Fexofenadine hydrochloride
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Fexofenadine hydrochloride is an antihistamine that is FDA approved for the treatment of runny nose,itchy watery eyes, sneezing, itching of the nose or throat. Common adverse reactions include stomach ache, backache, dizziness , headache , somnolence,fatigue, pain in limb.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
- Runny nose
- Itchy, watery eyes
- Sneezing
- Itching of the nose or throat
Dosing
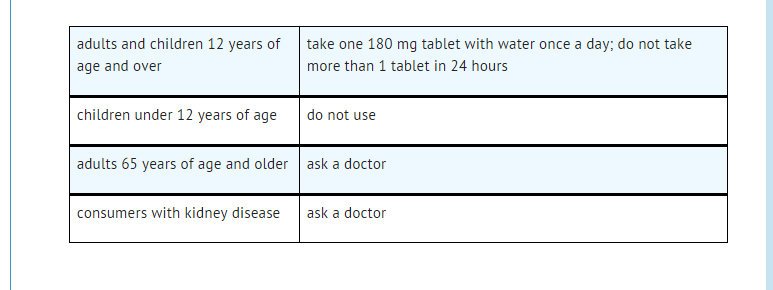
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Fexofenadine in adult patients.
Non–Guideline-Supported Use
Indications
- Hymenoptera immunotherapy, pretreatment[1]
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Indications
Temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
Dosing

Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Fexofenadine in pediatric patients.
Non–Guideline-Supported Use
Indications
- Hymenoptera immunotherapy, Pretreatment[1]
- Perennial allergic rhinitis[2]
Contraindications
There is limited information regarding Fexofenadine hydrochloride Contraindications in the drug label.
Warnings
Do not use
- If you have ever had an allergic reaction to this product or any of its ingredients.
Ask a doctor before use if you have
- Kidney disease. Your doctor should determine if you need a different dose.
When using this product
- Do not take more than directed
- Do not take at the same time as aluminum or magnesium antacids
- Do not take with fruit juices (see Directions)
Stop use and ask a doctor if
- An allergic reaction to this product occurs. Seek medical help right away.
If pregnant or breast-feeding
Ask a health professional before use.
Keep out of reach of children
- In case of overdose, get medical help or contact a Poison Control Center right away. (1-800-222-1222)
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Clinical Trial Experience of Fexofenadine
Postmarketing Experience
Common
- Gastrointestinal: stomach ache
- Musculoskeletal: backache
- Neurologic: dizziness , headache , somnolence
- Other: fatigue, pain in limb
Drug Interactions
There is limited information regarding Fexofenadine hydrochloride Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Fexofenadine hydrochloride in women who are pregnant.
Pregnancy Category (AUS):
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of fexofenadine hydrochloride in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of fexofenadine hydrochloride during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of fexofenadine hydrochloride with respect to nursing mothers.
Pediatric Use
There is no FDA guidance on the use of fexofenadine hydrochloride with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of fexofenadine hydrochloridewith respect to geriatric patients.
Gender
There is no FDA guidance on the use of fexofenadine hydrochloride with respect to specific gender populations.
Race
There is no FDA guidance on the use of fexofenadine hydrochloride with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of fexofenadine hydrochloride in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of fexofenadine hydrochloride in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of fexofenadine hydrochloride in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance on the use of fexofenadine hydrochloride in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
There is limited information regarding Monitoring of fexofenadine hydrochloride in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of fexofenadine hydrochloride in the drug label.
Overdosage
There is limited information regarding Chronic Overdose of fexofenadine hydrochloride in the drug label.
Pharmacology
There is limited information regarding Fexofenadine hydrochloride Pharmacology in the drug label.
Mechanism of Action
There is limited information regarding mechanism of action: of fexofenadine hydrochloride in the drug label.
Structure
There is limited information regarding structure: : of fexofenadine hydrochloride in the drug label.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of fexofenadine hydrochloride in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of fexofenadine hydrochloride in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of fexofenadine hydrochloride in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of fexofenadine hydrochloride in the drug label.
How Supplied
There is limited information regarding Fexofenadine hydrochloride How Supplied in the drug label.
Storage
There is limited information regarding Fexofenadine hydrochloride Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Fexofenadine hydrochloride |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Fexofenadine hydrochloride |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of fexofenadine hydrochloride in the drug label.
Precautions with Alcohol
- Alcohol- fexofenadine hydrochloride interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
7 SELECT ALLERGY RELIEF
Look-Alike Drug Names
There is limited information regarding Fexofenadine hydrochloride Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ 1.0 1.1 Reimers A, Hari Y, Müller U (2000). "Reduction of side-effects from ultrarush immunotherapy with honeybee venom by pretreatment with fexofenadine: a double-blind, placebo-controlled trial". Allergy. 55 (5): 484–8. PMID 10843430 PMID: 10843430 Check
|pmid=value (help). - ↑ 2.0 2.1 Bunnag C, Jareoncharsri P, Tunsuriyawong P, Pumhirun P, Limprasertsiri S, Chochaipanichnon L; et al. (2000). "A non-comparative trial of the efficacy and safety of fexofenadine for treatment of perennial allergic rhinitis". Asian Pac J Allergy Immunol. 18 (3): 127–33. PMID 11270466 PMID: 11270466 Check
|pmid=value (help).
{{#subobject:
|Page Name=Fexofenadine hydrochloride
|Pill Name=
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Fexofenadine hydrochloride |Label Name=Fexofenadine ingredients and appearance.png
}}
{{#subobject:
|Label Page=Fexofenadine hydrochloride |Label Name=Fexofenadine fig01.jpg
}}