Transjugular intrahepatic portosystemic shunt
| Transjugular intrahepatic portosystemic shunt | |
 | |
|---|---|
| TIPS Procedure |
| https://www.youtube.com/watch?v=O2u4_hF3234%7C350}} |
|
Transjugular intrahepatic portosystemic shunt Microchapters |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Umar Ahmad, M.D.[2]
Overview
A transjugular intrahepatic portosystemic shunt, also known as TIPS, is an artificial channel in the liver connecting the portal vein to a hepatic vein (for blood). It is an endovascular channel (created via the blood vessels) through the jugular vein. It is used to treat portal hypertension (which often is due to scarring of the liver- usually due to liver cirrhosis, which frequently leads to intestinal bleeding- from varices, or the buildup of fluid within the abdomen- also known as ascites. A TIPS decreases the effective vascular resistance of the liver. The result is a large pressure drop within the hepatic sinusoids and a decreased portal venous pressure. This, in turn, lessens the pressure on the blood vessels in the intestine so that future bleeding is less likely to occur. The reduced pressure also makes less fluid to develop, although this benefit may take weeks or months to occur.
History
- In 1969, Transjugular intrahepatic portosystemic shunt (TIPS) was first described by Joseph Rösch et al in dogs (patency lasted 2 weeks).[1][2]
- In 1982, Colapinto and Gordon were the first to use this procedure in a human clinical trial but at the time the results were not encouraging and had a high mortality rate in which 9 people died within the month.[3][4]
- In the mid-1980's, J. C. Palmaz achieved long-term patency with the introduction of expandable metallic stents, and was thus approved by the local ethics committee in 1987. The procedure took an average of 8 hours and the long-term patency lasted as long as 9 months.[5]
- In 1990, Jean Marc Perarnau helped improved the puncture techinque which led to a reduction in procedure time.The procedure took an average of 1 to 2 hours.[6]
- In 1995, Nishimine K et al experimented on expanded polytetrafluoroethylene-covered (PTFE) stent grafts that last longer and are currently used for TIPS creation by most interventionalists.[7]
Indications
The following are accepted indications for TIPS:[8]
- Uncontrolled variceal hemorrhage from esophageal, gastric, and intestinal varices that do not respond to endoscopic and medical management[9][10][11]
- Refractory ascites[12]
- Hepatic pleural effusion (hydrothorax)[13]
Controversial indications for TIPS are due to the limited data available in uncontrolled-trials. The following are controversial indications for TIPS:
- Bridge to transplantation and retransplantation[14][15]
- Budd-Chiari syndrome[16]
- Hepatic Hydrothorax[17]
- Hepatorenal syndrome (HRS)[18]
- Veno-occlusive disease (VOD)[19]
Contraindications
Absolute contraindications for TIPS include the following:[20][21][22][23][24][25][26]
- Severe encephalopathy
- Severe and progressive liver failure (on the basis of the Child-Pugh score; scores A and B have a better outcome than score C). Rapid increase in bilirubin concentration requires immediate TIPS occlusion to prevent death.
- Severe right heart failure
- Polycystic liver disease
Relative contraindications for TIPS include the following:[25][20][27][28][29][30][31]
- Pulmonary hypertension
- Portal and hepatic vein thrombosis
- Bilirubin >3 mg/dl
- Hepatopulmonary syndrome
- MELD score above 18
- Active infection
- Tumor within the expected path of the shunt
- Cardiac Failure. Pre-TIPS and post-TIPS E/A ratio measurements.
Preprocedure
Patient prep
Preparing before for the procedure includes the following important steps:[32][33]
- Obtain informed consent
- Review preprocedural vascular ultrasound studies or computed tomography (CT) scans of the abdomen to confirm the patency of the portal vein and assess for anatomic limitations
- Confirm that the patient has no contrast allergy
- Check for a platelet count higher than 50,000/μL[25]
- Relatively normal international normalized ratio (INR)
- Consider broad-spectrum antibiotic prophylaxis, preferably ceftriaxone[34]
- Position the patient supine, with the neck turned away from the side of vein puncture. Avoid pillows unless they are needed.
- Determine the Model for End-stage Liver Disease (MELD) score; this helps predict TIPS mortality, which is higher with a MELD score of 18 or above. Addition of sodium assessment to the MELD score may further enhance prediction of TIPS mortality.
- Anesthesia administration. General anesthesia is usually required for pediatric patients and is preferred in many institutions for adults as well. Procedural sedation may be used, depending on local practices. Sedation may vary depending on continent for example, in Europe, midazolam, piritamide, and propofol are preferentially given while in the U.S general anesthesia with endotracheal intubation is preferred. Midazolam with fentanyl citrate is a reasonable combination for achieving procedural sedation. Local anesthesia is achieved with approximately 5 mL of lidocaine 1% at the jugular puncture site.
- If the patient has ascites with significant volume, perform paracentesis first.
Equipment
Equipment used for transjugular intrahepatic portosystemic shunt (TIPS) creation includes the following:[35][36][37][38]
- Fluoroscopy
- Pressure transducer
- Basic angiography set
- Medical CO2 and its kit
- Sheath, 5 French, and curved catheter
- ultrasound machine with a linear-array probe
- Angioplasty balloons, typically 8 mm × 40 mm
- Chlorhexidine or povidone-iodine solution for skin disinfection
- Bare stents may be used, if needed, to extend to the right atrium
- Heparinized saline (1000-2000 U heparin in 1000 mL of 0.9% NaCl)
- Guide wires - 0.035-inches. Terumo glidewire and exchange-length 0.035-inches
- TIPS kit- Sheath, 10 French, 40 cm; guide catheter, 51 cm, with metal stiffener; portal venous access needle, 60 cm
- Covered stent - Gore Viatorr or another brand, such as Wallgraft (Boston Scientific, Natick, MA; the advantage of the Gore Viatorr is the design, which consists of a distal 2-cm-long unlined segment that is deployed in the portal vein and therefore gives better anchorage without obstructing the flow and allows for nutrient portal perfusion
Procedure
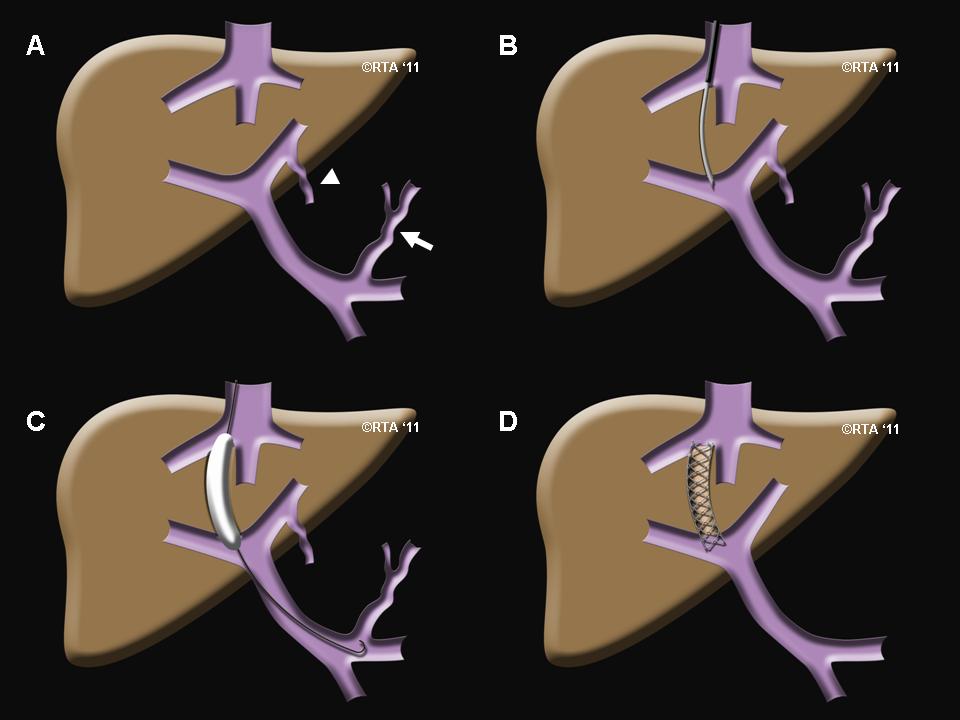
Transjugular intrahepatic portosystemic shunt (TIPS) creation proceeds as follows:[39][40][41][42][43][44][45]
- Positioning- The patient will be positioned on their back.
- Monitoring- The patient will then be connected to monitors that track the heart rate, blood pressure and pulse during the procedure.
- Line placement- A nurse or technologist will insert an intravenous (IV) line into a vein in the patients hand or arm so that sedative medication can be given intravenously. Moderate sedation may be used. As an alternative, the patient may receive general anesthesia.
- Sterilization- The area of the patient's body where the catheter is to be inserted will be sterilized and covered with a surgical drape. Clean the skin on the neck with chlorhexidine or povidone-iodine solution.
- Anesthetic- Numb the area just above the right collarbone with a local anesthetic.
- Incision for catheter- Make a small (≤1 cm) horizontal skin incision.
- Identification of vessel- Using ultrasonographic guidance and a micropuncture or an 18-gauge access needle, puncture the anterior wall of the vein, and enter the vein. Aspirate venous blood to confirm the needle position..
- Determine conditions- Using real time x-ray guidance, advance the 0.035-in. guide wire, and insert the accompanying 5-French sheath over the wire. Use a curved catheter and Terumo hydrophilic wire to access the right hepatic vein. Wedge the catheter in the hepatic vein. Obtain wedged hepatic and inferior vena cava (IVC)/right atrial pressure measurements, and calculate the gradient to confirm the diagnosis of portal hypertension, and also to determine the severity of the condition. To help plan for the placement of the TIPS stent, a contrast material will be injected in the hepatic vein to identify the portal venous system.
- Tract formation - Insert an Amplatz wire, and then exchange the Cobra-2 catheter and 5-French sheath for the 10-French sheath with dilator. Remove the dilator, and introduce the inner sheath, loaded together with the metal stiffener (stent). Insert the system up to 1 cm from the point of intersection of the two veins. Remove the wire, and insert the catheter with the needle. Turn the system, using the metal arrow on the stiffener anteriorly (on the assumption of placement in the right hepatic vein), and advance the needle with the catheter in an anteroinferior direction parallel to the spine, aiming for the portal vein. Start the throw approximately 2 cm from the confluence of the right hepatic vein and the IVC. The right portal vein is typically accessed 0.5-1.5 vertebral body widths lateral to the spine between T10 and T12. Remove the needle, and attach a small syringe with 1 mL of contrast to the catheter. Aspirate while gently withdrawing. A loss of resistance is felt once the catheter enters the portal vein.
- Preparing the stent- Confirm by aspiration and then injection of contrast. Once in the portal vein, insert a Terumo Glidewire. Advance the wire into the superior mesenteric vein or the splenic vein, and advance the catheter. Obtain portal and central pressure measurements to confirm the gradient. Exchange the Terumo wire for an 180-cm Amplatz wire. Perform angioplasty of the tract (with a 6- or 8-mm balloon). Advance a 10-French sheath into the portal vein; replacing the dilator is likely to be helpful. Exchange the catheter for a calibrated pigtail catheter. Perform a double-flush angiogram via both the pigtail and the side arm of the sheath (with the inner metal stiffener removed and sheath pulled back into the hepatic vein) to obtain images of hepatic and portal veins. Use these images to calculate the length of stent required. Measure from the portal vein to the confluence of the hepatic vein and the IVC. To correctly size a Viatorr stent, add 1-2 cm to the measured length. Reinsert the Amplatz wire, and advance the sheath back into the portal vein.
- Placement of stent- Remove the pigtail catheter then insert the stent over the wire into position; be careful to keep the Viatorr stent confined by the packaging sheath until it is fully introduced into the 10-French sheath, and do not advance the stent beyond the sheath tip in the portal vein. Sheath and stent should extend approximately 3 cm into the portal vein. Withdraw the outer TIPS sheath, and then deploy the stent as per its prescribed mechanism. With the Viatorr, an upstream 2-cm-long uncovered segment is deployed by withdrawing the sheath. Positioning may be fine-tuned at this point to achieve the goal of placing the uncovered portion in the portal vein and the covered portion in the tract/hepatic vein. Undersizing the initial tract angioplasty may help give a tactile sense of the junction of the portal vein and the tract. Once the stent is in the correct position, the balloon is inflated, expanding the stent into place.
- After the stent- Perform a portogram to assess flow through stent and any waisting. Measure pressures to ensure a typical goal portohepatic gradient of less than 12 mmHg (for confirmation of a reduction in portal hypertension). If necessary, dilate the stent with an 8-mm balloon. If the stent does not reach the confluence of the hepatic vein and the IVC, it may be extended with an additional uncovered stent. The balloon is then deflated and removed along with the catheter.
- Closing up- Pressure will be applied to prevent any bleeding and the opening in the skin is covered with a bandage. No sutures are needed.
- Observation- The patient will be admitted to the hospital following the procedure, where they will be closely observed.
- Completion- This procedure is usually completed in an hour or two but may take up to several hours depending on the complexity of the condition and vascular anatomy.
Postprocedure
Monitoring and follow-up
The high frequency of shunt stenosis warrants close surveillance with Doppler ultrasonography or portography. To ensure patency, patients undergo a baseline Doppler study within 24 hours of the procedure to document functional parameters, including the direction of portal vein flow and flow velocities throughout the shunt and within the hepatic vein. This is then followed again 3 months, 6 months, 12 months, and annually thereafter.[46] Although TIPS venography with direct portal and right atrial pressure measurements is the criterion standard for stent assessment, high sensitivity and specificity for shunt function has been reported with certain Doppler criteria, such as follows:[47][48][49][50]
| Doppler criteria |
|---|
| Low mean Portal Vein velocity (<30 cm/s) |
| High peak shunt velocity (190 cm/s) |
| Low peak shunt velocity (<50 to 90 cm/s) |
| Return of Antegrade flow in the intrahepatic Portal Veins |
| Significant change in shunt velocity (>50 cm/s) as compared with the immediate postprocedural result |
Doppler ultrasound is the most valuable means to estimate shunt function, such is the following indications:[51][52][53]
| Shunt Patency | |||
|---|---|---|---|
| Doppler Reading | Shunt status | Pre-TIPS | Post-TIPS |
| Flow Velocity | Normal Patent Shunt
Shunt insufficiency Shunt malfunction |
(Vmax: 10–20 cm/sec)
(Vmax: 10–20 cm/sec) (Vmax: 10–20 cm/sec) |
80–160 cm/sec
<60 or >180 cm/sec <40 or >200 cm/sec |
| Portal Flow direction | Normal Patent Shunt
Shunt malfunction |
Hepatopedal
Hepatopedal |
Hepatofungal (90%)
Hepatopedal |
- In general, the pre-procedural velocity increases by TIPS by a factor of 2–4 (40-60 cm/sec).
- It should be emphasized that measurements in the stent-shunt or in the draining hepatic vein are only reliable in cases of simple stenoses. In cases with a complex structure of the intimal proliferation in the stent or in the draining hepatic vein, the measurements are not reliable and normal values cannot exclude stenosis. Therefore, in case of normal values within the stent, the findings in the portal vein define whether shunt function is sufficient or not.
- If simple stenosis is seen, the Bernoulli equation (Δp = 4 v2) can be applied to calculate the pressure gradient Δp (in mmHg) across the stenosis from the flow velocity measured in the stenosis (Vmax in m/sec). Accordingly, a flow velocity (Vmax) of 180 cm/sec (1.8 m/sec) indicates a pressure gradient across the stenosis of 13 mmHg. It could be demonstrated that calculated gradients using the Bernoulli equation closely correlate with gradients determined by catheter measurement (r = 0.84).
- The early post-procedural setting consists of monitoring of the blood pressure, hemoglobin/hematocrit and maybe urine volume during 24 hours. With few exceptions, intensive care is not necessary.
- Generally, in patients with variceal bleeding, ß-blockers are withdrawn and in patients with refractory ascites, diuretic medication is reduced by half.
- Duplex-sonographic examination is performed before patient’s discharge.
Outcome
The following are some of the possible outcomes of the TIPS procedure:[54][55][56][57][58][59][60][61][62][63][64][65][66][67]
- The technical success of TIPS placement is related to the experience and skill of the interventional radiologist. Data from three large centers (University of California; University of Pennsylvania; and the Freiberg group) demonstrated technical success rates of more than 90%.
- Successful TIPS placement results in a portosystemic gradient of less than 12 mm Hg and immediate control of variceal-related bleeding. A target portosystemic gradient of 12 mm Hg is used as varices tend not to bleed when the gradient is less than 12 mm Hg. When technical failure occurs, it is usually due to an anatomic situation that prevents acceptable portal venous puncture.
- Significant reduction in ascites usually occurs within 1 month of the procedure, and this is estimated to occur in 50-90% of cases.
- Late stenosis and occlusion are usually related to pseudointimal hyperplasia within the stent or, more commonly, intimal hyperplasia within the hepatic vein. In most cases, the stenotic stent can be crossed with a guide wire and recanalized with balloon dilation or repeat stent placement to improve long-term patency rates. Primary patency after TIPS placement has been reported to be 66% and 42% after 1 and 2 years, respectively. Primary-assisted patency rates at 1 and 2 years are reported to be 83% and 79%, respectively, and secondary patency rates at 1 and 2 years are reported to be 96% and 90% respectively.
- Patients with severe systemic disease with an APACHE II score higher than 20 have a greater risk for early mortality, compared with others.
- Patients with active bleeding during the procedure also have increased early mortality. The 30-day mortality is iwithn a range of 3-30%; the variation within this range is related to whether the procedure was performed on an emergency basis or an elective basis.
Complications
Possible complication after the TIPS procedure may lead to the following:[68][69][66][70][71][72][73][74][75][76][77][78]
| Major complications | |
|---|---|
| Immediate complications | Delayed complications |
|
|
Encephalopathy (immediate complication) may be amenable to medical therapy ; if it is severe, the shunt may have to be narrowed or embolized[80]. As for In-stent stenosis a greater than 50% stenosis is seen in 25% of TIPS cases; this can be addressed with angioplasty. In the acute phase, the stent may undergo thrombosis. To treat this, the stent can be lysed, or mechanical thrombectomy can be performed. If later in-stent stenosis occurs, perform angioplasty or insert another stent, as required.[81]
| Technical complications |
|---|
|
Centers using sonography during the puncture process have experienced near-complete abolishment of these technical complications. Also, in particular, the Viatorr stent is designed to be placed with great accuracy and misplacement is almost impossible.
Pearls
Interesting facts about the procedure are:[84][85]
- Always perform Doppler ultrasonography to assess the portal vein before starting the TIPS procedure to confirm that the portal vein is not thrombosed.
- If covered shunt patency is difficult to assess on follow-up Doppler ultrasonography, CT may be performed.
- If insertion is unsuccessful, pull the catheter back into the sheath, reinsert the needle, and try again.
- When using Amplatz wires, always keep the tip of the wire under control. If the wire tip is not properly controlled, it can easily perforate the liver or the mesentery.
- To confirm entry into portal vein, inject contrast material. Contrast flowing toward the right atrium indicates hepatic vein location. Static contrast likely indicates biliary system location. Contrast flowing to liver periphery can indicate location in the hepatic artery or portal vein; however, the portal radicals are larger in size.
- If the middle hepatic vein is used, remember to rotate the TIPS sheath and stiffener posteriorly because the middle portal vein lies posterior and inferior to the hepatic vein.
- In contrast to serial paracentesis, TIPS leads to a significant improvement of these parameters including total blood urea nitrogen (BUN) and total body protein, muscle mass, and albumin concentration.
- Technical advancements in skills and stents have reduced complications and improved patency of TIPS. The major obstacle remains hepatic encephalopathy, which requires proper selection of patients and smaller shunts.
- In patients with acute variceal bleeding and high risk of early rebleeding, recent studies showed improved survival recommending early TIPS implantation. With respect to the prevention of rebleeding (secondary prophylaxis), TIPS remains the second-line treatment; unless new studies with covered stents demonstrate its superiority over standard medical therapy.
- In 2 relevant meta-analyses, TIPS improved survival in patients with refractory ascites, justifying its earlier application.
- It clearly reverses the circulatory dysfunction which leads to normalization of the renal function.
- With a 10-year survival rate of 80%, TIPS is the preferred treatment in patients with Budd-Chiari syndrome who do not respond sufficiently to medical treatment. The meta-analysis of individual patient data by Salerno showed that patients who had undergone TIPS lived significantly longer than patients treated with paracentesis.
- TIPS also seems to improve the estimated transplant free survival in patients with MELD scores between 10 and 20, suggesting that even patients with severe disease may benefit from TIPS.
Related Chapters
Reference
- ↑ Rösch J, Hanafee WN, Snow H (1969). "Transjugular portal venography and radiologic portacaval shunt: an experimental study". Radiology. 92 (5): 1112–4. doi:10.1148/92.5.1112. PMID 5771827.
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Colapinto RF, Stronell RD, Birch SJ, Langer B, Blendis LM, Greig PD, Gilas T (1982). "Creation of an intrahepatic portosystemic shunt with a Grüntzig balloon catheter". Can Med Assoc J. 126 (3): 267–8. PMC 1862861. PMID 6977404.
- ↑ Gordon JD, Colapinto RF, Abecassis M, Makowka L, Langer B, Blendis LM, Taylor B, Stronell RD (1987). "Transjugular intrahepatic portosystemic shunt: a nonoperative approach to life-threatening variceal bleeding". Can J Surg. 30 (1): 45–9. PMID 3493058.
- ↑ Palmaz JC, Sibbitt RR, Reuter SR, Garcia F, Tio FO (1985). "Expandable intrahepatic portacaval shunt stents: early experience in the dog". AJR Am J Roentgenol. 145 (4): 821–5. doi:10.2214/ajr.145.4.821. PMID 3876006.
- ↑ Conn HO (1993). "Transjugular intrahepatic portal-systemic shunts: the state of the art". Hepatology. 17 (1): 148–58. PMID 8423036.
- ↑ Nishimine K, Saxon RR, Kichikawa K, Mendel-Hartvig J, Timmermans HA, Shim HJ, Uchida BT, Barton RE, Keller FS, Rösch J (1995). "Improved transjugular intrahepatic portosystemic shunt patency with PTFE-covered stent-grafts: experimental results in swine". Radiology. 196 (2): 341–7. doi:10.1148/radiology.196.2.7617843. PMID 7617843.
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Chau TN, Patch D, Chan YW, Nagral A, Dick R, Burroughs AK (1998). ""Salvage" transjugular intrahepatic portosystemic shunts: gastric fundal compared with esophageal variceal bleeding". Gastroenterology. 114 (5): 981–7. PMID 9558287.
- ↑ Choi YH, Yoon CJ, Park JH, Chung JW, Kwon JW, Choi GM (2003). "Balloon-occluded retrograde transvenous obliteration for gastric variceal bleeding: its feasibility compared with transjugular intrahepatic portosystemic shunt". Korean J Radiol. 4 (2): 109–16. doi:10.3348/kjr.2003.4.2.109. PMC 2698068. PMID 12845306.
- ↑ Gaba RC, Omene BO, Podczerwinski ES, Knuttinen MG, Cotler SJ, Kallwitz ER, Berkes JL, Walzer NM, Bui JT, Owens CA (2012). "TIPS for treatment of variceal hemorrhage: clinical outcomes in 128 patients at a single institution over a 12-year period". J Vasc Interv Radiol. 23 (2): 227–35. doi:10.1016/j.jvir.2011.10.015. PMID 22178037.
- ↑ Garcia-Tsao G (2005). "Transjugular intrahepatic portosystemic shunt in the management of refractory ascites". Semin Intervent Radiol. 22 (4): 278–86. doi:10.1055/s-2005-925554. PMC 3036291. PMID 21326706.
- ↑ Siegerstetter V, Deibert P, Ochs A, Olschewski M, Blum HE, Rössle M (2001). "Treatment of refractory hepatic hydrothorax with transjugular intrahepatic portosystemic shunt: long-term results in 40 patients". Eur J Gastroenterol Hepatol. 13 (5): 529–34. PMID 11396532.
- ↑ Choi DX, Jain AB, Orloff MS (2009). "Utility of transjugular intrahepatic portosystemic shunts in liver-transplant recipients". J. Am. Coll. Surg. 208 (4): 539–46. doi:10.1016/j.jamcollsurg.2009.01.008. PMID 19476787.
- ↑ King A, Masterton G, Gunson B, Olliff S, Redhead D, Mangat K, Oniscu G, Hayes P, Tripathi D (2011). "A case-controlled study of the safety and efficacy of transjugular intrahepatic portosystemic shunts after liver transplantation". Liver Transpl. 17 (7): 771–8. doi:10.1002/lt.22281. PMID 21714062.
- ↑ Molmenti EP, Segev DL, Arepally A, Hong J, Thuluvath PJ, Rai R, Klein AS (2005). "The utility of TIPS in the management of Budd-Chiari syndrome". Ann. Surg. 241 (6): 978–81, discussion 982–3. PMC 1357177. PMID 15912047.
- ↑ Zhang LH, Tong H, Wei B, Wu H, Tang CW (2017). "Transjugular Intrahepatic Portosystemic Shunt as the Treatment for Refractory Hepatic Hydrothorax with Portal Vein Thrombosis". Chin. Med. J. 130 (16): 1999–2000. doi:10.4103/0366-6999.211887. PMC 5555138. PMID 28776556.
- ↑ Guevara M, Rodes J. Hepatorenal syndrome. Int J Biochem Cell Biol. 2005 Jan;37(1):22-6. PMID 15381144.
- ↑ Azoulay D, Castaing D, Lemoine A, Hargreaves GM, Bismuth H (2000). "Transjugular intrahepatic portosystemic shunt (TIPS) for severe veno-occlusive disease of the liver following bone marrow transplantation". Bone Marrow Transplant. 25 (9): 987–92. doi:10.1038/sj.bmt.1702386. PMID 10800068.
- ↑ 20.0 20.1 Rössle M, Ochs A, Gülberg V, Siegerstetter V, Holl J, Deibert P, Olschewski M, Reiser M, Gerbes AL (2000). "A comparison of paracentesis and transjugular intrahepatic portosystemic shunting in patients with ascites". N. Engl. J. Med. 342 (23): 1701–7. doi:10.1056/NEJM200006083422303. PMID 10841872.
- ↑ Pomier-Layrargues G, Bouchard L, Lafortune M, Bissonnette J, Guérette D, Perreault P (2012). "The transjugular intrahepatic portosystemic shunt in the treatment of portal hypertension: current status". Int J Hepatol. 2012: 167868. doi:10.1155/2012/167868. PMC 3408669. PMID 22888442.
- ↑ Baron A, Gulberg V, Sauter G, Waggershauser T, Reiser M, Gerbes AL (1998). "Effects of transjugular intrahepatic portosystemic shunt (TIPS) on quantitative liver function tests". Hepatogastroenterology. 45 (24): 2315–21. PMID 9951915.
- ↑ Gülberg V, Haag K, Rössle M, Gerbes AL (2002). "Hepatic arterial buffer response in patients with advanced cirrhosis". Hepatology. 35 (3): 630–4. doi:10.1053/jhep.2002.31722. PMID 11870377.
- ↑ Radeleff B, Sommer CM, Heye T, Lopez-Benitez R, Sauer P, Schmidt J, Kauczor HU, Richter GM (2009). "Acute increase in hepatic arterial flow during TIPS identified by intravascular flow measurements". Cardiovasc Intervent Radiol. 32 (1): 32–7. doi:10.1007/s00270-008-9435-0. PMID 18830743.
- ↑ 25.0 25.1 25.2 Rössle M, Siegerstetter V, Huber M, Ochs A (1998). "The first decade of the transjugular intrahepatic portosystemic shunt (TIPS): state of the art". Liver. 18 (2): 73–89. PMID 9588766.
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Malinchoc M, Kamath PS, Gordon FD, Peine CJ, Rank J, ter Borg PC (2000). "A model to predict poor survival in patients undergoing transjugular intrahepatic portosystemic shunts". Hepatology. 31 (4): 864–71. doi:10.1053/he.2000.5852. PMID 10733541.
- ↑ Ferral H, Vasan R, Speeg KV, Serna S, Young C, Postoak DW, Wholey MH, McMahan CA (2002). "Evaluation of a model to predict poor survival in patients undergoing elective TIPS procedures". J Vasc Interv Radiol. 13 (11): 1103–8. PMID 12427809.
- ↑ Schepke M, Roth F, Fimmers R, Brensing KA, Sudhop T, Schild HH, Sauerbruch T (2003). "Comparison of MELD, Child-Pugh, and Emory model for the prediction of survival in patients undergoing transjugular intrahepatic portosystemic shunting". Am. J. Gastroenterol. 98 (5): 1167–74. doi:10.1111/j.1572-0241.2003.07515.x. PMID 12809844.
- ↑ Kamath PS, Wiesner RH, Malinchoc M, Kremers W, Therneau TM, Kosberg CL, D'Amico G, Dickson ER, Kim WR (2001). "A model to predict survival in patients with end-stage liver disease". Hepatology. 33 (2): 464–70. doi:10.1053/jhep.2001.22172. PMID 11172350.
- ↑ Salerno F, Merli M, Cazzaniga M, Valeriano V, Rossi P, Lovaria A, Meregaglia D, Nicolini A, Lubatti L, Riggio O (2002). "MELD score is better than Child-Pugh score in predicting 3-month survival of patients undergoing transjugular intrahepatic portosystemic shunt". J. Hepatol. 36 (4): 494–500. PMID 11943420.
- ↑ "Transjugular Intrahepatic Portosystemic Shunt Technique: Creation of Transjugular Intrahepatic Portosystemic Shunt, Complications".
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Rössle, Martin (2013). "TIPS: 25years later". Journal of Hepatology. 59 (5): 1081–1093. doi:10.1016/j.jhep.2013.06.014. ISSN 0168-8278.
- ↑ Maleux G, Nevens F, Heye S, Verslype C, Marchal G (2006). "The use of carbon dioxide wedged hepatic venography to identify the portal vein: comparison with direct catheter portography with iodinated contrast medium and analysis of predictive factors influencing level of opacification". J Vasc Interv Radiol. 17 (11 Pt 1): 1771–9. doi:10.1097/01.RVI.0000242185.26944.60. PMID 17142707.
- ↑ Clark W, Golkar F, Luberice K, Toomey P, Paul H, Marcadis A, Okpaleke C, Vice M, Hernandez J, Alsina A, Rosemurgy AS (2011). "Uncovering the truth about covered stents: is there a difference between covered versus uncovered stents with transjugular intrahepatic portosystemic shunts?". Am. J. Surg. 202 (5): 561–4. doi:10.1016/j.amjsurg.2011.06.021. PMID 21944293.
- ↑ "Transjugular Intrahepatic Portosystemic Shunt Technique: Creation of Transjugular Intrahepatic Portosystemic Shunt, Complications".
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Copelan A, Kapoor B, Sands M (2014). "Transjugular intrahepatic portosystemic shunt: indications, contraindications, and patient work-up". Semin Intervent Radiol. 31 (3): 235–42. doi:10.1055/s-0034-1382790. PMC 4139433. PMID 25177083.
- ↑ Smail A, Azoulay D, Castaing D (2004). "[The technique of Trans-Jugular Porto-systemic Shunt (TIPS)]". J Chir (Paris) (in French). 141 (1): 21–6. PMID 15029059.
- ↑ Martínez-Cuesta A, Elduayen B, Vivas I, Delgado C, González-Crespo I, Bilbao JI (2000). "CO(2) wedged hepatic venography: technical considerations and comparison with direct and indirect portography with iodinated contrast". Abdom Imaging. 25 (6): 576–82. PMID 11029087.
- ↑ Feyssa E, Ortiz J, Grewal K, Azhar A, Parsikia A, Tufail K, Hashemi N, Brady P, Araya V (2011). "MELD score less than 15 predicts prolonged survival after transjugular intrahepatic portosystemic shunt for refractory ascites after liver transplantation". Transplantation. 91 (7): 786–92. doi:10.1097/TP.0b013e31820e014e. PMID 21304440.
- ↑ Ahmed R, Santhanam P, Rayyan Y (2015). "MELD-Na as a prognostic indicator of 30- and 90-day mortality in patients with end-stage liver disease after creation of transjugular intrahepatic portosystemic shunt". Eur J Gastroenterol Hepatol. 27 (10): 1226–7. doi:10.1097/MEG.0000000000000412. PMID 26111072.
- ↑ "Transjugular Intrahepatic Portosystemic Shunt Technique: Creation of Transjugular Intrahepatic Portosystemic Shunt, Complications".
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Ferral H, Gomez-Reyes E, Fimmel CJ (2016). "Post-Transjugular Intrahepatic Portosystemic Shunt Follow-Up and Management in the VIATORR Era". Tech Vasc Interv Radiol. 19 (1): 82–8. doi:10.1053/j.tvir.2016.01.009. PMID 26997092.
- ↑ Zizka J, Eliás P, Krajina A, Michl A, Lojík M, Ryska P, Masková J, Hůlek P, Safka V, Vanásek T, Bukac J (2000). "Value of Doppler sonography in revealing transjugular intrahepatic portosystemic shunt malfunction: a 5-year experience in 216 patients". AJR Am J Roentgenol. 175 (1): 141–8. doi:10.2214/ajr.175.1.1750141. PMID 10882264.
- ↑ Nicolas C, Le Gouge A, d'Alteroche L, Ayoub J, Georgescu M, Vidal V, Castaing D, Cercueil JP, Chevallier P, Roumy J, Trillaud H, Boyer L, Le Pennec V, Perret C, Giraudeau B, Perarnau JM, Stic-Tips G (2017). "Evaluation of Doppler-ultrasonography in the diagnosis of transjugular intrahepatic portosystemic shunt dysfunction: A prospective study". World J Hepatol. 9 (27): 1125–1132. doi:10.4254/wjh.v9.i27.1125. PMC 5620422. PMID 29026464. Vancouver style error: initials (help)
- ↑ "Transjugular Intrahepatic Portosystemic Shunt Technique: Creation of Transjugular Intrahepatic Portosystemic Shunt, Complications".
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Rössle M, Haag K, Ochs A, Sellinger M, Nöldge G, Perarnau JM, Berger E, Blum U, Gabelmann A, Hauenstein K (1994). "The transjugular intrahepatic portosystemic stent-shunt procedure for variceal bleeding". N. Engl. J. Med. 330 (3): 165–71. doi:10.1056/NEJM199401203300303. PMID 8264738.
- ↑ Nolte, W; Hartmann, H; Ramadori, G (1998). "Duplex ultrasonography in predicting patency of TIPS". Gastroenterology. 115 (5): 1305–1305. doi:10.1016/S0016-5085(98)70117-9. ISSN 0016-5085.
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Guevara M, Ginès P, Bandi JC, Gilabert R, Sort P, Jiménez W, Garcia-Pagan JC, Bosch J, Arroyo V, Rodés J (1998). "Transjugular intrahepatic portosystemic shunt in hepatorenal syndrome: effects on renal function and vasoactive systems". Hepatology. 28 (2): 416–22. doi:10.1002/hep.510280219. PMID 9696006.
- ↑ Brensing KA, Textor J, Perz J, Schiedermaier P, Raab P, Strunk H, Klehr HU, Kramer HJ, Spengler U, Schild H, Sauerbruch T (2000). "Long term outcome after transjugular intrahepatic portosystemic stent-shunt in non-transplant cirrhotics with hepatorenal syndrome: a phase II study". Gut. 47 (2): 288–95. PMC 1727992. PMID 10896924.
- ↑ Wong F, Pantea L, Sniderman K (2004). "Midodrine, octreotide, albumin, and TIPS in selected patients with cirrhosis and type 1 hepatorenal syndrome". Hepatology. 40 (1): 55–64. doi:10.1002/hep.20262. PMID 15239086.
- ↑ Testino G, Ferro C, Sumberaz A, Messa P, Morelli N, Guadagni B, Ardizzone G, Valente U (2003). "Type-2 hepatorenal syndrome and refractory ascites: role of transjugular intrahepatic portosystemic stent-shunt in eighteen patients with advanced cirrhosis awaiting orthotopic liver transplantation". Hepatogastroenterology. 50 (54): 1753–5. PMID 14696397.
- ↑ Kiafar C, Gilani N (2008). "Hepatic hydrothorax: current concepts of pathophysiology and treatment options". Ann Hepatol. 7 (4): 313–20. PMID 19034230.
- ↑ Gordon FD, Anastopoulos HT, Crenshaw W, Gilchrist B, McEniff N, Falchuk KR, LoCicero J, Lewis WD, Jenkins RL, Trey C (1997). "The successful treatment of symptomatic, refractory hepatic hydrothorax with transjugular intrahepatic portosystemic shunt". Hepatology. 25 (6): 1366–9. doi:10.1002/hep.510250611. PMID 9185754.
- ↑ Wilputte JY, Goffette P, Zech F, Godoy-Gepert A, Geubel A (2007). "The outcome after transjugular intrahepatic portosystemic shunt (TIPS) for hepatic hydrothorax is closely related to liver dysfunction: a long-term study in 28 patients". Acta Gastroenterol. Belg. 70 (1): 6–10. PMID 17619531.
- ↑ Garcia-Pagán JC, Heydtmann M, Raffa S, Plessier A, Murad S, Fabris F, Vizzini G, Gonzales Abraldes J, Olliff S, Nicolini A, Luca A, Primignani M, Janssen HL, Valla D, Elias E, Bosch J (2008). "TIPS for Budd-Chiari syndrome: long-term results and prognostics factors in 124 patients". Gastroenterology. 135 (3): 808–15. doi:10.1053/j.gastro.2008.05.051. PMID 18621047.
- ↑ Kauffmann, G. W.; Richter, G. M. (1999). "Transjugular intrahepatic portosystemic stent-shunt (TIPSS): technique and indications". European Radiology. 9 (4): 685–692. doi:10.1007/s003300050733. ISSN 0938-7994.
- ↑ Tripathi D, Helmy A, Macbeth K, Balata S, Lui HF, Stanley AJ, Redhead DN, Hayes PC (2004). "Ten years' follow-up of 472 patients following transjugular intrahepatic portosystemic stent-shunt insertion at a single centre". Eur J Gastroenterol Hepatol. 16 (1): 9–18. PMID 15095847.
- ↑ Gaba RC, Khiatani VL, Knuttinen MG, Omene BO, Carrillo TC, Bui JT, Owens CA (2011). "Comprehensive review of TIPS technical complications and how to avoid them". AJR Am J Roentgenol. 196 (3): 675–85. doi:10.2214/AJR.10.4819. PMID 21343513.
- ↑ Saugel B, Phillip V, Gaa J, Berger H, Lersch C, Schultheiss C, Thies P, Schneider H, Höllthaler J, Herrmann A, Schmid RM, Huber W (2012). "Advanced hemodynamic monitoring before and after transjugular intrahepatic portosystemic shunt: implications for selection of patients--a prospective study". Radiology. 262 (1): 343–52. doi:10.1148/radiol.11110043. PMID 22025732.
- ↑ 66.0 66.1 Armstrong PK, MacLeod C (2003). "Infection of transjugular intrahepatic portosystemic shunt devices: three cases and a review of the literature". Clin. Infect. Dis. 36 (4): 407–12. doi:10.1086/346156. PMID 12567297.
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Ahmed R, Santhanam P, Rayyan Y (2015). "MELD-Na as a prognostic indicator of 30- and 90-day mortality in patients with end-stage liver disease after creation of transjugular intrahepatic portosystemic shunt". Eur J Gastroenterol Hepatol. 27 (10): 1226–7. doi:10.1097/MEG.0000000000000412. PMID 26111072.
- ↑ Tripathi D, Helmy A, Macbeth K, Balata S, Lui HF, Stanley AJ, Redhead DN, Hayes PC (2004). "Ten years' follow-up of 472 patients following transjugular intrahepatic portosystemic stent-shunt insertion at a single centre". Eur J Gastroenterol Hepatol. 16 (1): 9–18. PMID 15095847.
- ↑ Bala TM, Panda M (2006). "Cardiac perforation and tamponade: a potentially fatal complication during transjugular intrahepatic portosystemic shunt placement". South. Med. J. 99 (9): 1000–2. doi:10.1097/01.smj.0000233256.23569.b8. PMID 17004537.
- ↑ Jalan R, Elton RA, Redhead DN, Finlayson ND, Hayes PC (1995). "Analysis of prognostic variables in the prediction of mortality, shunt failure, variceal rebleeding and encephalopathy following the transjugular intrahepatic portosystemic stent-shunt for variceal haemorrhage". J. Hepatol. 23 (2): 123–8. PMID 7499782.
- ↑ Clavien PA, Selzner M, Tuttle-Newhall JE, Harland RC, Suhocki P (1998). "Liver transplantation complicated by misplaced TIPS in the portal vein". Ann. Surg. 227 (3): 440–5. PMC 1191283. PMID 9527068.
- ↑ Gschwantler M, Gebauer A, Vavrik J, Madani B, Rohrmoser M, Schrutka-Kölbl C, Brownstone E, Fleischer J, Tscholakoff D, Weiss W (1997). "Acute and chronic complications after implantation of a transjugular intrahepatic portosystemic shunt--a prospective study in 53 patients". Z Gastroenterol. 35 (11): 999–1005. PMID 9429285.
- ↑ Masson S, Mardini HA, Rose JD, Record CO (2008). "Hepatic encephalopathy after transjugular intrahepatic portosystemic shunt insertion: a decade of experience". QJM. 101 (6): 493–501. doi:10.1093/qjmed/hcn037. PMID 18440957.
- ↑ Freedman AM, Sanyal AJ, Tisnado J, Cole PE, Shiffman ML, Luketic VA, Purdum PP, Darcy MD, Posner MP (1993). "Complications of transjugular intrahepatic portosystemic shunt: a comprehensive review". Radiographics. 13 (6): 1185–210. doi:10.1148/radiographics.13.6.8290720. PMID 8290720.
- ↑ Pereira K, Baker R, Salsamendi J, Doshi M, Kably I, Bhatia S (2016). "An Approach to Endovascular and Percutaneous Management of Transjugular Intrahepatic Portosystemic Shunt (TIPS) Dysfunction: A Pictorial Essay and Clinical Practice Algorithm". Cardiovasc Intervent Radiol. 39 (5): 639–651. doi:10.1007/s00270-015-1247-4. PMID 26604117.
- ↑ Colombato L (2007). "The role of transjugular intrahepatic portosystemic shunt (TIPS) in the management of portal hypertension". J. Clin. Gastroenterol. 41 Suppl 3: S344–51. doi:10.1097/MCG.0b013e318157e500. PMID 17975487.
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.
- ↑ Mallavarapu RK, Grimsley EW (2007). "Incarcerated umbilical hernia after transjugular intrahepatic portosystemic shunt procedure for refractory ascites". Clin. Gastroenterol. Hepatol. 5 (9): A26. doi:10.1016/j.cgh.2007.07.018. PMID 17825762.
- ↑ Casado M, Bosch J, García-Pagán JC, Bru C, Bañares R, Bandi JC, Escorsell A, Rodríguez-Láiz JM, Gilabert R, Feu F, Schorlemer C, Echenagusia A, Rodés J (1998). "Clinical events after transjugular intrahepatic portosystemic shunt: correlation with hemodynamic findings". Gastroenterology. 114 (6): 1296–303. PMID 9609767.
- ↑ Shah RP, Sze DY (2016). "Complications During Transjugular Intrahepatic Portosystemic Shunt Creation". Tech Vasc Interv Radiol. 19 (1): 61–73. doi:10.1053/j.tvir.2016.01.007. PMID 26997090.
- ↑ Fidelman N, Kwan SW, LaBerge JM, Gordon RL, Ring EJ, Kerlan RK (2012). "The transjugular intrahepatic portosystemic shunt: an update". AJR Am J Roentgenol. 199 (4): 746–55. doi:10.2214/AJR.12.9101. PMID 22997364.
- ↑ Boyer TD, Haskal ZJ (2005). "American Association for the Study of Liver Diseases Practice Guidelines: the role of transjugular intrahepatic portosystemic shunt creation in the management of portal hypertension". J Vasc Interv Radiol. 16 (5): 615–29. doi:10.1097/01.RVI.0000157297.91510.21. PMID 15872315.
- ↑ "Transjugular Intrahepatic Portosystemic Shunt Technique: Creation of Transjugular Intrahepatic Portosystemic Shunt, Complications".
- ↑ Rössle M (2013). "TIPS: 25 years later". J. Hepatol. 59 (5): 1081–93. doi:10.1016/j.jhep.2013.06.014. PMID 23811307.