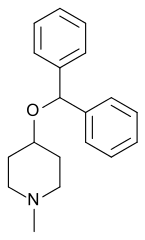
Diphenylpyraline
 | |
| Clinical data | |
|---|---|
| Synonyms | 4-(diphenylmethoxy)-1-methyl-piperidine |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, Topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Elimination half-life | 24-40 hours[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C19H23NO |
| Molar mass | 281.392 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
|
WikiDoc Resources for Diphenylpyraline |
|
Articles |
|---|
|
Most recent articles on Diphenylpyraline Most cited articles on Diphenylpyraline |
|
Media |
|
Powerpoint slides on Diphenylpyraline |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Diphenylpyraline at Clinical Trials.gov Trial results on Diphenylpyraline Clinical Trials on Diphenylpyraline at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Diphenylpyraline NICE Guidance on Diphenylpyraline
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Diphenylpyraline Discussion groups on Diphenylpyraline Patient Handouts on Diphenylpyraline Directions to Hospitals Treating Diphenylpyraline Risk calculators and risk factors for Diphenylpyraline
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Diphenylpyraline |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Diphenylpyraline (DPP; sold as Allergen, Arbid, Belfene, Diafen, Hispril, Histyn, Lergobine, Lyssipol, Mepiben, Neargal) is a first-generation antihistamine with anticholinergic effects of the diphenylpiperidine class.[2][3][4] It is marketed in Europe for the treatment of allergies.[2][3][5] DPP has also been found to act as a dopamine reuptake inhibitor and produces hyperactivity in rodents.[6] It has been shown to be useful in the treatment of Parkinsonism.[7]
Chemistry
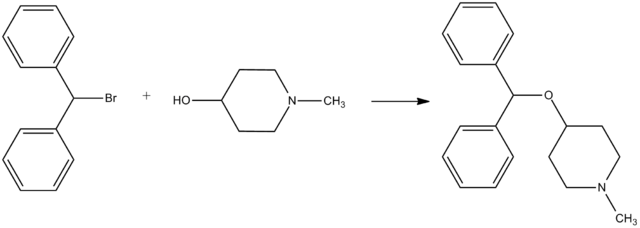
Diphenylpyraline, 4-diphenylmethoxy-1-methylpiperidine, is synthesized by alkylating 4-hydroxy-1-methylpiperidine with benzhydrylbromide.
See also
References
- ↑ Graham G, Bolt AG (June 1974). "Half-life of diphenylpyraline in man". Journal of Pharmacokinetics and Biopharmaceutics. 2 (3): 191–5. doi:10.1007/BF01059761. PMID 4156058.
- ↑ 2.0 2.1 Swiss Pharmaceutical Society (2000). Index Nominum 2000: International Drug Directory (Book with CD-ROM). Boca Raton: Medpharm Scientific Publishers. ISBN 3-88763-075-0.
- ↑ 3.0 3.1 Puhakka H, Rantanen T, Virolainen E (1977). "Diphenylpyraline (Lergobine) in the treatment of patients suffering from allergic and vasomotor rhinitis". J Int Med Res. 5 (1): 37–41. PMID 14039.
- ↑ Kubo N, Shirakawa O, Kuno T, Tanaka C (March 1987). "Antimuscarinic effects of antihistamines: quantitative evaluation by receptor-binding assay". Japanese Journal of Pharmacology. 43 (3): 277–82. doi:10.1254/jjp.43.277. PMID 2884340.
- ↑ Hruby, Victor J.; Ruben Vardanyan; Vardanyan, ۊRuben (2006). Synthesis of essential drugs. Amsterdam: Elsevier. ISBN 0-444-52166-6.
- ↑ Lapa G, Mathews T, Harp J, Budygin E, Jones S (2005). "Diphenylpyraline, a histamine H1 receptor antagonist, has psychostimulant properties". Eur J Pharmacol. 506 (3): 237–40. doi:10.1016/j.ejphar.2004.11.017. PMID 15627433.
- ↑ Ohno T, Kobayashi S, Hayashi M, Sakurai M, Kanazawa I (2001). "Diphenylpyraline-responsive parkinsonism in cerebrotendinous xanthomatosis: long-term follow up of three patients". J Neurol Sci. 182 (2): 95–7. doi:10.1016/S0022-510X(00)00441-X. PMID 11137513.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- Template:drugs.com link with non-standard subpage
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- H1 receptor antagonists
- Piperidines
- Ethers
- Stimulants
- Drug