Terfenadine
 | |
| Clinical data | |
|---|---|
| Trade names | Seldane, Triludan, Teldane |
| AHFS/Drugs.com | Multum Consumer Information |
| MedlinePlus | a600034 |
| Pregnancy category |
|
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | 70% |
| Elimination half-life | 3.5 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
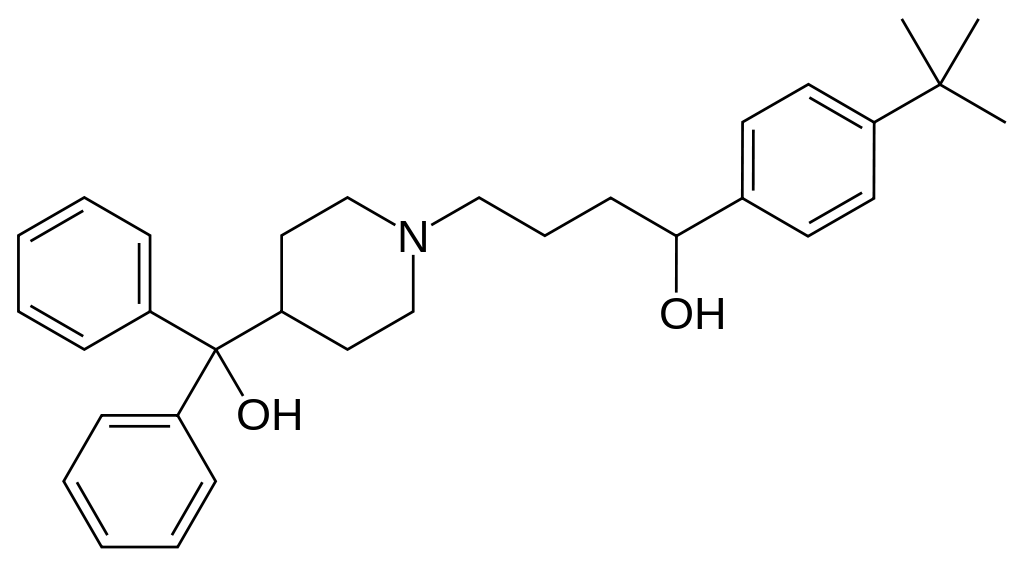
| Formula | C32H41NO2 |
| Molar mass | 471.673 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Terfenadine |
|
Articles |
|---|
|
Most recent articles on Terfenadine Most cited articles on Terfenadine |
|
Media |
|
Powerpoint slides on Terfenadine |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Terfenadine at Clinical Trials.gov Clinical Trials on Terfenadine at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Terfenadine
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Terfenadine Discussion groups on Terfenadine Patient Handouts on Terfenadine Directions to Hospitals Treating Terfenadine Risk calculators and risk factors for Terfenadine
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Terfenadine |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Terfenadine is an antihistamine formerly used for the treatment of allergic conditions. It was brought to market by Hoechst Marion Roussel (now Sanofi-Aventis) and marketed under various brand names, including Seldane in the United States, Triludan in the United Kingdom, and Teldane in Australia. According to its manufacturer, terfenadine had been used by over 100 million patients worldwide as of 1990.It was superseded by fexofenadine in the 1990s due to the risk of a particular type of disruption of the electrical rhythms of the heart (specifically cardiac arrhythmia caused by QT interval prolongation).
Terfenadine is a prodrug, generally completely metabolized to the active form fexofenadine in the liver by the enzyme cytochrome P450 CYP3A4 isoform. Due to its near complete metabolism by the liver immediately after leaving the gut, terfenadine normally is not measurable in the plasma. Terfenadine itself, however, is cardiotoxic at higher doses, while its major active metabolite is not. Terfenadine, in addition to its antihistamine effects, also acts as a potassium channel blocker (Kv11.1 encoded by the gene hERG). Since its active metabolite is not a potassium channel blocker, no cardiotoxicity is associated with fexofenadine.Toxicity is possible after years of continued use with no previous problems as a result of an interaction with other medications such as erythromycin, or foods such as grapefruit. The addition of, or dosage change in, these CYP3A4 inhibitors makes it harder for the body to metabolize and remove terfenadine. In larger plasma concentrations, it may lead to toxic effects on the heart's rhythm (e.g. ventricular tachycardia and torsades de pointes).
History
In the United States, Seldane was brought to market in 1985 as the first nonsedating antihistamine for the treatment of allergic rhinitis.In June 1990, evidence of serious ventricular arrhythmias among those taking Seldane prompted the FDA to issue a report on the risk factors associated with concomitant use of the drug with macrolide antibiotics and ketoconazole.Two months later, the FDA required the manufacturer to send a letter to all physicians, alerting them to the problem; in July 1992, the existing precautions were elevated to a black box warningand the issue attracted mass media attention in reports that people with liver disease or who took ketoconazole, an antifungal agent, or the antibiotic erythromycin, could suffer cardiac arrhythmia if they also took Seldane.
In January 1997, the same month when the U.S. Food and Drug Administration (FDA) had earlier approved a generic version of Seldane made by IVAX Corporation of Miami, the FDA recommended terfenadine-containing drugs be removed from the market and physicians consider alternative medications for their patients.Seldane (and Seldane-D, terfenadine combined with the decongestant pseudoephedrine) were removed from the U.S. market by their manufacturer in late 1997 after the FDA approval of Allegra-D (fexofenadine/pseudoephedrine). >"FDA Approves Allegra-D, Manufacturer To Withdraw Seldane From Marketplace". Food and Drug Administration. Archived from <refs/topics/ANSWERS/ANS00843.html the original on 2008-02-23. Retrieved 2010-11-11.</ref> Terfenadine-containing drugs were subsequently removed from the Canadian market in 1999,[1]
See also
References
- ↑ Status of Terfenadine-Containing Drugs in Canada from Health CanadaTerfenadine- General Practice notebook from GPnotebook.co.uk
<ref> tag with name "jama96" defined in <references> is not used in prior text.
<ref> tag with name "ladainews" defined in <references> is not used in prior text.
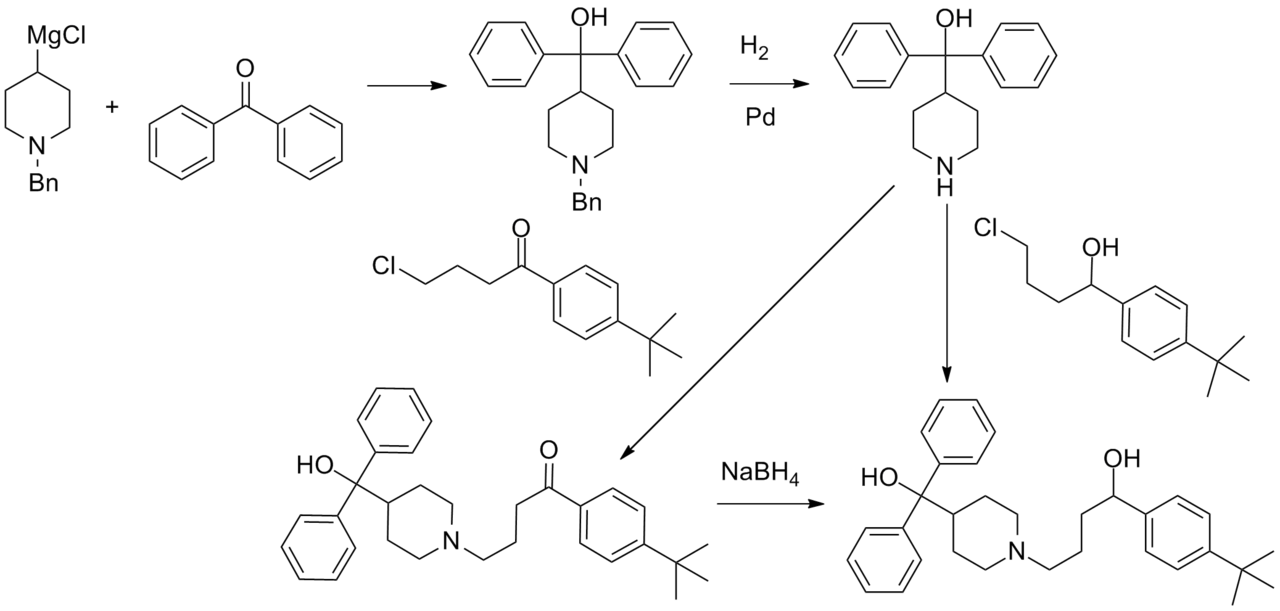
<ref> tag with name "Cir1996" defined in <references> is not used in prior text.Synthesis
External links
- Hoechst Marion Roussel Committed to Education Regarding Seldane Usage, an April 30, 1996 press release
- ↑ Arzneimittel-Forsch 32, 1157 (1982).
- Pages with script errors
- Pages with reference errors
- Template:drugs.com link with non-standard subpage
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Articles with changed InChI identifier
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles with non-default infobox title
- Drugboxes which contain changes to verified fields
- H1 receptor antagonists
- Prodrugs
- Piperidines
- Alcohols
- Withdrawn drugs