarrestin complex recruits the clathrin adaptor AP-2 during endocytosis". Proc. Natl. Acad. Sci. U.S.A. 96 (7): 3712–7. doi:10.1073/pnas.96.7.3712. PMID 10097102.
- ↑ Margeta-Mitrovic M, Jan YN, Jan LY (2000). "A trafficking checkpoint controls GABA(B) receptor heterodimerization". Neuron. 27 (1): 97–106. doi:10.1016/S0896-6273(00)00012-X. PMID 10939334.
- ↑ White JH, Wise A, Main MJ, Green A, Fraser NJ, Disney GH, Barnes AA, Emson P, Foord SM, Marshall FH (1998). "Heterodimerization is required for the formation of a functional GABA(B) receptor". Nature. 396 (6712): 679–82. doi:10.1038/25354. PMID 9872316.
- ↑ Liu X, Yue Y, Li B, Nie Y, Li W, Wu WH, Ma L (2007). "A G protein-coupled receptor is a plasma membrane receptor for the plant hormone abscisic acid". Science. 315 (5819): 712–6. doi:10.1126/science.1135882. PMID 17347412.
- ↑ Bakthavatsalam D, Brazill D, Gomer RH, Eichinger L, Rivero F, Noegel AA (2007). "A G protein-coupled receptor with a lipid kinase domain is involved in cell-density sensing". Curr Biol. 17 (10): 892–7. doi:10.1016/j.cub.2007.04.029. PMID 17481898.
See also
External links
- A phylogenetic tree of all human GPCRs showing family relationships from Vassilatis, et al.[1]
- Reference for molecular and mathematical models for the initial receptor response
- IUPHAR GPCR Database
- G Protein-Coupled Receptor Database (GPCRDB)
- G-protein-coupled+receptors at the US National Library of Medicine Medical Subject Headings (MeSH)
- Wikipedia:MeSH D12.776#MeSH D12.776.543.750.100 --- receptors.2C g-protein-coupled
- UMich Orientation of Proteins in Membranes families/superfamily-6
ar:مستقبل مقترن بالبروتين ج de:G-Protein-gekoppelter Rezeptor it:Recettori accoppiati a proteine G he:קולטן המצומד לחלבון G nl:G-proteïnegekoppelde receptor
- ↑ Vassilatis DK, Hohmann JG, Zeng H, Li F, Ranchalis JE, Mortrud MT, Brown A, Rodriguez SS, Weller JR, Wright AC, Bergmann JE, Gaitanaris GA (2003). "The G protein-coupled receptor repertoires of human and mouse". Proc Natl Acad Sci U S A. 100 (8): 4903–8. doi:10.1073/pnas.0230374100. PMID 12679517.