Digoxin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alonso Alvarado, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Digoxin is a cardiac glycoside that is FDA approved for the treatment of atrial fibrillation, atrial flutter, and heart failure. Common adverse reactions include dizziness, headache, mental disorder, nausea, and vomiting.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Heart failure
- Dosing Information
- Loading dose: 10-15 mcg/kg PO administer half the total loading dose initially, then ¼ the loading dose every 6 to 8 hours twice.
- 8-12 mcg/kg IV administer half the total loading dose initially, then ¼ the loading dose every 6 to 8 hours twice.
- Maintenance dose: 3.4-5.1 mcg/kg/day PO once daily.
- 2.4-3.6 mcg/kh/day IV once daily.
Atrial fibrillation
- Dosing Information
- Loading dose: 10-15 mcg/kg PO administer half the total loading dose initially, then ¼ the loading dose every 6 to 8 hours twice.
- 8-12 mcg/kg IV administer half the total loading dose initially, then ¼ the loading dose every 6 to 8 hours twice.
- Maintenance dose: 3.4-5.1 mcg/kg/day PO once daily.
- 2.4-3.6 mcg/kh/day IV once daily.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of SandboxAlonso in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of SandboxAlonso in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Heart failure
- Dosing Information
- Loading dose
- 5-10 years old: 20-45 mcg/kg PO administer half the total loading dose initially, then ¼ the loading dose every 6 to 8 hours twice.
- >10 year old: 10-15 mcg/kg PO administer half the total loading dose initially, then ¼ the loading dose every 6 to 8 hours twice.
- Maintenence dose:
- 5-10 years: 6.4 – 12.9 mcg/kg/day PO or 3.2 – 6.4 mcg/kg/day Twice daily.
- More than 10 years: 3.4 – 5.1 mcg/kg/day PO.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of SandboxAlonso in pediatric patients.
Non–Guideline-Supported Use
Supraventricular tachycardia, Recurrent; Prophylaxis
- Dosage information
Fetal tachycardia - Supraventricular tachycardia
- Dosage information
Contraindications
- Ventricular fibrillation
- Known hypersensitivity to digoxin (reactions seen include unexplained rash, swelling of the mouth, lips or throat or a difficulty in breathing). A hypersensitivity reaction to other digitalis preparations usually constitutes a contraindication to digoxin.
Warnings
Ventricular Fibrillation in Patients With Accessory AV Pathway (Wolff-Parkinson-White Syndrome)
Patients with Wolff-Parkinson-White syndrome who develop atrial fibrillation are at high risk of ventricular fibrillation. Treatment of these patients with digoxin leads to greater slowing of conduction in the atrioventricular node than in accessory pathways, and the risks of rapid ventricular response leading toventricular fibrillation are thereby increased.
Sinus Bradycardia and Sino-atrial Block
Digoxin may cause severe sinus bradycardia or sino-atrial block particularly in patients with pre-existing sinus node disease and may cause advanced or complete heart block in patients with pre-existing incomplete AV block. Consider insertion of a pacemaker before treatment with digoxin.
Digoxin Toxicity
Signs and symptoms of digoxin toxicity include anorexia, nausea, vomiting, visual changes and cardiac arrhythmias [first-degree, second-degree (Wenckebach), or third-degree heart block (including asystole); atrial tachycardia with block; AV dissociation; accelerated junctional (nodal) rhythm; unifocal or multiform ventricular premature contractions (especially bigeminy or trigeminy); ventricular tachycardia; and ventricular fibrillation]. Toxicity is usually associated with digoxin levels greater than 2 ng/mL although symptoms may also occur at lower levels. Low body weight, advanced age or impaired renal function, hypokalemia, hypercalcemia, or hypomagnesemia may predispose to digoxin toxicity. Obtain serum digoxin levels in patients with signs or symptoms of digoxin therapy and interrupt or adjust dose if necessary. Assess serum electrolytes and renal function periodically. The earliest and most frequent manifestation of digoxin toxicity in infants and children is the appearance of cardiac arrhythmias, including sinus bradycardia. In children, the use of digoxin may produce any arrhythmia The most common are conduction disturbances or supraventricular tachyarrhythmias, such as atrial tachycardia (with or without block) and junctional (nodal) tachycardia. Ventricular arrhythmias are less common. Sinus bradycardia may be a sign of impending digoxin intoxication, especially in infants, even in the absence of first-degree heart block. Any arrhythmias or alteration in cardiac conduction that develops in a child taking digoxin should initially be assumed to be a consequence of digoxin intoxication.
Misidentification of Digoxin Toxicity
Given that adult patients with heart failure have some symptoms in common with digoxin toxicity, it may be difficult to distinguish digoxin toxicity from heart failure. Misidentification of their etiology might lead the clinician to continue or increase digoxin dosing, when dosing should actually be suspended. When the etiology of these signs and symptoms is not clear, measure serum digoxin levels.
Risk of Ventricular Arrhythmias During Electrical Cardioversion
It may be desirable to reduce the dose of or discontinue digoxin for 1-2 days prior to electrical cardioversion of atrial fibrillation to avoid the induction of ventricular arrhythmias, but physicians must consider the consequences of increasing the ventricular response if digoxin is decreased or withdrawn. If digitalis toxicity is suspected, elective cardioversion should be delayed. If it is not prudent to delay cardioversion, the lowest possible energy level should be selected to avoid provoking ventricular arrhythmias.
Risk of Ischemia in Patients With Acute Myocardial Infarction
Digoxin is not recommended in patients with acute myocardial infarction because digoxin may increase myocardial oxygen demand and lead to ischemia.
Vasoconstriction in Patients With Myocarditis
Digoxin can precipitate vasoconstriction and may promote production of pro-inflammatory cytokines; therefore, avoid use in patients with myocarditis.
Decreased Cardiac Output in Patients With Preserved Left Ventricular Systolic Function
Patients with heart failure associated with preserved left ventricular ejection fraction may experience decreased cardiac output with use of digoxin. Such disorders include restrictive cardiomyopathy, constrictive pericarditis, amyloid heart disease, and acutecor pulmonale. Patients with idiopathic hypertrophic subaortic stenosis may have worsening of the outflow obstruction due to the inotropic effects of digoxin. Patients with amyloid heart disease may be more susceptible to digoxin toxicity at therapeutic levels because of an increased binding of digoxin to extracellular amyloid fibrils. Digoxin should generally be avoided in these patients, although it has been used for ventricular rate control in the subgroup of patients with atrial fibrillation.
Reduced Efficacy in Patients With Hypocalcemia
Hypocalcemia can nullify the effects of digoxin in humans; thus, digoxin may be ineffective until serum calcium is restored to normal. These interactions are related to the fact that digoxin affects contractility and excitability of the heart in a manner similar to that of calcium.
Altered Response in Thyroid Disorders and Hypermetabolic States
Hypothyroidism may reduce the requirements for digoxin. Heart failure and/or atrial arrhythmias resulting from hypermetabolic or hyperdynamic states (e.g., hyperthyroidism, hypoxia, or arteriovenous shunt) are best treated by addressing the underlying condition. Atrial arrhythmias associated with hypermetabolic states are particularly resistant to digoxin treatment. Patients with beri beri heart disease may fail to respond adequately to digoxin if the underlying thiamine deficiency is not treated concomitantly.
Adverse Reactions
Clinical Trials Experience
Nervous System
- Visual disturbances (blurred or yellow vision), headache, weakness, dizziness, apathy, confusion, and mental disturbances (such as anxiety, depression, delirium, and hallucination).
Cardiovascular
- Arrhythmia( first-degree, second-degree (Wenckebach), or third-degree heart block (including asystole); atrial tachycardia with block; AV dissociation; accelerated junctional (nodal) rhythm; unifocal or multiform ventricular premature contractions (especially bigeminy or trigeminy);ventricular tachycardia; and ventricular fibrillation).
Gastrointestinal
Miscellaneous
- Gynecomastia, thrombocytopenia and maculopapular rash.
Infant and Children
The side effects of digoxin in infants and children differ from those seen in adults in several respects. Although digoxin may produce anorexia, nausea, vomiting, diarrhea, and CNS disturbances in young patients, these are rarely the initial symptoms of overdosage. Rather, the earliest and most frequent manifestation of excessive dosing with digoxin in infants and children is the appearance of cardiac arrhythmias, including sinus bradycardia. In children, the use of digoxin may produce any arrhythmia. The most common are conduction disturbances or supraventricular tachyarrhythmias, such as atrial tachycardia (with or without block) and junctional (nodal) tachycardia. Ventricular arrhythmias are less common. Sinus bradycardia may be a sign of impending digoxin intoxication, especially in infants, even in the absence of first-degree heart block. Any arrhythmia or alteration in cardiac conduction that develops in a child taking digoxin should be assumed to be caused by digoxin, until further evaluation proves otherwise.
Postmarketing Experience
There is limited information regarding Digoxin Postmarketing Experience in the drug label.
Drug Interactions
Drugs that Affect Renal Function
Caution should be exercised when combining digoxin with any drug that may cause significant deterioration in renal function (e.g., ACE inhibitors, angiotensin receptor blockers,nonsteroidal anti-inflammatory drugs [NSAIDs], COX-2 inhibitors) since a decline in glomerular filtration or tubular secretion may impair the excretion of digoxin.
Antiarrthymics
- Dofetilid : Concomitant administration with digoxin was associated with a higher rate of torsades de pointes
- Sotalol: Proarrhythmic events were more common in patients receiving sotalol and digoxin than on either alone; it is not clear whether this represents an interaction or is related to the presence of CHF, a known risk factor for proarrhythmia, in patients receiving digoxin.
- Dronedarone: Sudden death was more common in patients receiving digoxin with dronedarone than on either alone; it is not clear whether this represents an interaction or is related to the presence of advanced heart disease, a known risk factor for sudden death in patients receiving digoxin.
Parathyroid Hormone Analog
- Teriparatide: Sporadic case reports have suggested that hypercalcemia may predispose patients to digitalis toxicity. Teriparatide transiently increases serum calcium.
Potassium-depleting diuretics
Potassium-depleting diuretics are a major contributing factor to digitalis toxicity.
Calcium
Calcium, particularly if administered rapidly by the intravenous route, may produce serious arrhythmias in digitalized patients.
Quinidine, verapamil, amiodarone, propafenone, indomethacin, itraconazole, alprazolam, and spironolactone
Quinidine, verapamil, amiodarone, propafenone, indomethacin, itraconazole, alprazolam, and spironolactone raise the serum digoxin concentration due to a reduction in clearance and/or in volume of distribution of the drug, with the implication that digitalis intoxication may result.
Erythromycin, clarithromycin, and tetracycline
Erythromycin and clarithromycin (and possibly othermacrolide antibiotics) and tetracycline may increase digoxin absorption in patients who inactivate digoxin by bacterial metabolism in the lower intestine, so that digitalis intoxication may result (see Pharmacology).
Decrease gut motility, which may increase digoxin absorption.
Antacids, kaolin-pectin, sulfasalazine, neomycin, cholestyramine, certain anticancer drugs, and metoclopramide
May interfere with intestinal digoxin absorption, resulting in unexpectedly low serum concentrations.
May decrease serum digoxin concentration, especially in patients with renal dysfunction, by increasing the non-renal clearance of digoxin.
Thyroid administration to a digitalized, hypothyroid patient may increase the dose requirement of digoxin.
Sympathomimetics
Concomitant use of digoxin and sympathomimetics increases the risk of cardiac arrhythmias.
Succinylcholine
Succinylcholine may cause a sudden extrusion of potassium from muscle cells, and may thereby cause arrhythmias in digitalized patients.
Calcium Channel Blockers
Use with digoxin may be useful in combination to control atrial fibrillation, their additive effects on AV node conduction can result in advanced or complete heart block.
Beta blockers
Both digitalis glycosides and beta-blockers slow atrioventricular conduction and decrease heart rate. Concomitant use can increase the risk of bradycardia. Digoxin concentrations are increased by about 15% when digoxin and carvedilol are administered concomitantly. Therefore, increased monitoring of digoxin is recommended when initiating, adjusting, or discontinuing carvedilol.
Drug/Laboratory Test Interactions
- Endogenous substances of unknown composition (digoxin-like immunoreactive substances [DLIS]) can interfere with standard radioimmunoassays for digoxin. The interference most often causes results to be falsely positive or falsely elevated, but sometimes it causes results to be falsely reduced. Some assays are more subject to these failings than others. Several LC/MS/MS methods are available that may provide less susceptibility to DLIS interference.
- DLIS are present in up to half of all neonates and in varying percentages of pregnant women, patients with hypertrophic cardiomyopathy, patients with renal or hepatic dysfunction, and other patients who are volume-expanded for any reason. The measured levels of DLIS (as digoxin equivalents) are usually low (0.2-0.4 ng/mL), but sometimes they reach levels that would be considered therapeutic or even toxic.
- In some assays, spironolactone, canrenone, and potassium canrenoate may be falsely detected as digoxin, at levels up to 0.5 ng/mL. Some traditional Chinese and Ayurvedic medicine substances like Chan Su, Siberian Ginseng, Asian Ginseng, Ashwagandha, or Dashen can cause similar interference.
Spironolactone and DLIS are much more extensively protein-bound than digoxin. As a result, assays of free digoxin levels in protein-free ultrafiltrate (which tend to be about 25% less than total levels, consistent with the usual extent of protein binding) are less affected by spironolactone or DLIS.
- It should be noted that ultrafiltration does not solve all interference problems with alternative medicines. The use of an LC/MS/MS method may be the better option according to the good results it provides, especially in terms of specificity and limit of quantization.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C
Animal reproduction studies have not been conducted with digoxin. It is also not known whether digoxin can cause fetal harm when administered to a pregnant woman or can affect reproductive capacity. Digoxin should be given to a pregnant woman only if clearly needed.
Pregnancy Category (AUS): A
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Digoxin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Digoxin during labor and delivery.
Nursing Mothers
Studies have shown that digoxin concentrations in the mother’s serum and milk are similar. However, the estimated exposure of a nursing infant to digoxin via breastfeeding will be far below the usual infant maintenance dose. Therefore, this amount should have no pharmacologic effect upon the infant. Nevertheless, caution should be exercised when digoxin is administered to a nursing woman.
Pediatric Use
Newborn infants display considerable variability in their tolerance to digoxin. Premature and immature infants are particularly sensitive to the effects of digoxin, and the dosage of the drug must not only be reduced but must be individualized according to their degree of maturity. Digitalis glycosides can cause poisoning in children due to accidental ingestion.
Geriatic Use
The majority of clinical experience gained with digoxin has been in the elderly population. This experience has not identified differences in response or adverse effects between the elderly and younger patients. However, this drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, which should be based on renal function, and it may be useful to monitor renal function
Gender
In a study of 184 patients, the clearance of digoxin was 12% lower in female than in male patients. This difference is not likely to be clinically important.
Race
The impact of race differences on digoxin pharmacokinetics have not been formally studied. Because digoxin is primarily eliminated as unchanged drug via the kidney and because there are no important differences in creatinine clearance among races, pharmacokinetic differences due to race are not expected.)
Renal Impairment
Since the clearance of digoxin correlates withcreatinine clearance, patients with renal impairment generally demonstrate prolonged digoxin elimination half-lives and greater exposures to digoxin. Therefore, titrate carefully in these patients based on clinical response and based on monitoring of serum digoxin concentrations, as appropriate.
Hepatic Impairment
Because only a small percentage (approximately 13%) of a dose of digoxin undergoes metabolism, hepatic impairment would not be expected to significantly alter the pharmacokinetics of digoxin. In a small study, plasma digoxin concentration profiles in patients with acute hepatitis generally fell within the range of profiles in a group of healthy subjects. No dosage adjustments are recommended for patients with hepatic impairment; however, serum digoxin concentrations should be used, as appropriate, to help guide dosing in these patients.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Digoxin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Digoxin in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
- In selecting a LANOXIN dosing regimen, it is important to consider factors that affect digoxin blood levels (e.g., body weight, age, renal function, concomitant drugs) since toxic levels of digoxin are only slightly higher than therapeutic levels. Dosing can be either initiated with a loading dose followed by maintenance dosing if rapid titration is desired or initiated with maintenance dosing without a loading dose.
- Consider interruption or reduction in digoxin dose prior to electrical cardioversion.
Use digoxin solution to obtain the appropriate dose in infants, young pediatric patients, or patients with very low body weight.
- Intravenous:
- In selecting a digoxin dosing regimen, it is important to consider factors that affect digoxin blood levels (e.g., body weight, age, renal function, concomitant drugs) since toxic levels of digoxin are only slightly higher than therapeutic levels. Dosing can be either initiated with a loading dose followed by maintenance dosing if rapid titration is desired or initiated with maintenance dosing without a loading dose.
- Parenteral administration of digoxin should be used only when the need for rapid digitalization is urgent or when the drug cannot be taken orally. Intramuscular injection can lead to severe pain at the injection site, thus intravenous administration is preferred. If the drug must be administered by the intramuscular route, it should be injected deep into the muscle followed by massage. For adults, no more than 500 mcg of Digoxin Injection should be injected into a single site. For pediatric patients, see the full prescribing information for pediatric digoxin injection (not available from West-Ward) for specific recommendations.
- Administer the dose over a period of 5 minutes or longer and avoid bolus administration to prevent systemic and coronary vasoconstriction. Mixing of Digoxin Injection with other drugs in the same container or simultaneous administration in the same intravenous line is not recommended.
- Digoxin Injection can be administered undiluted or diluted with a 4-fold or greater volume of Sterile Water for Injection, 0.9% Sodium Chloride Injection, or 5% Dextrose Injection. The use of less than a 4-fold volume of diluent could lead to precipitation of the digoxin. Immediate use of the diluted product is recommended.
- If tuberculin syringes are used to measure very small doses do not flush with the parenteral solution after its contents are expelled into an indwelling vascular catheter to avoid over administration of digoxin.
- Consider interruption or reduction in digoxin dose prior to electrical cardioversion
Switching from Intravenous Digoxin to Oral Digoxin
When switching from intravenous to oral digoxin formulations, make allowances for differences in bioavailability when calculating maintenance dosages
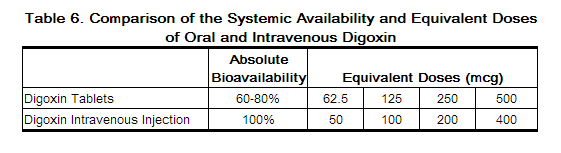
Monitoring
Monitor for signs and symptoms of digoxin toxicity and clinical response. Adjust dose based on toxicity, efficacy, and blood levels. Serum digoxin levels less than 0.5 ng/mL have been associated with diminished efficacy, while levels above 2 ng/mL have been associated with increased toxicity without increased benefit. Interpret the serum digoxin concentration in the overall clinical context, and do not use an isolated measurement of serum digoxin concentration as the basis for increasing or decreasing the digoxin dose. Serum digoxin concentrations may be falsely elevated by endogenous digoxin-like substances [see Drug Interactions (7.4)]. If the assay is sensitive to these substances, consider obtaining a baseline digoxin level before starting digoxin and correct post-treatment values by the reported baseline level. Obtain serum digoxin concentrations just before the next scheduled digoxin dose or at least 6 hours after the last dose. The digoxin concentration is likely to be 10-25% lower when sampled right before the next dose (24 hours after dosing) compared to sampling 8 hours after dosing (using once-daily dosing). However, there will be only minor differences in digoxin concentrations using twice daily dosing whether sampling is done at 8 or 12 hours after a dose.
IV Compatibility
Solution
Compatible
- 0.9% Sodium Chloride
- 5% Dextrose
Overdosage
Acute Overdose
Signs and Symptoms
- Adults: The most common signs and symptoms of digoxin toxicity are nausea, vomiting, anorexia, and fatigue that occur in 30 to 70% of patients who are overdosed. Extremely high serum concentrations produce hyperkalemia especially in patients with impaired renal function. Almost every type of cardiac arrhythmia has been associated with digoxin overdose and multiple rhythm disturbances in the same patient are common. Peak cardiac effects occur 3 to 6 hours following ingestion and may persist for 24 hours or longer. Arrhythmias that are considered more characteristic of digoxin toxicity are new-onset Mobitz type 1 A-V block, accelerated junctional rhythms, non-paroxysmal atrial tachycardia withA-V block, and bi-directional ventricular tachycardia. Cardiac arrest from asystole or ventricular fibrillation is usually fatal.
- Digoxin toxicity is related to serum concentration. As digoxin serum levels increase above 1.2 ng/mL, there is a potential for increase in adverse reactions. Furthermore, lower potassium levels increases the risk for adverse reactions. In adults with heart disease, clinical observations suggest that an overdose of digoxin of 10 to 15 mg results in death of half of patients. A dose above 25 mg ingested by an adult without heart disease appeared to be uniformly fatal if no Digoxin Immune Fab (DIGIBIND®, DIGIFAB®) was administered.
- Among the extra-cardiac manifestations, gastrointestinal symptoms (e.g., nausea, vomiting, anorexia) are very common (up to 80% incidence) and precede cardiac manifestations in approximately half of the patients in most literature reports. Neurologic manifestations (e.g., dizziness, various CNS disturbances), fatigue, and malaise are very common. Visual manifestations may also occur with aberration in color vision (predominance of yellow green) the most frequent. Neurological and visual symptoms may persist after other signs of toxicity have resolved. In chronic toxicity, nonspecific extra-cardiac symptoms, such as malaise and weakness, may predominate.
- Children: In pediatric patients, signs and symptoms of toxicity can occur during or shortly after the dose of digoxin. Frequent non-cardiac effects are similar to those observed in adults although nausea and vomiting are not seen frequently in infants and small pediatric patients. Other reported manifestations of overdose are weight loss in older age groups, failure to thrive in infants, abdominal pain caused by mesenteric artery ischemia, drowsiness, and behavioral disturbances including psychotic episodes. Arrhythmias and combinations of arrhythmias that occur in adult patients can also occur in pediatric patients although sinus tachycardia, supraventricular tachycardia, and rapid atrial fibrillation are seen less frequently in pediatric patients. Pediatric patients are more likely to develop A-V conduction disturbances, or sinus bradycardia. Any arrhythmia in a child treated with digoxin should be considered related to digoxin until otherwise ruled out. In pediatric patients aged 1 to 3 years without heart disease, clinical observations suggest that an overdose of digoxin of 6 to 10 mg would result in death of half of the patients. In the same population, a dose above 10 mg resulted in death if no Digoxin Immune Fab were administered.
Management
- Patients who have intentionally or accidently ingested massive doses of digoxin should receive activated charcoal orally or by nasogastric tube regardless of the time since ingestion since digoxin recirculates to the intestine by enterohepatic circulation. In addition to cardiac monitoring, temporarily discontinue digoxin until the adverse reaction resolves. Correct factors that may be contributing to the adverse reactions [see Warnings and Precautions].
- In particular, correct hypokalemia and hypomagnesemia.
- Digoxin is not effectively removed from the body by dialysisbecause of its large extravascular volume of distribution. Life threatening arrhythmias (ventricular tachycardia, ventricular fibrillation, high degree A-V block, bradyarrhythma, sinus arrest) or hyperkalemia requires administration of Digoxin Immune Fab.
- Digoxin Immune Fab has been shown to be 80 to 90% effective in reversing signs and symptoms of digoxin toxicity. Bradycardia and heart block caused by digoxin are parasympathetically mediated and respond to atropine.
- A temporary cardiac pacemaker may also be used. Ventricular arrhythmias may respond to lidocaine or phenytoin. When a large amount of digoxin has been ingested, especially in patients with impaired renal function, hyperkalemiamay be present due to release of potassium from skeletal muscle. In this case, treatment with Digoxin Immune Fab is indicated; an initial treatment with glucose and insulin may be needed if the hyperkalemia is life-threatening.
- Once the adverse reaction has resolved, therapy with digoxin may be reinstituted following a careful reassessment of dose.
Chronic Overdose
Signs and Symptoms
The most common signs and symptoms of digoxin toxicity are nausea, vomiting, anorexia, and fatigue that occur in 30 to 70% of patients who are overdosed.
Management
- If there is suspicion of toxicity, discontinue digoxin and place the patient on a cardiac monitor. Correct factors such as electrolyte abnormalities, thyroid dysfunction, and concomitant medications [see Dosage and Administration].
- Correct hypokalemia by administering potassium so that serum potassium is maintained between 4.0 and 5.5 mmol/L. Potassium is usually administered orally, but when correction of the arrhythmia is urgent and serum potassium concentration is low, potassium may be administered by the intravenous route.
- Monitor electrocardiogram for any evidence of potassium toxicity (e.g., peaking of T waves) and to observe the effect on the arrhythmia. Avoid potassium salts in patients with bradycardia or heart block. Symptomatic arrhythmias may be treated with Digoxin Immune Fab.
Pharmacology
Mechanism of Action
All of digoxin’s actions are mediated through its effects on Na-K ATPase. This enzyme, the “sodium pump,” is responsible for maintaining the intracellular milieu throughout the body by moving sodium ions out of and potassium ions into cells. By inhibiting Na-K ATPase, digoxin
- causes increased availability of intracellular calcium in the myocardium and conduction system, with consequent increased inotropy, increased automaticity, and reduced conduction velocity
- indirectly causes parasympathetic stimulation of the autonomic nervous system, with consequent effects on the sino-atrial (SA) and atrioventricular (AV) nodes
- reduces catecholamine reuptake at nerve terminals, rendering blood vessels more sensitive to endogenous or exogenous catecholamines
- increases baroreceptor sensitization, with consequent increased carotid sinus nerve activity and enhanced sympathetic withdrawal for any given increment in mean arterial pressure
- increases (at higher concentrations) sympathetic outflow from the central nervous system (CNS) to both cardiac and peripheral sympathetic nerves
allows (at higher concentrations) progressive efflux of intracellular potassium, with consequent increase in serum potassium levels.
The cardiologic consequences of these direct and indirect effects are an increase in the force and velocity of myocardial systolic contraction (positive inotropic action), a slowing of the heart rate (negative chronotropic effect), decreased conduction velocity through the AV node, and a decrease in the degree of activation of the sympathetic nervous system and renin-angiotensin system (neurohormonal deactivating effect).
Structure
Digoxin is one of the cardiac (or digitalis) glycosides, a closely related group of drugs having in common specific effects on the myocardium. These drugs are found in a number of plants. Digoxin is extracted from the leaves of Digitalis lanata. The term “digitalis” is used to designate the whole group of glycosides. The glycosides are composed of 2 portions: a sugar and a cardenolide (hence “glycosides”).
Digoxin is described chemically as (3β,5β,12β)-3-[(O-2,6-dideoxy-β-D-ribo-hexopyranosyl-(1→4)-O-2,6-dideoxy-β-D-ribo-hexopyranosyl-(1→4)-2,6-dideoxy-β-D-ribo-hexopyranosyl)oxy]-12,14-dihydroxy-card-20(22)-enolide. Its molecular formula is C41H64O14, its molecular weight is 780.95.
Digoxin exists as clear to white odorless crystals or white, odorless crystalline powder that melts with decomposition above 230°C. The drug is practically insoluble in water and in ether; slightly soluble in diluted (50%) alcohol and in chloroform; and freely soluble in pyridine.
Digoxin injection USP is a sterile solution of digoxin for intravenous or intramuscular injection. Each mL contains: digoxin 0.25 mg, propylene glycol 40% (v/v), anhydrous ethanol 10% (v/v), dibasic sodium phosphate 0.3% (w/v) and anhydrous citric acid 0.08% (w/v) to adjust pH between 6.8 and 7.2, and water for injection. Dilution is not required.
Pharmacodynamics
The times to onset of pharmacologic effect and to peak effect of preparations of digoxin are shown in the table below.

Hemodynamic Effects: Short- and long-term therapy with the drug increases cardiac output and lowers pulmonary artery pressure, pulmonary capillary wedge pressure, and systemic vascular resistance in patients with heart failure. These hemodynamic effects are accompanied by an increase in the left ventricular ejection fraction and a decrease in end-systolic and end-diastolic dimensions.
ECG Changes: The use of therapeutic doses of digoxin may cause prolongation of thePR interval and depression of theST segment on the electrocardiogram. Digoxin may produce false positive ST-T changes on the electrocardiogram during exercise testing. These electrophysiologic effects are not indicative of toxicity. Digoxin does not significantly reduce heart rate during exercise.
Pharmacokinetics
Distribution: Following drug administration, a 6 to 8 hour tissue distribution phase is observed. This is followed by a much more gradual decline in the serum concentration of the drug, which is dependent on the elimination of digoxin from the body. The peak height and slope of the early portion (absorption/distribution phases) of the serum concentration-time curve are dependent upon the route of administration and the absorption characteristics of the formulation. Clinical evidence indicates that the early high serum concentrations do not reflect the concentration of digoxin at its site of action, but that with chronic use, the steady-state post-distribution serum concentrations are in equilibrium with tissue concentrations and correlate with pharmacologic effects. In individual patients, these post-distribution serum concentrations may be useful in evaluating therapeutic and toxic effects.
Digoxin is concentrated in tissues and therefore has a large apparent volume of distribution (approximately 475 to 500 L). Digoxin crosses both the blood-brain barrier and the placenta. At delivery, the serum digoxin concentration in the newborn is similar to the serum concentration in the mother. Approximately 25% of digoxin in the plasma is bound to protein. Serum digoxin concentrations are not significantly altered by large changes in fat tissue weight, so that its distribution space correlates best with lean (i.e., ideal) body weight, not total body weight.
Metabolism: Only a small percentage (13%) of a dose of digoxin is metabolized in healthy volunteers. The urinary metabolites, which include dihydrodigoxin, digoxigenin bisdigitoxoside, and their glucuronide and sulfate conjugates are polar in nature and are postulated to be formed via hydrolysis, oxidation, and conjugation. The metabolism of digoxin is not dependent upon the cytochrome P-450 system, and digoxin is not known to induce or inhibit thecytochrome P-450 system.
Excretion: Elimination of digoxin follows first-order kinetics (that is, the quantity of digoxin eliminated at any time is proportional to the total body content). Following intravenous administration to healthy volunteers, 50 to 70% of a digoxin dose is excreted unchanged in the urine. Renal excretion of digoxin is proportional to creatinine clearance and is largely independent of urine flow. In healthy volunteers with normal renal function, digoxin has a half-life of 1.5 to 2 days. The half-life in anuric patients is prolonged to 3.5 to 5 days. Digoxin is not effectively removed from the body by dialysis, exchange transfusion, or during cardiopulmonary bypass because most of the drug is bound to extravascular tissues.
Nonclinical Toxicology
Digoxin showed no genotoxic potential in in vitro studies (Ames test and mouse lymphoma). No data are available on the carcinogenic potential of digoxin, nor have studies been conducted to assess its potential to affect fertility.
Clinical Studies
Chronic Heart Failure
Two 12-week, double-blind, placebo-controlled studies enrolled 178 (RADIANCE trial) and 88 (PROVED trial) patients with NYHA class II or III heart failure previously treated with digoxin, a diuretic, and an ACE inhibitor (RADIANCE only) and randomized them to placebo or treatment with LANOXIN. Both trials demonstrated better preservation of exercise capacity in patients randomized to LANOXIN. Continued treatment with LANOXIN reduced the risk of developing worsening heart failure, as evidenced by heart failure-related hospitalizations and emergency care and the need for concomitant heart failure therapy.
Dig Trial of LANOXIN in Patients with Heart Failure
The Digitalis Investigation Group (DIG) main trial was a 37-week, multicenter, randomized, double-blind mortality study comparing digoxin to placebo in 6800 adult patients with heart failure and left ventricular ejection fraction ≤0.45. At randomization, 67% were NYHA class I or II, 71% had heart failure of ischemic etiology, 44% had been receiving digoxin, and most were receiving a concomitant ACE inhibitor (94%) and diuretics (82%). As in the smaller trials described above, patients who had been receiving open-label digoxin were withdrawn from this treatment before randomization. Randomization to digoxin was again associated with a significant reduction in the incidence of hospitalization, whether scored as number of hospitalizations for heart failure (relative risk 75%), risk of having at least one such hospitalization during the trial (RR 72%), or number of hospitalizations for any cause (RR 94%). On the other hand, randomization to digoxin had no apparent effect on mortality (RR 99%, with confidence limits of 91 to 107%).
Chronic Atrial Fibrillation
Digoxin has also been studied as a means of controlling the ventricular response to chronic atrial fibrillation in adults. Digoxin reduced the resting heart rate, but not the heart rate during exercise.
In 3 different randomized, double-blind trials that included a total of 315 adult patients, digoxin was compared to placebo for the conversion of recent-onset atrial fibrillation to sinus rhythm. Conversion was equally likely, and equally rapid, in the digoxin and placebo groups. In a randomized 120-patient trial comparing digoxin, sotalol, and amiodarone, patients randomized to digoxin had the lowest incidence of conversion to sinus rhythm, and the least satisfactory rate control when conversion did not occur.
In at least one study, digoxin was studied as a means of delaying reversion to atrial fibrillation in adult patients with frequent recurrence of this arrhythmia. This was a randomized, double-blind, 43-patient crossover study. Digoxin increased the mean time between symptomatic recurrent episodes by 54%, but had no effect on the frequency of fibrillatory episodes seen during continuous electrocardiographic monitoring.
How Supplied
DIgoxin Tablets
LANOXIN (digoxin) Tablets, Scored 125 mcg (0.125 mg): Bottles of 100 with child-resistant cap (NDC 24987-242-55), (NDC 24987-242-57) and 1,000 (NDC 24987-242-75), (NDC 24987-242-76); unit dose pack of 100 (NDC 24987-242-56). Imprinted with LANOXIN and Y3B (yellow).
LANOXIN (digoxin) Tablets, Scored 250 mcg (0.25 mg): Bottles of 100 with child-resistant cap (NDC 24987-249-55), (NDC 24987-249-57), 1,000 (NDC -24987-249-75), (NDC-24987-249-76), and 5,000 (NDC 24987-249-80); unit dose pack of 100 (NDC 24987-249-56). Imprinted with LANOXIN and X3A (white).
- National Drug Code (NDC): see above
- Manufactured by: Manufactured by DSM Pharmaceuticals, Inc. Greenville, NC 27834
- Distributed by:Covis Pharmaceuticals, Inc. Cary, NC 27511
Digoxin Injection
Digoxin Injection, USP is available as: 500 mcg/2 mL (250 mcg/mL) ampuls packaged in 25s
- National Drug Code (NDC):(NDC 0641-1410-35)
- Manufactured by: HIKMA FARMACÊUTICA (PORTUGAL), S.A. Estrada do Rio da Mό, 8, 8A e 8B – Fervença – 2705-906 Terrugem SNT, PORTUGAL
- Distributed by:WEST-WARD PHARMACEUTICALS Eatontown, NJ 07724 USA
Digoxin Solution
Digoxin Solution:Each 1 mL of clear, colorless Digoxin Oral Solution contains 0.05 mg (50 mcg). The Digoxin Oral Solution bottles are to be used with the graduated droppers provided in the carton. Starting at 0.2 mL, this 1 mL dropper is marked in divisions of 0.1 mL, corresponding to 5 mcg or 0.005 mg of digoxin. The calibrated dropper supplied with the 60 mL bottle of Digoxin Oral Solution is not appropriate to measure doses below 0.2 mL. Doses less than 0.2 mL require appropriate methods or measuring devices designed to administer an accurate amount to the patient, such as a graduated syringe
- National Drug Code (NDC):NDC:17856-0057
- Manufactured by:Roxane Laboratories, Inc. Columbus, Ohio 43216
- Distributed by:Atlantic Biologicals Corps
Storage
DIgoxin Tablets
- Storage: Store at 25°C (77°F); excursions permitted to 15 to 30°C (59 to 86°F) in a dry place.
- Keep out of reach of children.
Digoxin Injection
- Storage: Store at 20˚-25˚C (68˚-77˚F), excursions permitted to 15˚-30˚C (59˚-86˚F).
Digoxin Solution
- Storage:Store at 25°C (77°F); excursions permitted to 15° to 30°C (59° to 86°F). * Protect from light.
Images
Drug Images
{{#ask: Page Name::Digoxin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Digoxin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Advise patients that digoxin is a cardiac glycoside used to treat heart failure and heart arrhythmias.
- Instruct patients to take this medication as directed by their physician.
- Advise patients that many drugs can interact with digoxin. Instruct patients to inform their doctor and pharmacist if they are taking any over the counter medications, including herbal *medication, or are started on a new prescription.
- Advise patient that blood tests will be necessary to ensure that their digoxin dose is appropriate for them.
- Advise patients to contact their doctor or a health care professional if they experience nausea, vomiting, persistent diarrhea, confusion, weakness, or visual disturbances (including *blurred vision, green-yellow color disturbances, halo effect) as these could be signs that the dose of digoxin may be too high.
- Advise parents or caregivers that the symptoms of having too high digoxin doses may be difficult to recognize in infants and pediatric patients. Symptoms such as weight loss, failure to thrive in infants, abdominal pain, and behavioral disturbances may be indications of digoxin toxicity.
- Suggest to the patient to monitor and record their heart rate and blood pressure daily.
- Instruct women of childbearing potential who become or are planning to become pregnant to consult a physician prior to initiation or continuing therapy with digoxin.
Precautions with Alcohol
Alcohol-SandboxAlonso interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Digoxin
- Digox
- Lanoxin
Look-Alike Drug Names
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Pfammatter, JP.; Stocker, FP. (1998). "Re-entrant supraventricular tachycardia in infancy: current role of prophylactic digoxin treatment". Eur J Pediatr. 157 (2): 101–6. PMID 9504781. Unknown parameter
|month=ignored (help) - ↑ Sanatani, S.; Potts, JE.; Reed, JH.; Saul, JP.; Stephenson, EA.; Gibbs, KA.; Anderson, CC.; Mackie, AS.; Ro, PS. (2012). "The study of antiarrhythmic medications in infancy (SAMIS): a multicenter, randomized controlled trial comparing the efficacy and safety of digoxin versus propranolol for prophylaxis of supraventricular tachycardia in infants". Circ Arrhythm Electrophysiol. 5 (5): 984–91. doi:10.1161/CIRCEP.112.972620. PMID 22962431. Unknown parameter
|month=ignored (help) - ↑ Lilja, H.; Karlsson, K.; Lindecrantz, K.; Sabel, KG. (1984). "Treatment of intrauterine supraventricular tachycardia with digoxin and verapamil". J Perinat Med. 12 (3): 151–4. PMID 6502442.
{{#subobject:
|Label Page=Digoxin |Label Name=Lanoxintablet1.jpg
}}
{{#subobject:
|Label Page=Digoxin |Label Name=Lanoxintablet2.jpg
}}
{{#subobject:
|Label Page=Digoxin |Label Name=Lanoxintablet3.png
}}
{{#subobject:
|Label Page=Digoxin |Label Name=Digoxininjection.jpg
}}
{{#subobject:
|Label Page=Digoxin |Label Name=Digoxininjection1.png
}}
{{#subobject:
|Label Page=Digoxin |Label Name=Digoxinsolution.jpg
}}
{{#subobject:
|Label Page=Digoxin |Label Name=Digoxinsolution1.png
}}