Heart failure resident survival guide
For acute heart failure prevention click here.
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Mahmoud Sakr, M.D. [2]; Ayokunle Olubaniyi, M.B,B.S [3]; Rim Halaby, M.D. [4] Nehal Eid, M.D.[5]
| Acute Heart Failure Resident Survival Guide Microchapters |
|---|
| Overview |
| Classification |
| Causes |
| FIRE |
| Diagnosis |
| Treatment
Stage A and B |
| Do's |
| Don'ts |
Overview
Heart failure is a complex clinical syndrome characterized by impaired ventricular filling, impaired blood ejection, or both, leading to the inability of the heart to meet the metabolic demands of the body or to do so only at elevated filling pressures. The diagnosis relies on symptoms, physical examination findings, objective evidence of cardiac dysfunction, and supportive testing. The main symptoms and signs of heart failure include dyspnea, volume overload leading to pulmonary edema and/or peripheral edema, fatigue, and exercise intolerance. Acute decompensated heart failure (ADHF) is a life-threatening syndrome that can occur with new-onset heart failure or worsening of chronic heart failure. Symptoms and signs of ADHF may include dyspnea secondary to pulmonary edema, peripheral edema, hypotension, and impaired end-organ perfusion manifested by worsening renal function, altered mental status, oliguria, and cold clammy extremities.
The management of ADHF requires rapid identification of the precipitating cause, assessment of congestion and perfusion, oxygen or noninvasive ventilation when hypoxemia or respiratory distress is present, IV loop diuretic therapy for congestion, selective vasodilator therapy when blood pressure permits, and escalation to inotropes, vasopressors, invasive hemodynamic monitoring, or shock pathways when hypoperfusion is present. The goals of chronic heart failure treatment are to relieve symptoms, reduce hospitalization, improve survival, and prevent disease progression. Treatment of heart failure includes identification and management of precipitating factors, lifestyle modification, pharmacological therapy, device therapy when indicated, and referral for advanced therapies in selected patients.[1][2]
Classification
Classification by Severity of Congestive Heart Failure
Shown below is a table comparing American College of Cardiology Foundation/American Heart Association (ACCF/AHA) stages to New York Heart Association (NYHA) functional classification of heart failure severity.[3][1]
| ACCF/AHA Stages | New York Heart Association (NYHA) Classification | ||
| Stage | Interpretation | Class | Interpretation |
| A | At high risk for heart failure but without structural heart disease or symptoms/signs of HF | - | - |
| B | Pre-HF: structural heart disease, increased filling pressures, or risk markers, but without symptoms/signs of HF | I | No limitation of physical activity. Ordinary physical activity does not cause symptoms of HF |
| C | Structural heart disease with prior or current symptoms of HF | I | No limitation of physical activity. Ordinary physical activity does not cause symptoms of HF |
| II | Slight limitation of physical activity. Comfortable at rest, but ordinary physical activity results in symptoms of HF | ||
| III | Marked limitation of physical activity. Comfortable at rest, but less than ordinary activity causes symptoms of HF | ||
| IV | Unable to carry on any physical activity without symptoms of HF, or symptoms of HF at rest | ||
| D | Advanced HF with marked symptoms that interfere with daily life and recurrent hospitalizations despite attempts to optimize GDMT | IV | Unable to carry on any physical activity without symptoms of HF, or symptoms of HF at rest |
Classification by Other Factors
Left Ventricular Ejection Fraction (LVEF)
- Heart failure with reduced ejection fraction (HFrEF): left ventricular ejection fraction (LVEF) ≤40%
- Heart failure with mildly reduced ejection fraction (HFmrEF): LVEF 41–49%
- Heart failure with preserved ejection fraction (HFpEF): LVEF ≥50%
- Heart failure with improved ejection fraction (HFimpEF): previous LVEF ≤40% with follow-up LVEF >40%
Cardiac Output
- Low-output heart failure
- High-output heart failure or high cardiac output state
Left vs. Right Sided
- Left-sided heart failure: pulmonary edema, pulmonary congestion, and dyspnea
- Right-sided heart failure: peripheral edema, elevated jugular venous pressure, hepatomegaly, ascites, and systemic venous congestion
Backward vs. Forward Failure
- Backward failure: congestion and elevated filling pressures
- Forward failure: low systemic perfusion and end-organ hypoperfusion
Causes
Life-Threatening Causes
Acute decompensated heart failure is life-threatening and should be treated promptly irrespective of the underlying cause.
Common Causes
- Acute coronary syndrome
- Acute kidney injury
- Acute severe myocarditis
- Cardiac arrhythmias
- Cardiomyopathy
- Cardiotoxic agents, including alcohol, cocaine, and anthracyclines
- Decompensation of underlying chronic heart failure
- Hypertensive crisis
- Pulmonary embolus
- Systemic inflammatory response syndrome or systemic infection
- Valvular heart disease
- Medication nonadherence
- Excess sodium or fluid intake
- Medications that worsen HF, including NSAIDs, nondihydropyridine calcium channel blockers, and thiazolidinediones
Click here for the complete list of causes.
FIRE: Focused Initial Rapid Evaluation
A Focused Initial Rapid Evaluation (FIRE) should be performed to identify patients with severe acute decompensated heart failure in need of immediate intervention.[3][1][2]
Boxes in red signify that urgent management is needed.
Abbreviations:
BUN: Blood urea nitrogen;
COPD: Chronic obstructive pulmonary disease;
D5W: 5% dextrose solution in water;
HF: Heart failure;
IV: Intravenous;
MAP: Mean arterial pressure;
Na: Sodium;
NSAID: Non steroidal anti-inflammatory drug;
SBP: Systolic blood pressure;
S3: Third heart sound;
Identify cardinal findings that increase the pretest probability of acute decompensated heart failure ❑ Dyspnea | |||||||||||||||||
Does the patient have any of the following findings that require hospitalization and urgent management? ❑ Severe decompensated HF:
❑ Dyspnea at rest manifested by tachypnea or oxygen saturation <90% | |||||||||||||||||
Yes | No | ||||||||||||||||
Admit to a level of care that allows continuous ECG monitoring, frequent vital signs, oxygen monitoring, and rapid escalation if needed | |||||||||||||||||
Initial stabilization: Assess congestion and perfusion: Identify precipitating factor and treat accordingly:
❑ COPD Treat congestion and optimize volume status:
❑ Low sodium diet, commonly <2 g daily, unless contraindicated or individualized differently
❑ Do not use low-dose dopamine routinely to improve diuresis or renal blood flow; reserve vasoactive therapy for patients with hypotension, hypoperfusion, or shock Vasodilators
Treat low perfusion:
Invasive hemodynamic monitoring:
VTE prevention: Chronic medical therapy and GDMT during hospitalization:
❑ Do not initiate or uptitrate ACE inhibitors, ARBs, or ARNI during hemodynamic instability, shock, symptomatic hypotension, severe AKI, or clinically significant hyperkalemia Monitor laboratory tests: Management of hyponatremia:
❑ Optimize HF therapy and decongestion | |||||||||||||||||
Complete Diagnostic Approach
A complete diagnostic approach should be carried out after a focused initial rapid evaluation is conducted and following initiation of any urgent intervention.[3][4][1]
Abbreviations:
ANA: Antinuclear antibody;
ARDS: Acute respiratory distress syndrome;
BNP: B-type natriuretic peptide;
BUN: Blood urea nitrogen;
CAD: Coronary artery disease;
CBC: Complete blood count;
CCB: Calcium channel blocker;
CHF: Congestive heart failure;
CT: Computed tomography;
CXR: Chest X-ray;
DM: Diabetes mellitus;
ECG: Electrocardiogram;
JVP: Jugular venous pressure;
HF: Heart failure;
HTN: Hypertension;
LVEF: Left ventricular ejection fraction;
LVH: Left ventricular hypertrophy;
MI: Myocardial infarction;
MRI: Magnetic resonance imaging;
NT-proBNP: N-terminal pro-brain natriuretic peptide;
OCPs: Oral contraceptive pills;
PAWP: Pulmonary artery wedge pressure;
SBP: Systolic blood pressure;
S1: First heart sound;
S3: Third heart sound;
TSH: Thyroid stimulating hormone
Characterize the symptoms: Symptoms of left-sided fluid accumulation:
❑ Paroxysmal nocturnal dyspnea Obtain a detailed history:
❑ Medication history
❑ Family history
❑ Prior Radiation to the chest Determine the NYHA classification based on symptoms: | |||||||||||||||||||||||||||||||||
Examine the patient: General appearance: Vitals: ❑ Pulse
❑ Pulse oximetry; administer oxygen if hypoxemic, generally targeting SpO2 ≥90–92%, with individualized lower targets in patients at risk of hypercapnic respiratory failure Weight: Skin Neck examination: Respiratory examination Cardiovascular examination
Abdominal examination Extremity examination Neurological examination Determine status of congestion and perfusion based on physical exam:
Low perfusion at rest (warm vs. cold)
The patient is: | |||||||||||||||||||||||||||||||||
Order tests: Routine initial tests
❑ BNP or NT-proBNP, especially if the diagnosis is uncertain
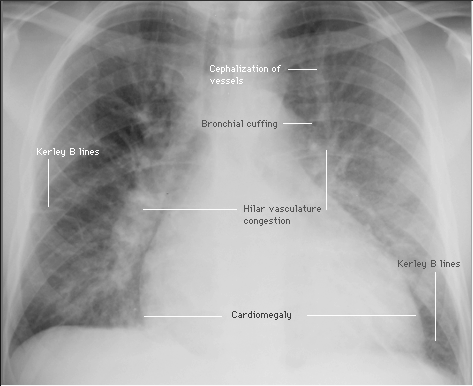 ❑ ECG to help identify the cause of heart failure
❑ 2-D echocardiography with Doppler
❑ Radionuclide ventriculography or MRI when echocardiography is inadequate or when myocardial scar, infiltrative disease, inflammation, or cardiomyopathy etiology requires further evaluation
Order additional tests selectively to evaluate alternative or specific etiologies: | |||||||||||||||||||||||||||||||||
Consider alternative diagnoses:
| |||||||||||||||||||||||||||||||||
Assess the stage of heart failure using the ACCF/AHA staging system to guide chronic therapy
| |||||||||||||||||||||||||||||||||
Stage C ❑ Patients with structural heart disease
❑ Prior or current signs or symptoms of heart failure | Stage D ❑ Advanced/refractory heart failure
| ||||||||||||||||||||||||||||||||
Prevention of Heart Failure in Stage A and B
Shown below is an algorithm depicting the management of stage A and B heart failure.[3][1]
Abbreviations: ACE I: Angiotensin converting enzyme inhibitor; ACS: Acute coronary syndrome; CVD: Cardiovascular disease; DM: Diabetes mellitus; EF: Ejection fraction; HF: Heart failure; HTN: Hypertension; ICD: Implantable cardioverter defibrillator; MI: Myocardial infarction; PAD: Peripheral artery disease; SGLT2: Sodium-glucose cotransporter 2
What is the stage of heart failure (HF)? | |||||||||||||||||||||||||||
| Stage A At high risk for HF but without structural heart disease or symptoms/signs of HF | Stage B / Pre-HF Structural heart disease, increased filling pressures, or risk markers but without symptoms/signs of HF | ||||||||||||||||||||||||||
❑ Encourage healthy lifestyle and regular physical activity | ❑ Encourage healthy lifestyle and regular physical activity | ||||||||||||||||||||||||||
Consider additional measures in selected patients: | |||||||||||||||||||||||||||
Treatment of Heart Failure in Stage C and D
Shown below is an algorithm depicting the management of stage C and D heart failure.[3][1][7]
Abbreviations: ACE I: Angiotensin converting enzyme inhibitor; ARB: Angiotensin II receptor blocker; ACS: Acute coronary syndrome; ARNI: Angiotensin receptor-neprilysin inhibitor; BID: Twice a day; BNP: Brain natriuretic peptide; CRT: Cardiac resynchronization therapy; CVD: Cardiovascular disease; DM: Diabetes mellitus; EF: Ejection fraction; GDMT: Guideline-directed medical therapy; GFR: Glomerular filtration rate; HF: Heart failure; HFimpEF: Heart failure with improved ejection fraction; HFmrEF: Heart failure with mildly reduced ejection fraction; HFrEF: Heart failure with reduced ejection fraction; HFpEF: Heart failure with preserved ejection fraction; HTN: Hypertension; ICD: Implantable cardioverter defibrillator; LVEF: Left ventricular ejection fraction; MCS: Mechanical circulatory support; MRA: Mineralocorticoid receptor antagonist; NYHA: New York Heart Association; MI: Myocardial infarction; PAD: Peripheral artery disease; SGLT2: Sodium-glucose cotransporter 2; TID: Three times a day

Foundational GDMT Framework
For patients with symptomatic chronic HFrEF, contemporary guideline-directed medical therapy is built around four foundational medication classes. When feasible, the usual strategy is to add all foundational classes before focusing on maximal uptitration of any single class, with individualized sequencing according to blood pressure, renal function, potassium, heart rate, congestion, and tolerability.[1][7]
- ARNI preferred, or ACE inhibitor/ARB when ARNI is not feasible
- Evidence-based beta blocker
- Mineralocorticoid receptor antagonist (MRA)
- SGLT2 inhibitor
Additional therapies such as hydralazine/isosorbide dinitrate, ivabradine, vericiguat, digoxin, ICD, CRT, cardiac rehabilitation, palliative care, mechanical circulatory support, or transplant evaluation are used in selected patients.
Medications
| Drug Class | Drug | Daily dose | Maximum daily dose / Notes |
|---|---|---|---|
| Loop diuretics | Furosemide (duration of action: 6 to 8 h) |
PO dose for chronic HF: 20 to 40 mg once or twice daily IV dose for acute HF:
Initial IV bolus administered slowly over 1 to 2 minutes, then continuous IV infusion rate individualized to response || Up to 600 mg/day may be used in severe edematous states with close monitoring; many hospitalized HF protocols escalate to high-dose furosemide equivalents based on response | |
| Bumetanide (duration of action: 4 to 6 h) |
PO dose for chronic HF: 0.5 to 1.0 mg once or twice daily IV dose may be used for acute HF; approximate equivalence: bumetanide 1 mg ≈ furosemide 40 mg |
10 mg/day | |
| Torsemide (duration of action: 12 to 16 h) |
PO dose for chronic HF: 10 to 20 mg once daily | 200 mg/day; torsemide is generally used orally and IV torsemide is not available in the United States | |
| Thiazide diuretics | Chlorothiazide (duration of action: 6 to 12 h) |
PO: 250 to 500 mg once or twice daily; IV chlorothiazide may be used in selected hospitalized patients with diuretic resistance | 1000 mg/day |
| Hydrochlorothiazide (duration of action: 6 to 12 h) |
PO: 25 mg once or twice daily | 200 mg/day | |
| Metolazone (duration of action: 12 to 24 h) |
PO: 2.5 mg once daily when used for sequential nephron blockade | 20 mg/day; monitor electrolytes closely | |
| K+-sparing diuretic | Amiloride (duration of action: 24 h) |
PO: 5 mg once daily | 20 mg/day; avoid routine combination with MRA because of hyperkalemia risk |
| Spironolactone (duration of action: 1 to 3 h) |
PO: 12.5 to 25.0 mg once daily | 50 mg/day; monitor potassium and renal function | |
| Triamterene (duration of action: 7 to 9 h) |
PO: 50 to 75 mg twice daily | 200 mg/day; avoid routine combination with MRA because of hyperkalemia risk | |
| ACE inhibitors | Enalapril | 2.5 mg twice daily | 10 to 20 mg twice daily |
| Lisinopril | 2.5 to 5 mg once daily | 20 to 40 mg once daily | |
| Ramipril | 1.25 to 2.5 mg once daily | 10 mg once daily | |
| ARBs | Candesartan | 4 to 8 mg once daily | 32 mg once daily |
| Losartan | 25 to 50 mg once daily | 50 to 150 mg once daily | |
| Valsartan | 20 to 40 mg twice daily | 160 mg twice daily | |
| ARNI Foundational GDMT for HFrEF |
Sacubitril/valsartan | 24 mg sacubitril/26 mg valsartan twice daily in patients with SBP 100–120 mm Hg, low prior ACE inhibitor/ARB dose, or renal/hepatic impairment; otherwise 49 mg sacubitril/51 mg valsartan twice daily | 97 mg sacubitril/103 mg valsartan twice daily; discontinue ACE inhibitor for at least 36 hours before starting ARNI |
| Beta blockers Foundational GDMT for HFrEF |
Bisoprolol | 1.25 mg once daily | 10 mg once daily |
| Carvedilol | 3.125 mg twice daily | 50 mg twice daily | |
| Carvedilol CR | 10 mg once daily | 80 mg once daily | |
| Metoprolol succinate extended release | 12.5 to 25.0 mg once daily | 200 mg once daily | |
| Aldosterone antagonists / MRAs Foundational GDMT for HFrEF |
Spironolactone | 12.5 to 25.0 mg once daily | 25 mg once or twice daily; monitor potassium and renal function |
| Eplerenone | 25 mg once daily | 50 mg once daily | |
| SGLT2 inhibitors Foundational GDMT for HFrEF; recommended in HFmrEF/HFpEF to reduce HF hospitalization and cardiovascular events |
Dapagliflozin | 10 mg once daily | No titration required |
| Empagliflozin | 10 mg once daily | No titration required | |
| Inotropes | Dopamine | 5 to 10 mcg/kg/min, OR 10 to 15 mcg/kg/min depending on hemodynamic goal | Reserve for selected patients with hypotension or shock; not recommended routinely for renal protection or enhanced diuresis |
| Dobutamine | 2.5 to 5 mcg/kg/min initially; titrate as needed, often 5 to 20 mcg/kg/min | Use for low-output states or cardiogenic shock when indicated | |
| Milrinone | 0.125 to 0.75 mcg/kg/min | Adjust for renal function; loading dose often avoided in unstable or hypotensive patients | |
| Vasodilators | Nitroglycerin | 5 to 10 mcg/min initially; increase by 5 to 10 mcg/min every 3 to 5 minutes as tolerated | May be titrated to high doses in hypertensive pulmonary edema with close blood pressure monitoring |
| Nitroprusside | 0.3 mcg/kg/min initially; titrate carefully according to blood pressure and clinical response | Common range 0.3 to 5 mcg/kg/min; high-dose or prolonged use increases cyanide/thiocyanate toxicity risk; close monitoring required | |
| Nesiritide | 2 mcg/kg bolus; then 0.01 mcg/kg/min continuous infusion | Max 0.03 mcg/kg/min; not routinely used because outcome benefit is not established | |
| Digoxin | Digoxin |
Loading dose: usually not required for chronic HF; if used for rate control, individualize by renal function, age, and urgency | |
| Add-On Therapies Beyond the Four Pillars 1. Hydralazine and isosorbide dinitrate |
Fixed-dose combination | 37.5 mg hydralazine/20 mg isosorbide dinitrate 3 times daily | 75 mg hydralazine/40 mg isosorbide dinitrate 3 times daily |
| Individual doses | Hydralazine: 25 to 100 mg 3 or 4 times daily Isosorbide dinitrate: 20 to 40 mg 3 times daily |
Hydralazine: 300 mg daily in divided doses Isosorbide dinitrate: 120 mg daily in divided doses | |
| 2. Ivabradine | Ivabradine | 5 mg twice daily with meals (2.5 mg twice daily if history of conduction disease or bradycardia risk) | 7.5 mg twice daily; titrate to resting heart rate 50-60 bpm when indicated |
| 3. Vericiguat | Vericiguat | 2.5 mg once daily | 10 mg once daily |
Referral for Advanced Therapies
The "I-NEED-HELP" mnemonic identifies patients who may benefit from referral to an advanced HF center:[8]
- Inotropes (previous or ongoing requirement)
- NYHA class III-IV or persistently elevated natriuretic peptides
- End-organ dysfunction (worsening renal/hepatic function)
- Ejection fraction ≤35%
- Defibrillator shocks (recurrent)
- Hospitalizations (>1 HF hospitalization in 12 months)
- Edema (refractory despite escalating diuretics)
- Low blood pressure (SBP consistently ≤90 mm Hg)
- Progressive intolerance to GDMT
Specific referral triggers include:[1]
- ≥2 HF hospitalizations or ED visits in 12 months
- Progressive decline in renal function
- Intolerance to RAAS inhibitors due to hypotension or renal dysfunction
- Inability to walk 1-2 blocks due to dyspnea or fatigue
- Worsening right HF or secondary pulmonary hypertension
- Frequent SBP ≤90 mm Hg
- Cardiac cachexia
- Persistent hyponatremia (Na <134 mEq/L)
- Refractory or recurrent ventricular arrhythmias; frequent ICD shocks
- Predicted 1-year mortality >20% using validated risk models when available
The "Golden Window" for referral: Patients should be referred before progressive or irreversible end-organ damage has occurred.
Do's
Acute Decompensated Heart Failure
- Differentiate systolic and diastolic heart failure among patients with ADHF in order to guide therapy:
- Inotropic agents are not used routinely in ADHF, but may be indicated in patients with systolic dysfunction and evidence of hypoperfusion, low cardiac output, or cardiogenic shock.
- While beta blocker initiation is generally avoided during the unstable or hypoperfused phase of acute decompensated systolic heart failure, control of tachycardia may be useful in selected stable patients, especially those with diastolic heart failure, to prolong left ventricular filling time.
- ACE inhibitors, ARBs, or ARNI may be continued, resumed, or initiated once the patient is hemodynamically stable and renal function and potassium are acceptable; avoid initiation or uptitration during shock, symptomatic hypotension, severe AKI, or clinically significant hyperkalemia.
- Rely on the patient's volume status, congestion, renal function, perfusion, and urine output to guide the aggressiveness of diuresis in ADHF.
- Continue chronic medications during acute decompensation in the following conditions:
- ACE inhibitor/ARB/ARNI: may be continued if the patient is hemodynamically stable without severe AKI, symptomatic hypotension, or clinically significant hyperkalemia
- Beta blocker: may be continued in the absence of hypotension, shock, severe bradycardia, or need for escalating inotropes/vasopressors
- Aldosterone antagonist: may be continued in the absence of hypotension, hyperkalemia, and significant renal dysfunction
- Digoxin may reduce heart failure hospitalizations but has not shown a mortality benefit in HFrEF. It can be used in selected patients with HFrEF and atrial fibrillation to help reduce ventricular response, especially when beta blockers are not tolerated or insufficient. Lower serum digoxin concentrations are generally preferred, particularly in older patients and patients with renal impairment. Drugs that increase the concentration of digoxin include amiodarone, quinidine, and verapamil.[9][10][11][12][13][14]
- DVT prophylaxis unless contraindicated.[15][16]
- Consider adding another diuretic, such as metolazone or a thiazide-type diuretic, for worsening congestion despite adequate loop diuretic escalation, with close electrolyte and renal monitoring.[17][18]
- Daily serum electrolytes, BUN, creatinine, and magnesium should be measured during IV diuresis or active titration of heart failure medications.
- Convert IV diuretics to oral therapy in anticipation of discharge once the patient is clinically stable and adequately decongested.
- Schedule early follow-up visit within 7 to 14 days and early telephone follow-up within 2 to 3 days of hospital discharge.[19][20]
Chronic Heart Failure
- Guideline-directed medical therapy (GDMT) for HFrEF includes four foundational medication classes: ARNI/ACE inhibitor/ARB, evidence-based beta blocker, mineralocorticoid receptor antagonist, and SGLT2 inhibitor. Additional therapies, such as hydralazine/isosorbide dinitrate, ivabradine, vericiguat, digoxin, ICD, CRT, cardiac rehabilitation, palliative care, or advanced HF therapies, are used in selected patients.[1][7]
- Order an echocardiogram as soon as possible in the absence of a recent one or if the patient's clinical status is deteriorating.
- Continue GDMT in patients with HFimpEF unless contraindicated, because withdrawal may lead to relapse of LV dysfunction and HF.
Don'ts
- Avoid, if possible, NSAIDs, sympathomimetics, tricyclic antidepressants, class I antiarrhythmics, most class III antiarrhythmics except amiodarone when appropriate, and nondihydropyridine calcium channel blockers such as diltiazem and verapamil in patients with HFrEF or acute decompensated HF, as they can be harmful.[21][22][23][24][25][26]
- Do not administer parenteral inotropes in normotensive patients with acute decompensated HF without evidence of decreased organ perfusion or shock.[27]
- Do not use low-dose dopamine routinely to improve diuresis or renal blood flow.
- Do not combine an ACEI, ARB, and aldosterone antagonist in patients with HFrEF unless there is a compelling specialist-directed indication, because this combination increases risk of renal dysfunction and hyperkalemia.
- Do not use aldosterone receptor antagonists in patients with hyperkalemia or significant renal insufficiency when serum creatinine is more than 2.5 mg/dL in men or more than 2.0 mg/dL in women, estimated glomerular filtration rate <30 mL/min/1.73 m2, or potassium more than 5.0 mEq/L.[28][29]
- Do not use statins routinely for heart failure alone without another indication such as ASCVD, diabetes, or primary prevention based on cardiovascular risk.[30][31]
- Do not administer K+-sparing diuretics such as amiloride or triamterene together with an aldosterone antagonist unless specifically directed and closely monitored, because of elevated risk of hyperkalemia.
- Do not initiate or uptitrate RAAS inhibitors, ARNI, beta blockers, or MRA during shock, severe hypotension, severe AKI, or clinically significant hyperkalemia; wait until the patient is clinically stable and monitoring is in place.
- Do not delay advanced HF referral until irreversible end-organ dysfunction has developed.
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, Deswal A, Drazner MH, Dunlay SM, Evers LR, Fang JC, Fedson SE, Fonarow GC, Hayek SS, Hernandez AF, Khazanie P, Kittleson MM, Lee CS, Link MS, Milano CA, Nnacheta LC, Sandhu AT, Stevenson LW, Vardeny O, Vest AR, Yancy CW (2022). "2022 AHA/ACC/HFSA Guideline for the Management of Heart Failure". Circulation. 145 (18): e895–e1032. doi:10.1161/CIR.0000000000001063. PMID 35363499 Check
|pmid=value (help). - ↑ 2.0 2.1 Hollenberg SM, Stevenson LW, Ahmad T, Bozkurt B, Butler J, Davis LL, Drazner MH, Kirkpatrick JN, Morris AA, Page RL 2nd, Siddiqi HK, Storrow AB, Teerlink JR (2024). "2024 ACC Expert Consensus Decision Pathway on Clinical Assessment, Management, and Trajectory of Patients Hospitalized With Heart Failure Focused Update". J Am Coll Cardiol. 84 (13): 1241–1267. doi:10.1016/j.jacc.2024.06.002. PMID 39127954 Check
|pmid=value (help). - ↑ 3.0 3.1 3.2 3.3 3.4 Yancy CW, Jessup M, Bozkurt B, Butler J, Casey DE, Drazner MH; et al. (2013). "2013 ACCF/AHA guideline for the management of heart failure: executive summary: a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines". Circulation. 128 (16): 1810–52. doi:10.1161/CIR.0b013e31829e8807. PMID 23741057.
- ↑ Hunt SA, Abraham WT, Chin MH, Feldman AM, Francis GS, Ganiats TG; et al. (2009). "2009 Focused update incorporated into the ACC/AHA 2005 Guidelines for the Diagnosis and Management of Heart Failure in Adults". J Am Coll Cardiol. 53 (15): e1–e90. doi:10.1016/j.jacc.2008.11.013. PMID 19358937.
- ↑ Perna, ER; Macín, SM; Parras, JI; Pantich, R; Farías, EF; Badaracco, JR; Jantus, E; Medina, F; Brizuela, M (2002). "Cardiac troponin T levels are associated with poor short- and long-term prognosis in patients with acute cardiogenic pulmonary edema". Am Heart J. 143 (5): 814–20. PMID 12040342. Unknown parameter
|month=ignored (help) - ↑ McMurray JJ, Adamopoulos S, Anker SD, Auricchio A, Böhm M, Dickstein K; et al. (2012). "ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012". Eur Heart J. 33 (14): 1787–847. doi:10.1093/eurheartj/ehs104. PMID 22611136.
- ↑ 7.0 7.1 7.2 7.3 Maddox TM, Januzzi JL Jr, Allen LA, Breathett K, Brouse S, Butler J, Davis LL, Fonarow GC, Ibrahim NE, Lindenfeld J, Masoudi FA, Motiwala SR, Oliveros E, Walsh MN, Wasserman A, Yancy CW, Youmans QR (2024). "2024 ACC Expert Consensus Decision Pathway for Treatment of Heart Failure With Reduced Ejection Fraction". J Am Coll Cardiol. 83 (15): 1444–1488. doi:10.1016/j.jacc.2023.12.024. PMID 38466244 Check
|pmid=value (help). - ↑ van der Meer P, Gaggin HK, Dec GW (2019). "ACC/AHA Versus ESC Guidelines on Heart Failure: JACC Guideline Comparison". J Am Coll Cardiol. 73 (21): 2756–2768. doi:10.1016/j.jacc.2019.03.478. PMID 31146820.
- ↑ The Captopril-Digoxin Multicenter Research Group. Comparative effects of therapy with captopril and digoxin in patients with mild to moderate heart failure. JAMA. 1988;259:539–44.
- ↑ Dobbs SM, Kenyon WI, Dobbs RJ. Maintenance digoxin after an episode of heart failure: placebo-controlled trial in outpatients. Br Med J. 1977;1:749–52
- ↑ Lee DC, Johnson RA, Bingham JB, et al. Heart failure in outpatients: a randomized trial of digoxin versus placebo. N Engl J Med. 1982;306:699–705.
- ↑ Guyatt GH, Sullivan MJ, Fallen EL, et al. A controlled trial of digoxin in congestive heart failure. Am J Cardiol. 1988;61:371–5.
- ↑ Uretsky BF, Young JB, Shahidi FE, et al., for the PROVED Investigative Group. Randomized study assessing the effect of digoxin withdrawal in patients with mild to moderate chronic congestive heart failure: results of the PROVED trial. J Am Coll Cardiol. 1993;22:955–62.
- ↑ Packer M, Gheorghiade M, Young JB, et al. Withdrawal of digoxin from patients with chronic heart failure treated with angiotensin-converting enzyme inhibitors. RADIANCE Study. N Engl J Med. 1993;329:1–7.
- ↑ Alikhan R, Cohen AT, Combe S, Samama MM, Desjardins L, Eldor A; et al. (2003). "Prevention of venous thromboembolism in medical patients with enoxaparin: a subgroup analysis of the MEDENOX study". Blood Coagul Fibrinolysis. 14 (4): 341–6. PMID 12945875.
- ↑ Guyatt GH, Akl EA, Crowther M, Gutterman DD, Schuünemann HJ, American College of Chest Physicians Antithrombotic Therapy and Prevention of Thrombosis Panel (2012). "Executive summary: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed". Chest. 141 (2 Suppl): 7S–47S. doi:10.1378/chest.1412S3. PMC 3278060. PMID 22315257.
- ↑ Grosskopf I, Rabinovitz M, Rosenfeld JB (1986). "Combination of furosemide and metolazone in the treatment of severe congestive heart failure". Isr J Med Sci. 22 (11): 787–90. PMID 3793436.
- ↑ Rosenberg J, Gustafsson F, Galatius S, Hildebrandt PR (2005). "Combination therapy with metolazone and loop diuretics in outpatients with refractory heart failure: an observational study and review of the literature". Cardiovasc Drugs Ther. 19 (4): 301–6. doi:10.1007/s10557-005-3350-2. PMID 16189620.
- ↑ Krumholz HM, Chen YT, Wang Y, Vaccarino V, Radford MJ, Horwitz RI (2000). "Predictors of readmission among elderly survivors of admission with heart failure". Am Heart J. 139 (1 Pt 1): 72–7. PMID 10618565.
- ↑ Hernandez AF, Greiner MA, Fonarow GC, Hammill BG, Heidenreich PA, Yancy CW; et al. (2010). "Relationship between early physician follow-up and 30-day readmission among Medicare beneficiaries hospitalized for heart failure". JAMA. 303 (17): 1716–22. doi:10.1001/jama.2010.533. PMID 20442387.
- ↑ Heerdink ER, Leufkens HG, Herings RM, et al. NSAIDs associated with increased risk of congestive heart failure in elderly patients taking diuretics. Arch Intern Med. 1998;158:1108–12.
- ↑ Herchuelz A, Derenne F, Deger F, et al. Interaction between nonsteroidal anti-inflammatory drugs and loop diuretics: modulation by sodium balance. J Pharmacol Exp Ther. 1989;248:1175–81.
- ↑ Gottlieb SS, Robinson S, Krichten CM, et al. Renal response to indomethacin in congestive heart failure secondary to ischemic or idiopathic dilated cardiomyopathy. Am J Cardiol. 1992;70:890–3.
- ↑ The Cardiac Arrhythmia Suppression Trial (CAST) Investigators. Preliminary report: effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction. N Engl J Med. 1989;321:406–12.
- ↑ The Cardiac Arrhythmia Suppression Trial II Investigators. Effect of the antiarrhythmic agent moricizine on survival after myocardial infarction. N Engl J Med. 1992;327:227–33.
- ↑ Pratt CM, Eaton T, Francis M, et al. The inverse relationship between baseline left ventricular ejection fraction and outcome of antiarrhythmic therapy: a dangerous imbalance in the risk-benefit ratio. Am Heart J. 1989;118:433–40.
- ↑ Cuffe MS, Califf RM, Adams KF, Benza R, Bourge R, Colucci WS, Massie BM, O'Connor CM, Pina I, Quigg R, Silver MA, Gheorghiade M (2002). "Short-term intravenous milrinone for acute exacerbation of chronic heart failure: a randomized controlled trial". JAMA. 287 (12): 1541–7. PMID 11911756. Retrieved 2012-04-06. Unknown parameter
|month=ignored (help) - ↑ Juurlink DN, Mamdani MM, Lee DS, Kopp A, Austin PC, Laupacis A; et al. (2004). "Rates of hyperkalemia after publication of the Randomized Aldactone Evaluation Study". N Engl J Med. 351 (6): 543–51. doi:10.1056/NEJMoa040135. PMID 15295047.
- ↑ Bozkurt B, Agoston I, Knowlton AA (2003). "Complications of inappropriate use of spironolactone in heart failure: when an old medicine spirals out of new guidelines". J Am Coll Cardiol. 41 (2): 211–4. PMID 12535810.
- ↑ Horwich TB, MacLellan WR, Fonarow GC (2004). "Statin therapy is associated with improved survival in ischemic and non-ischemic heart failure". J Am Coll Cardiol. 43 (4): 642–8. doi:10.1016/j.jacc.2003.07.049. PMID 14975476.
- ↑ Gissi-HF Investigators. Tavazzi L, Maggioni AP, Marchioli R, Barlera S, Franzosi MG; et al. (2008). "Effect of rosuvastatin in patients with chronic heart failure (the GISSI-HF trial): a randomised, double-blind, placebo-controlled trial". Lancet. 372 (9645): 1231–9. doi:10.1016/S0140-6736(08)61240-4. PMID 18757089.