Congestive heart failure treatment of patients with current or prior symptoms of heart failure (Stage C): Difference between revisions
(/* Patients with Reduced LVEF(HFrEF) (DO NOT EDIT){{cite journal| author=Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM | display-authors=etal| title=2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. | journal=Circulation | year= 2022 | volume= 145 | issue= 18 | pages= e876-e894 | pmid=35363500 | doi=10.1161/CIR.000000...) |
(/* Pharmacological Treatmetn for HFrEF (DO NOT EDIT){{cite journal| author=Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM | display-authors=etal| title=2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. | journal=Circulation | year= 2022 | volume= 145 | issue= 18 | pages= e876-e894 | pmid=35363500 | doi=10.1161/CIR.0000...) |
||
| Line 94: | Line 94: | ||
== 2022 AHA/ACC/HFSA Heart Failure Guideline/ 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure/2016 ACC/AHA/HFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline,2013 ACC/AHA Guideline, 2009 ACC/AHA Focused Update and 2005 Guidelines for the Diagnosis and Management of Heart Failure in the Adult (DO NOT EDIT) <ref name="pmid35363500">{{cite journal| author=Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM | display-authors=etal| title=2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. | journal=Circulation | year= 2022 | volume= 145 | issue= 18 | pages= e876-e894 | pmid=35363500 | doi=10.1161/CIR.0000000000001062 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=35363500 }} </ref><ref name="pmid23747642">{{cite journal| author=Yancy CW, Jessup M, Bozkurt B, Masoudi FA, Butler J, McBride PE et al.| title=2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. | journal=J Am Coll Cardiol | year= 2013 | volume= | issue= | pages= | pmid=23747642 | doi=10.1016/j.jacc.2013.05.019 | pmc= | url=http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=23747642 }} </ref><ref name="pmid19324967">Jessup M, Abraham WT, Casey DE, Feldman AM, Francis GS, Ganiats TG et al. (2009) [http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&retmode=ref&cmd=prlinks&id=19324967 2009 focused update: ACCF/AHA Guidelines for the Diagnosis and Management of Heart Failure in Adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the International Society for Heart and Lung Transplantation.] ''Circulation'' 119 (14):1977-2016.[http://dx.doi.org/10.1161/CIRCULATIONAHA.109.192064 DOI:10.1161/CIRCULATIONAHA.109.192064] PMID: [http://pubmed.gov/19324967 19324967]</ref>== | == 2022 AHA/ACC/HFSA Heart Failure Guideline/ 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure/2016 ACC/AHA/HFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline,2013 ACC/AHA Guideline, 2009 ACC/AHA Focused Update and 2005 Guidelines for the Diagnosis and Management of Heart Failure in the Adult (DO NOT EDIT) <ref name="pmid35363500">{{cite journal| author=Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM | display-authors=etal| title=2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. | journal=Circulation | year= 2022 | volume= 145 | issue= 18 | pages= e876-e894 | pmid=35363500 | doi=10.1161/CIR.0000000000001062 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=35363500 }} </ref><ref name="pmid23747642">{{cite journal| author=Yancy CW, Jessup M, Bozkurt B, Masoudi FA, Butler J, McBride PE et al.| title=2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. | journal=J Am Coll Cardiol | year= 2013 | volume= | issue= | pages= | pmid=23747642 | doi=10.1016/j.jacc.2013.05.019 | pmc= | url=http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=23747642 }} </ref><ref name="pmid19324967">Jessup M, Abraham WT, Casey DE, Feldman AM, Francis GS, Ganiats TG et al. (2009) [http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&retmode=ref&cmd=prlinks&id=19324967 2009 focused update: ACCF/AHA Guidelines for the Diagnosis and Management of Heart Failure in Adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the International Society for Heart and Lung Transplantation.] ''Circulation'' 119 (14):1977-2016.[http://dx.doi.org/10.1161/CIRCULATIONAHA.109.192064 DOI:10.1161/CIRCULATIONAHA.109.192064] PMID: [http://pubmed.gov/19324967 19324967]</ref>== | ||
===Pharmacological | ===Pharmacological Treatment for [[HFrEF]] (DO NOT EDIT)<ref name="pmid35363500">{{cite journal| author=Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM | display-authors=etal| title=2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. | journal=Circulation | year= 2022 | volume= 145 | issue= 18 | pages= e876-e894 | pmid=35363500 | doi=10.1161/CIR.0000000000001062 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=35363500 }} </ref><ref name="pmid23747642">{{cite journal| author=Yancy CW, Jessup M, Bozkurt B, Masoudi FA, Butler J, McBride PE et al.| title=2013 ACCF/AHA Guideline for the Management of Heart Failure: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. | journal=J Am Coll Cardiol | year= 2013 | volume= | issue= | pages= | pmid=23747642 | doi=10.1016/j.jacc.2013.05.019 | pmc= |url=http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=23747642 }} </ref><ref name="pmid19324967">Jessup M, Abraham WT, Casey DE, Feldman AM, Francis GS, Ganiats TG et al. (2009) [http://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&retmode=ref&cmd=prlinks&id=19324967 2009 focused update: ACCF/AHA Guidelines for the Diagnosis and Management of Heart Failure in Adults: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines: developed in collaboration with the International Society for Heart and Lung Transplantation.] ''Circulation'' 119 (14):1977-2016. [http://dx.doi.org/10.1161/CIRCULATIONAHA.109.192064 DOI:10.1161/CIRCULATIONAHA.109.192064] PMID:[http://pubmed.gov/19324967 19324967]</ref>=== | ||
====Renin-Angiotensin System Inhibition With ACEi or ARB or ARNi==== | ====Renin-Angiotensin System Inhibition With ACEi or ARB or ARNi==== | ||
| Line 106: | Line 106: | ||
|- | |- | ||
|bgcolor="LightGreen"|<nowiki>"</nowiki>'''2.''' In patients with previous or current symptoms of chronic HFrEF, the use of ACEi is beneficial to reduce morbidity and mortality when the use of ARNi is not feasible.<ref name="pmid2883575">{{cite journal| author=CONSENSUS Trial Study Group| title=Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). | journal=N Engl J Med | year= 1987 | volume= 316 | issue= 23 | pages= 1429-35 | pmid=2883575 | doi=10.1056/NEJM198706043162301 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=2883575 }} </ref><ref name="pmid2057034">{{cite journal| author=SOLVD Investigators. Yusuf S, Pitt B, Davis CE, Hood WB, Cohn JN| title=Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. | journal=N Engl J Med | year= 1991 | volume= 325 | issue= 5 | pages= 293-302 | pmid=2057034 | doi=10.1056/NEJM199108013250501 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=2057034 }} </ref><ref name="pmid10587334">{{cite journal| author=Packer M, Poole-Wilson PA, Armstrong PW, Cleland JG, Horowitz JD, Massie BM | display-authors=etal| title=Comparative effects of low and high doses of the angiotensin-converting enzyme inhibitor, lisinopril, on morbidity and mortality in chronic heart failure. ATLAS Study Group. | journal=Circulation | year= 1999 | volume= 100 | issue= 23 | pages= 2312-8 | pmid=10587334 | doi=10.1161/01.cir.100.23.2312 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=10587334 }} </ref><ref name="pmid1386652">{{cite journal| author=Pfeffer MA, Braunwald E, Moyé LA, Basta L, Brown EJ, Cuddy TE | display-authors=etal| title=Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. | journal=N Engl J Med | year= 1992 | volume= 327 | issue= 10 | pages= 669-77 | pmid=1386652 | doi=10.1056/NEJM199209033271001 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=1386652 }} </ref><ref name="pmid8104270">{{cite journal| author=| title=Effect of ramipril on mortality and morbidity of survivors of acute myocardial infarction with clinical evidence of heart failure. The Acute Infarction Ramipril Efficacy (AIRE) Study Investigators. | journal=Lancet | year= 1993 | volume= 342 | issue= 8875 | pages= 821-8 | pmid=8104270 | doi= | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=8104270 }} </ref><ref name="pmid7477219">{{cite journal| author=Køber L, Torp-Pedersen C, Carlsen JE, Bagger H, Eliasen P, Lyngborg K | display-authors=etal| title=A clinical trial of the angiotensin-converting-enzyme inhibitor trandolapril in patients with left ventricular dysfunction after myocardial infarction. Trandolapril Cardiac Evaluation (TRACE) Study Group. | journal=N Engl J Med | year= 1995 | volume= 333 | issue= 25 | pages= 1670-6 | pmid=7477219 | doi=10.1056/NEJM199512213332503 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=7477219 }} </ref><ref name="pmid7654275">{{cite journal| author=Garg R, Yusuf S| title=Overview of randomized trials of angiotensin-converting enzyme inhibitors on mortality and morbidity in patients with heart failure. Collaborative Group on ACE Inhibitor Trials. | journal=JAMA | year= 1995 | volume= 273 | issue= 18 | pages= 1450-6 | pmid=7654275 | doi= | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=7654275 }} </ref><ref name="pmid20625347">{{cite journal| author=Woodard-Grice AV, Lucisano AC, Byrd JB, Stone ER, Simmons WH, Brown NJ| title=Sex-dependent and race-dependent association of XPNPEP2 C-2399A polymorphism with angiotensin-converting enzyme inhibitor-associated angioedema. | journal=Pharmacogenet Genomics | year= 2010 | volume= 20 | issue= 9 | pages= 532-6 | pmid=20625347 | doi=10.1097/FPC.0b013e32833d3acb | pmc=2945219 | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=20625347 }} </ref> ''([[ACC AHA guidelines classification scheme#Level of Evidence|Level of Evidence: | |bgcolor="LightGreen"|<nowiki>"</nowiki>'''2.''' In patients with previous or current symptoms of chronic HFrEF, the use of ACEi is beneficial to reduce morbidity and mortality when the use of ARNi is not feasible.<ref name="pmid2883575">{{cite journal| author=CONSENSUS Trial Study Group| title=Effects of enalapril on mortality in severe congestive heart failure. Results of the Cooperative North Scandinavian Enalapril Survival Study (CONSENSUS). | journal=N Engl J Med | year= 1987 | volume= 316 | issue= 23 | pages= 1429-35 | pmid=2883575 | doi=10.1056/NEJM198706043162301 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=2883575 }} </ref><ref name="pmid2057034">{{cite journal| author=SOLVD Investigators. Yusuf S, Pitt B, Davis CE, Hood WB, Cohn JN| title=Effect of enalapril on survival in patients with reduced left ventricular ejection fractions and congestive heart failure. | journal=N Engl J Med | year= 1991 | volume= 325 | issue= 5 | pages= 293-302 | pmid=2057034 | doi=10.1056/NEJM199108013250501 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=2057034 }} </ref><ref name="pmid10587334">{{cite journal| author=Packer M, Poole-Wilson PA, Armstrong PW, Cleland JG, Horowitz JD, Massie BM | display-authors=etal| title=Comparative effects of low and high doses of the angiotensin-converting enzyme inhibitor, lisinopril, on morbidity and mortality in chronic heart failure. ATLAS Study Group. | journal=Circulation | year= 1999 | volume= 100 | issue= 23 | pages= 2312-8 | pmid=10587334 | doi=10.1161/01.cir.100.23.2312 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=10587334 }} </ref><ref name="pmid1386652">{{cite journal| author=Pfeffer MA, Braunwald E, Moyé LA, Basta L, Brown EJ, Cuddy TE | display-authors=etal| title=Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. | journal=N Engl J Med | year= 1992 | volume= 327 | issue= 10 | pages= 669-77 | pmid=1386652 | doi=10.1056/NEJM199209033271001 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=1386652 }} </ref><ref name="pmid8104270">{{cite journal| author=| title=Effect of ramipril on mortality and morbidity of survivors of acute myocardial infarction with clinical evidence of heart failure. The Acute Infarction Ramipril Efficacy (AIRE) Study Investigators. | journal=Lancet | year= 1993 | volume= 342 | issue= 8875 | pages= 821-8 | pmid=8104270 | doi= | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=8104270 }} </ref><ref name="pmid7477219">{{cite journal| author=Køber L, Torp-Pedersen C, Carlsen JE, Bagger H, Eliasen P, Lyngborg K | display-authors=etal| title=A clinical trial of the angiotensin-converting-enzyme inhibitor trandolapril in patients with left ventricular dysfunction after myocardial infarction. Trandolapril Cardiac Evaluation (TRACE) Study Group. | journal=N Engl J Med | year= 1995 | volume= 333 | issue= 25 | pages= 1670-6 | pmid=7477219 | doi=10.1056/NEJM199512213332503 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=7477219 }} </ref><ref name="pmid7654275">{{cite journal| author=Garg R, Yusuf S| title=Overview of randomized trials of angiotensin-converting enzyme inhibitors on mortality and morbidity in patients with heart failure. Collaborative Group on ACE Inhibitor Trials. | journal=JAMA | year= 1995 | volume= 273 | issue= 18 | pages= 1450-6 | pmid=7654275 | doi= | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=7654275 }} </ref><ref name="pmid20625347">{{cite journal| author=Woodard-Grice AV, Lucisano AC, Byrd JB, Stone ER, Simmons WH, Brown NJ| title=Sex-dependent and race-dependent association of XPNPEP2 C-2399A polymorphism with angiotensin-converting enzyme inhibitor-associated angioedema. | journal=Pharmacogenet Genomics | year= 2010 | volume= 20 | issue= 9 | pages= 532-6 | pmid=20625347 | doi=10.1097/FPC.0b013e32833d3acb | pmc=2945219 | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=20625347 }} </ref> ''([[ACC AHA guidelines classification scheme#Level of Evidence|Level of Evidence: A]])'' <nowiki>"</nowiki> | ||
|- | |- | ||
|bgcolor=" | |bgcolor="LightGreen"|<nowiki>"</nowiki>'''3.''' In patients with previous or current symptoms of chronic HFrEF who are intolerant to ACEi because of cough or angioedema and when the use of ARNi is not feasible, the use of ARB is recommended to reduce morbidity and mortality. <ref name="pmid11759645">{{cite journal| author=Cohn JN, Tognoni G, Valsartan Heart Failure Trial Investigators| title=A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. | journal=N Engl J Med | year= 2001 | volume= 345 | issue= 23 | pages= 1667-75 | pmid=11759645 | doi=10.1056/NEJMoa010713 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=11759645 }} </ref><ref name="pmid14610160">{{cite journal| author=Pfeffer MA, McMurray JJ, Velazquez EJ, Rouleau JL, Køber L, Maggioni AP | display-authors=etal| title=Valsartan, captopril, or both in myocardial infarction complicated by heart failure, left ventricular dysfunction, or both. | journal=N Engl J Med | year= 2003 | volume= 349 | issue= 20 | pages= 1893-906 | pmid=14610160 | doi=10.1056/NEJMoa032292 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=14610160 }} [https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=&cmd=prlinks&id=15230551 Review in: ACP J Club. 2004 Jul-Aug;141(1):3] </ref><ref name="pmid19922995">{{cite journal| author=Konstam MA, Neaton JD, Dickstein K, Drexler H, Komajda M, Martinez FA | display-authors=etal| title=Effects of high-dose versus low-dose losartan on clinical outcomes in patients with heart failure (HEAAL study): a randomised, double-blind trial. | journal=Lancet | year= 2009 | volume= 374 | issue= 9704 | pages= 1840-8 | pmid=19922995 | doi=10.1016/S0140-6736(09)61913-9 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=19922995 }} [https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=&cmd=prlinks&id=20436124 Review in: Evid Based Med. 2010 Apr;15(2):51-2] </ref><ref name="pmid18700309">{{cite journal| author=Dominiak M| title=[Commentary to the article: ONTARGET Investigators, Yusuf S, Teo KK, Pogue J et al. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med 2008; 358: 1547-59]. | journal=Kardiol Pol | year= 2008 | volume= 66 | issue= 6 | pages= 705-6; discussion 707 | pmid=18700309 | doi= | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=18700309 }} </ref><ref name="pmid18757085">{{cite journal| author=Telmisartan Randomised AssessmeNt Study in ACE iNtolerant subjects with cardiovascular Disease (TRANSCEND) Investigators. Yusuf S, Teo K, Anderson C, Pogue J, Dyal L | display-authors=etal| title=Effects of the angiotensin-receptor blocker telmisartan on cardiovascular events in high-risk patients intolerant to angiotensin-converting enzyme inhibitors: a randomised controlled trial. | journal=Lancet | year= 2008 | volume= 372 | issue= 9644 | pages= 1174-83 | pmid=18757085 | doi=10.1016/S0140-6736(08)61242-8 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=18757085 }} [https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=&cmd=prlinks&id=19238604 Review in: Ann Intern Med. 2009 Feb 17;150(4):JC2-6] </ref> ''([[ACC AHA guidelines classification scheme#Level of Evidence|Level of Evidence: A]])'' <nowiki>"</nowiki> | ||
|- | |- | ||
|bgcolor=" | |bgcolor="LightGreen"|<nowiki>"</nowiki>'''5.''' In patients with chronic symptomatic HFrEF NYHA class II or III who tolerate an ACEi or ARB, replacement by an ARNi is recommended to further reduce morbidity and mortality. <ref name="pmid25176015">{{cite journal| author=McMurray JJ, Packer M, Desai AS, Gong J, Lefkowitz MP, Rizkala AR | display-authors=etal| title=Angiotensin-neprilysin inhibition versus enalapril in heart failure. | journal=N Engl J Med | year= 2014 | volume= 371 | issue= 11 | pages= 993-1004 | pmid=25176015 | doi=10.1056/NEJMoa1409077 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=25176015 }} [https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=&cmd=prlinks&id=25659916 Review in: Evid Based Med. 2015 Apr;20(2):61] [https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=&cmd=prlinks&id=25686189 Review in: Ann Intern Med. 2015 Feb 17;162(4):JC2] </ref><ref name="pmid31134724">{{cite journal| author=Wachter R, Senni M, Belohlavek J, Straburzynska-Migaj E, Witte KK, Kobalava Z | display-authors=etal| title=Initiation of sacubitril/valsartan in haemodynamically stabilised heart failure patients in hospital or early after discharge: primary results of the randomised TRANSITION study. | journal=Eur J Heart Fail | year= 2019 | volume= 21 | issue= 8 | pages= 998-1007 | pmid=31134724 | doi=10.1002/ejhf.1498 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=31134724 }} </ref><ref name="pmid30415601">{{cite journal| author=Velazquez EJ, Morrow DA, DeVore AD, Duffy CI, Ambrosy AP, McCague K | display-authors=etal| title=Angiotensin-Neprilysin Inhibition in Acute Decompensated Heart Failure. | journal=N Engl J Med | year= 2019 | volume= 380 | issue= 6 | pages= 539-548 | pmid=30415601 | doi=10.1056/NEJMoa1812851 | pmc= | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=30415601 }} </ref><ref name="pmid31475296">{{cite journal| author=Desai AS, Solomon SD, Shah AM, Claggett BL, Fang JC, Izzo J | display-authors=etal| title=Effect of Sacubitril-Valsartan vs Enalapril on Aortic Stiffness in Patients With Heart Failure and Reduced Ejection Fraction: A Randomized Clinical Trial. | journal=JAMA | year= 2019 | volume= 322 | issue= 11 | pages= 1077-1084 | pmid=31475296 | doi=10.1001/jama.2019.12843 | pmc=6749534 | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=31475296 }} </ref><ref name="pmid31240976">{{cite journal| author=Wang Y, Zhou R, Lu C, Chen Q, Xu T, Li D| title=Effects of the Angiotensin-Receptor Neprilysin Inhibitor on Cardiac Reverse Remodeling: Meta-Analysis. | journal=J Am Heart Assoc | year= 2019 | volume= 8 | issue= 13 | pages= e012272 | pmid=31240976 | doi=10.1161/JAHA.119.012272 | pmc=6662364 | url=https://www.ncbi.nlm.nih.gov/entrez/eutils/elink.fcgi?dbfrom=pubmed&tool=sumsearch.org/cite&retmode=ref&cmd=prlinks&id=31240976 }} </ref>([[ACC AHA guidelines classification scheme#Level of Evidence|Level of Evidence: B-R]])'' <nowiki>"</nowiki> | ||
====Beta Blockers==== | ====Beta Blockers==== | ||
Revision as of 13:50, 12 June 2022
| Resident Survival Guide |
| File:Critical Pathways.gif |
| Congestive Heart Failure Microchapters |
|
Pathophysiology |
|---|
|
Differentiating Congestive heart failure from other Diseases |
|
Diagnosis |
|
Treatment |
|
Medical Therapy: |
|
Surgical Therapy: |
|
ACC/AHA Guideline Recommendations
|
|
Specific Groups: |
|
Congestive heart failure treatment of patients with current or prior symptoms of heart failure (Stage C) On the Web |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Mahmoud Sakr, M.D. [2],Seyedmahdi Pahlavani, M.D. [3], Edzel Lorraine Co, D.M.D., M.D. [4]
Overview
Several measures [1] have been listed for patients in Stage A or B as previously mentioned and the Class 1 recommendations for those patients are also appropriate for patients with current or previous symptoms of HF. The effectiveness of diuretics and safety of their usage is an important aspect of the treatment that should be closely monitored along with daily measurement of weight and moderate sodium restriction. Physical activity is highly recommended, although heavy labor or sports shouldn’t be a part of it. A reduction in physical activity promotes physical deconditioning, and an increase of weight which may be associated with more strain on the failing heart.[2] Patients with chronic conditions such as HF are also recommended to be immunized with influenza and pneumococcal vaccines in order to reduce the risk of respiratory infection.
Drugs that should be Avoided or Used with Caution in Patients with HF
Pharmacological therapy should be closely monitored and several classes of drugs should be avoided in case of HF:
- Calcium Channel blockers: There is no direct role of these drugs in the management of HF, due to negative and possible deleterious effect in patients with HF due to systolic dysfunction[3]. Vasoelective Calcium Channel blockers as amlodipine and felodipine have not been linked to adverse effect in HF treatment, but there is no evidence of efficacy for these drugs in the management of HF.[4] However, amlodipine and felodipine appear to be safe for the treatment of concomitant disease in HF patients, such as angina or hypertension.
- Antiarrhythmic agents: Negative inotropic effect exerted by most antiarrhythmic drugs can precipitate HF in patients with reduced LV function. The reduction in LV function can also reduce the elimination of these drugs leading to further drug toxicity. Other antiarrhythmic drugs can induce some proarrhythmic effect, especially class 1 agents and class 3 agents Ibutilide and sotalol;[5] the same class 3 agents in addition to dofetilide can induce torsades to pointes.
Amiodarone is considered the safest of the antiarrhythmic drugs because of its minimal proarrhythmic effect and is generally the preferred drug for treating arrhythmias in HF patients.
- Nonsteroidal anti-inflammatory drugs (NSAID):[6] The administration of non-selective NSAIDs in HF patients is linked with an increased risk of HF exacerbation, increased renal dysfunction, and abnormal responses to ACEIs and diuretics. COX-2 selective inhibitors have not been fully investigated, but observational studies indicate that they may be linked with an increased rate of HF exacerbation and increased mortality.
Aspirin benefits and risks are not well established in patients with HF and Vascular disease (includingCAD). The potential interaction between ACEIs and beta blockers is of great importance. Although no data has proven that aspirin causes more frequent HF exacerbations and interactions with those drugs, health care providers should be aware of the possibility of such risks, but no recommendation for or against aspirin therapy in patients with heart failure can be made before further data are available.
- Oral Hypoglycemic agents:[7] two oral hypoglycemic agents, metformin and thiazolidinediones are considered to be risky in patients with HF.
Metformin - one of the most common side effects of metformin is lactic acidosis, which can be fatal in patients with HF.
Thiazolidinediones -[8] the biggest risk of using Thiazolidinediones is fluid retention which may cause severe worsening of patients with HF.
- Antidepressants: Depression is a common finding between patients suffering from HF that is usually related to high mortality rate and bad prognosis of those patients.[9] Limited data are available on the safety and the risks associated with the usage of antidepressants in patients with heart failure. Health care providers should be aware of major cardiovascular events (as HF, MI, Stroke, cardiovascular death ) that is associated with Tricyclic antidepressants (TCAs) and Selective serotonin reuptake inhibitors (SSRIs).
- Phosphodiesterase inhibitors PDE – The PDE-3 inhibitors as[10] Cilostazol and PDE-4 as [11] Anagrelide should be avoided in patients with HF, because of an increase risk of high-output heart failure and fluid retention that is associated with those drugs.[12]
PDE-5 inhibitors such as sildenafil, vardenafil, and tadalafil, are widely used in the management of erectile dysfunction in men. The use of those agents with any form of nitrate therapy is contraindicated because of severe hypotensive effect that can be life threatening.[13] In a trial where sildenafil and placebo were randomly assigned to 34 HF patients, no significant difference of symptomatic hypotension was observed, but HF patients with borderline low blood pressure and/or low volume status are in risk of severe hypotension and should avoid any PDE-5 inhibitors use.
- Chemotherapy – Cardiotoxic chemotherapeutic agents as Cyclophosphamide, Trastuzumab, Bevacizumab and Anthracyclines, should be avoided in HF patients [14]
- Tumor Necrosis Factor alpha inhibitors TNF-alpha: New onset or worsening of pre-existing heart failure have been linked to TNF-alpha inhibitors.[15] Infliximab has been specifically contraindicated in doses over 5mg/kg in patients with heart failure.
- Antihistamines:[16] some second generation antihistamines as terfenadine and astemizole have been reported to cause long QT syndrome and should not be used in HF patients.
Serum potassium should be closely monitored in HF patients, in order of preventing either hypokalemia or hyperkalemia, which could greatly affect cardiac excitability and conduction, leading to sudden cardiac death.[17] Serum potassium should be maintained between 4.0 to 5.0 mEq per liter range, because low potassium level may affect digitalis and antiarrhythmic drugs treatment, while high potassium level can prevent the use of treatments known to prolong life.[17]
Supervision of HF patients with close monitoring of treatment and diet is a very important aspect of the follow-up process in those individuals. Body weight and medications should be closely monitored, because any minor change in those parameters can have a significant effect over symptoms and hospitalization of patients with HF.[18] Patient education is a crucial aspect of the management of HF, patient and family surveillance over any new change of symptoms or body weight is important in allowing early detection of those changes and implementing new treatment strategies to reduce further complications.[19]
Pharmacological Therapy of HF
Improving symptoms, reducing mortality and slowing or reversing myocardial deterioration are the main goals of pharmacological therapy in HF patients. The therapy should be also directed at preventing arrhythmias, embolic events and other exacerbating factors. Different strategies have been implemented in the treatment of HF, but the ACC/AHA recommendations are based on a combination of 3 types of drugs: a Diuretic, an ACEI or an ARB, and a beta blocker[20]. A review of data from several trials, provide clear evidence supporting the central role of these drugs in HF patient Therapy. The Diuretics play an important role in controlling fluid retention and achieving a euvolemic state. On the other hand, ACEI and a beta blocker should be started and maintained in patients who can tolerate them because of their major effect in controlling symptoms and reducing mortality. Digoxin can be added to this regime as a fourth agent to reduce symptoms, reduce recurrent hospitalization, control great and rhythm, and enhance exercise tolerance.
Diuretics
Different classes of diuretics have been implicated in the treatment of CHF. Loop Diuretics such as furosemide, bumetanide and torsemide, act on the inhibition of sodium and chloride reabsorption in the loop of Henle, whereas thiazide diuretics, metolazone, and potassium sparing diuretics as spironolactone act on the distal portion of the renal tubule. [21]Loop diuretics increase sodium secretion up to 25 % of the filtered load of sodium, enhance free water clearance and maintain their efficacy unless severe renal impairment is present.[22]Thiazide diuretics in contrast, increase the fractional excretion of sodium to a maximum of 10% of the filtered load, decrease free water clearance and lose their efficacy in patients with impaired renal function. For such reasons, loop diuretics are much preferred, unless hypertension with mild fluid retention is present in HF patients, where thiazide diuretics may have a better antihypertensive effect in this case.
Different short-studies demonstrated the efficacy of diuretics in improving various symptoms of HF, reducing jugular venous pressure, pulmonary congestion, peripheral edema and body weight, all within days of initiation of therapy.[23] No long term studies have been conducted so far to reveal the effects of diuretic therapy on morbidity and mortality, but an improvement in cardiac function and exercise tolerance in patients with HF have been demonstrated in intermediate-term trials.[24]
Diuretics produce great symptomatic benefits,[25] much more rapidly than any other drugs for HF, they can adequately control the fluid retention of HF, relieving pulmonary edema and peripheral edema within hours or days. However, Diuretics should not be used alone in controlling Stage C HF. They should be combined with an ACEI and a beta blocker to avoid further clinical deterioration and maintain the HF symptoms under control.[24]
Appropriate dose of diuretics should used in treatment of HF,[26] because low doses will cause fluid retention, which can lead to a reduced response to ACEIs and ARBs and an increase risk of decompensation with the use of beta blockers, while excessive diuretics will lead to volume contraction which can increase the risk of hypotension and renal insufficiency with ACEIs, ARBs and beta blockers.[27]
Loop diuretics are usually the first diuretics to be used to control pulmonary and/or peripheral edema. Furosemide is the most commonly used, but some patients may respond favorably to other agents in this category (as torsemide and Bumetanide) because of superior absorption and longer time of action.[28]
The starting dosage is usually 20 to 40 mg of furosemide or its equivalent, but the exact dosing should always be monitored according to diuresis and other clinical symptoms, since the ultimate goal is to eliminate the evidence of fluid retention such as jugular venous pressure elevation and peripheral edema. Outpatients with HF are usually started with low dose until urine increases and weigh decreases by 0.5 to 1.0 kg daily. Diuretic therapy should be also combined with moderate dietary sodium restriction. Unstable or severe disease, should be controlled with intravenous diuretics( bolus or continuous infusion) and thiazide diuretics can be added for a synergistic effect.[29]
Reducing overall volume may decrease intracardiac filling pressure resulting in a lower cardiac output via the Frank-Starling relationship. This effect is usually a minor effect and does not alter the course of the therapy. On the other hand, an unexplained increase in BUN and creatinine should be closely monitored and suspected as a sign of abnormal tissue perfusion. In this case renal function and other end organ perfusion should be assessed to avoid any concomitant complications.
Angiotensin Converting Enzyme Inhibitors
Patients with moderate to severe HF or asymptomatic left ventricular dysfunction show a great improvement in survival rate with the usage of ACE inhibitors.[30] ACE inhibitors enhance the action of kinins and augment kinin-mediated prostaglandin production, so the effect of ACEIs cannot be solely explained by the suppression of Angiotensin II production. Furthermore, it has been proven that ACEIs modify cardiac remodeling more favorably than ARBs in experimental models of HF.[31] Several studies indicates that ACEIs can alleviate symptoms,[32] improve clinical status and sense of well being in patients with HF.
Another important aspect of ACEIs therapy is the reduction of mortality and hospitalization in such patients.[33] These benefits were noticed in patients with mild, moderate or even severe symptoms of HF, with or without CAD. In general ACEIs should be used together with a beta blocker and should not be used without diuretics in patients with a current or recent history of fluid retention, because of the important role of diuretics in maintaining sodium balance and preventing the development of peripheral and pulmonary edema.[24]
For the reasons mentioned above, ACEI therapy should be started in asymptomatic or symptomatic patients with HF. The beginning therapy should be a low dose ( eg, 2.5 mg of enalapril twice a day, 6.25 mg of captopril three time a day or 5 mg of lisinopril once a day). The dose should be gradually increased in one to two week if the initial therapy is tolerated and try to target a dose of 20 mg of enalapril twice a day, or 50 mg of captopril three times a day or up to 40 mg of lisinopril a day.[34] Plasma potassium and renal function should be assessed one to two weeks after starting or changing a dose and periodically thereafter. Physicians should attempt to target certain doses which have been proved to reduce the risk of cardiovascular events. If these target doses of ACEIs cannot be used or poorly tolerated than intermediate doses should be implemented. [35]
NSAIDs should be avoided since they may cause an increase in adverse effects of ACEIs in patients with HF and a decrease in the favorable effect of ACEIs therapy.[24] Some evidence suggests that aspirin may inhibit the acute hemodynamic benefits of ACEIs.[36] If the patient has a know history of coronary artery disease(CAD) then use of ASA along with ACEIs is recommended. However, but in patients with no history of CAD, there is no evidence to support the use of aspirin.
Treatment of HFrEF Stage C and D
Abbreviations:
ACEI: angiotensin-converting enzyme inhibitor, ARB: angiotensin receptor-blocker, ARNI: angiotensin
receptor-neprilysin inhibitor, BP: blood pressure, bpm: beats per minute, C/I: contraindication, COR: Class of
Recommendation, NT-proBNP: creatinine clearance, NYHA: cardiac resynchronization therapy–device, pts: diagnosis, HF: guideline-directed management and therapy, CrCl: creatinine clearance, CRT-D: cardiac resynchronization therapy–device, Dx: diagnosis, GDMT: guideline-directed management and therapy, HF: heart failure, HFrEF: heart failure with reduced ejection fraction, ISDN/HYD: isosorbide dinitrate hydral-nitrates, K+: potassium, LBBB: left bundle-branch block, LVAD: left ventricular assist device, LVEF: left ventricular ejection fraction, MI: myocardial infarction, NSR: normal sinus rhythm, NYHA: New York Heart Association
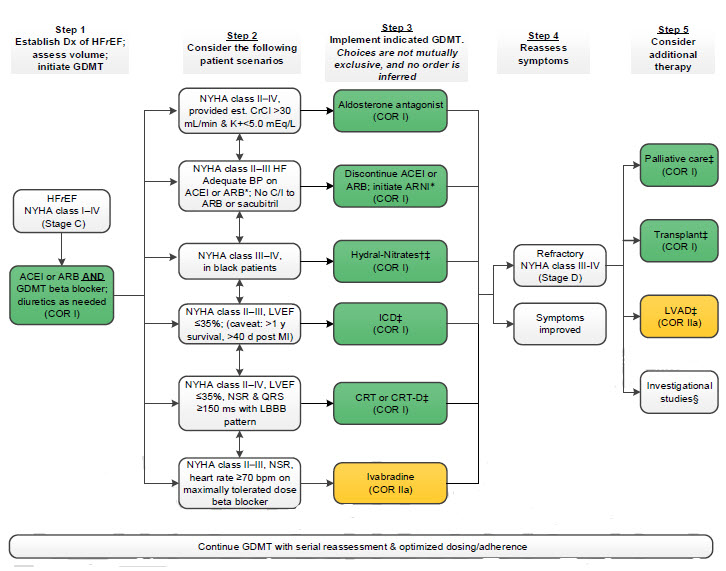
†Hydral-Nitrates green box: The combination of ISDN/HYD with ARNI has not been robustly tested. BP response should be carefully monitored.
§Participation in investigational studies is also appropriate for stage C, NYHA class II and III HF.
2022 AHA/ACC/HFSA Heart Failure Guideline/ 2017 ACC/AHA/HFSA Focused Update of the 2013 ACCF/AHA Guideline for the Management of Heart Failure/2016 ACC/AHA/HFSA Focused Update on New Pharmacological Therapy for Heart Failure: An Update of the 2013 ACCF/AHA Guideline,2013 ACC/AHA Guideline, 2009 ACC/AHA Focused Update and 2005 Guidelines for the Diagnosis and Management of Heart Failure in the Adult (DO NOT EDIT) [37][38][39]
Pharmacological Treatment for HFrEF (DO NOT EDIT)[37][38][39]
Renin-Angiotensin System Inhibition With ACEi or ARB or ARNi
| Class I | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| "1. In patients with HFrEF and NYHA class II to III symptoms, the use of ARNi is recommended to reduce morbidity and mortality. [40][41][42][43][44](Level of Evidence: A) " | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| "2. In patients with previous or current symptoms of chronic HFrEF, the use of ACEi is beneficial to reduce morbidity and mortality when the use of ARNi is not feasible.[45][46][34][47][48][49][33][50] (Level of Evidence: A) " | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| "3. In patients with previous or current symptoms of chronic HFrEF who are intolerant to ACEi because of cough or angioedema and when the use of ARNi is not feasible, the use of ARB is recommended to reduce morbidity and mortality. [51][52][53][54][55] (Level of Evidence: A) " | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| "5. In patients with chronic symptomatic HFrEF NYHA class II or III who tolerate an ACEi or ARB, replacement by an ARNi is recommended to further reduce morbidity and mortality. [40][41][42][43][44](Level of Evidence: B-R) "
Beta BlockersMineralocorticoid Receptor Antagonists (MRAs)Sodium-Glucose Contransporter 2 InhibitorsHydralazine and Isosorbide DinitrateOther Drug TreatmentDrugs of Unproven Value or That May Worsen HF
Treatment of Patients with HF and preserved ejection fraction HFpEF (DO NOT EDIT) [195]
Related ChaptersExternal Links
References
|
- Pages with reference errors
- Pages with citations using unsupported parameters
- CS1 maint: Multiple names: authors list
- Pages using citations with accessdate and no URL
- CS1 errors: PMID
- CS1 maint: Explicit use of et al.
- CS1 errors: PMC
- CS1 maint: PMC format
- Pages with broken file links
- Cardiology
- Disease
- Emergency medicine
- Intensive care medicine
- Medicine
- Up-To-Date
- Up-To-Date cardiology