Atosiban
 | |
| Clinical data | |
|---|---|
| Trade names | Tractocile, Antocin |
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | intravenous |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
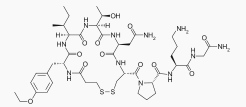
| Formula | C43H67N11O12S2 |
| Molar mass | 994.199 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Atosiban |
|
Articles |
|---|
|
Most recent articles on Atosiban |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Atosiban at Clinical Trials.gov Clinical Trials on Atosiban at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Atosiban
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Atosiban Risk calculators and risk factors for Atosiban
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Atosiban |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Atosiban (trade names Tractocile, Antocin, atosiban SUN) is an inhibitor of the hormones oxytocin and vasopressin. It is used as an intravenous medication as a labour repressant (tocolytic) to halt premature labor. Although initial studies suggested it could be used as a nasal spray and hence would not require hospital admission, it is not used in that form. It was developed by Ferring Pharmaceuticals in Sweden and first reported in the literature in 1985.[1] Originally marketed by Ferring Pharmaceuticals, it is licensed in proprietary and generic forms for the delay of imminent pre-term birth in pregnant adult women.
Mechanism of action
Atosiban is a nonapeptide, desamino-oxytocin analogue, and a competitive vasopressin/oxytocin receptor antagonist (VOTra). Atosiban inhibits the oxytocin-mediated release of inositol trisphosphate from the myometrial cell membrane. As a result, there is reduced release of intracellular, stored calcium from the sarcoplasmic reticulum of myometrial cells, and reduced influx of Ca2+ from the extracellular space through voltage gated channels. In addition, atosiban suppresses oxytocin-mediated release of PGE and PGF from the decidua.[2]
In human pre-term labour, atosiban, at the recommended dosage, antagonises uterine contractions and induces uterine quiescence. The onset of uterus relaxation following atosiban is rapid, uterine contractions being significantly reduced within 10 minutes to achieve stable uterine quiescence.
Indications
Atosiban is indicated to delay imminent pre-term birth in pregnant adult women with:[3]
- regular uterine contractions of at least 30 seconds duration at a rate of ≥ 4 per 30 minutes
- a cervical dilation of 1 to 3 cm (0-3 for nulliparas) and effacement of ≥ 50%
- a gestational age from 24 until 33 completed weeks
- a normal foetal heart rate
Other uses
Atosiban use after assisted reproduction
Atosiban is useful in improving the pregnancy outcome of in vitro fertilization-embryo transfer (IVF-ET) in patients with repeated implantation failure (RIF).[4] The pregnancy rate improved from zero to 43.7%.[5]
It was seen that the first- and second-trimester bleeding was more prevalent in ART than in spontaneous pregnancies.From 2004 to 2010, 33 first-trimester pregnancies with vaginal bleeding after ART with evident uterine contractions, author used atosiban and/or ritodrine, and there was no preterm delivery before 30 weeks.[6]
In a recent meta-analysis,[7] nifedipine is superior to β2-adrenergic-receptor agonists and magnesium sulfate for tocolysis in women with preterm labor (20–36 weeks), but it has been assigned to pregnancy category C by the Food and Drug Administration (FDA) so is not recommended before 20 weeks, or in the first trimester.[6] Recent reports supports the use of atosiban, even at very early pregnancy, to decrease the frequency of uterine contractions to enhance success of pregnancy.[4]
Clinical trials
Atosiban vs. nifedipine
Recently published a retrospective study (Saleh SS et al. 2013) comparing the efficacy and safety of atosiban and nifedipine in the suppression of pre-term labour concluded that atosiban and nifedipine are effective in delaying delivery for 7 days or more in women presenting with pre-term labour. A total of 68.3% of women in the atosiban group remained undelivered at 7 days or more, compared with 64.7% in the nifedipine group. They have the same efficacy and associated minor side-effects. However, flushing, palpitation and hypotension were significantly higher in the nifedipine group.[8]
A clinical trial (Salim R et al. 2012) compared tocolytic efficacy and tolerability of atosiban with that of nifedipine. Forty-eight (68.6%) women allocated to atosiban and 39 (52%) to nifedipine did not deliver and did not require an alternate agent at 48 hours respectively (P=.03). Study concludes that atosiban has fewer failures within 48 hours. Nifedipine may be associated with a longer postponement of delivery.[9]
Randomised controlled study (de Heus R et al. 2009), demonstrates for the first time, the direct effects of atosiban on fetal movement, heart rate and blood flow. Tocolysis with either atosiban or nifedipine combined with betamethasone administration have no direct fetal adverse effects.[10]
Atosiban vs. ritodrine
Multicentre, controlled trial of atosiban Vs. ritodrine in 128 women shows a significantly better tocolytic efficacy after 7 days in the atosiban group than in the ritodrine group (60.3 versus 34.9%), but not at 48 hours (68.3 versus 58.7%). Maternal adverse events were reported less frequently in the atosiban group (7.9 vs 70.8%), resulting in fewer early drug terminations due to adverse events (0 versus 20.0%). Therefore atosiban is superior to ritodrine in the treatment of preterm labour.[11]
Systematic Review
A systematic review of atosiban for tocolysis in preterm labour, six clinical studies - two compared atosiban to placebo and four atosiban to a beta-agonist showed a significant increase in the proportion of women undelivered by 48 hours in women receiving atosiban compared to placebo. When compared with beta-agonists, atosiban increased the proportion of women undelivered by 48 hours and was more safer compared to beta-agonists. Therefore oxytocin antagonists appear to be effective and safe for tocolysis in preterm labour.[12]
A 2005 systematic review by the Cochrane Collaboration showed that while atosiban had fewer side-effects than alternative drugs (such as ritodrine,), other beta blockers, calcium channel antagonists. It was no better than placebo in the major outcomes.[13]
See also
References
- ↑ Akerlund M, Carlsson AM, Melin P, Trojnar J (1985). "The effect on the human uterus of two newly developed competitive inhibitors of oxytocin and vasopressin". Acta Obstet Gynecol Scand. 64 (6): 499–504. doi:10.3109/00016348509156728. PMID 4061066.
- ↑ Sanu O, Lamont RF (2010). "Critical appraisal and clinical utility of atosiban in the management of preterm labor". Ther Clin Risk Manag. 6: 191–199. PMID 20463780.
- ↑ Tractocile-Summary of Product Characteristics. http://www.medicines.org.uk/emc/medicine/4305/SPC/
- ↑ 4.0 4.1 Chou PY, Wu MH, Pan HA, Hung KH, Chang FM. Use of an oxytocin antagonist in in vitro fertilization-embryo transfer for women with repeated implantation failure: a retrospective study.Taiwan J Obstet Gynecol. 2011 Jun;50(2):136-40. doi: 10.1016/j.tjog.2011.04.003
- ↑ Lan VT, Khang VN, Nhu GH, Tuong HM. Atosiban improves implantation and pregnancy rates in patients with repeated implantation failure.Lan VT, Khang VN, Nhu GH, Tuong HM. Reprod Biomed Online. 2012 Sep;25(3):254-60. doi: 10.1016/j.rbmo.2012.05.014. Epub 2012 Jun 16.
- ↑ 6.0 6.1 Wu MY, Chen SU, Yang YS. Using atosiban in uterine contractions of early pregnancies after assisted reproduction.J Formos Med Assoc. 2011 Dec;110(12):800. doi: 10.1016/j.jfma.2011.11.016. Epub 2011 Dec 20.
- ↑ Conde-Agudelo A, Romero R, Kusanovic JP. Nifedipine in the management of preterm labor: a systematic review and metaanalysis. Am J Obstet Gynecol. 2011;204:134.e1–134.e20
- ↑ Saleh SS, Al-Ramahi MQ, Al Kazaleh FA. Atosiban and nifedipine in the suppression of pre-term labour: a comparative study. J Obstet Gynaecol. 2013 Jan;33(1):43-5. doi: 10.3109/01443615.2012.721822.
- ↑ Salim R, Garmi G, Nachum Z, Zafran N, Baram S, Shalev E. Nifedipine compared with atosiban for treating preterm labor: a randomized controlled trial. Obstet Gynecol. 2012 Dec;120(6):1323-31. doi: http://10.1097/AOG.0b013e3182755dff.
- ↑ de Heus R, Mulder EJ,Derks JB, Visser GH. The effects of thetocolytics atosiban and nifedipine on fetal movements, heart rate and bloodflow. J Matern Fetal Neonatal Med. 2009 Jun;22(6):485-90. doi:10.1080/14767050802702349.
- ↑ Shim JY, Park YW, YoonBH, Cho YK, Yang JH, Lee Y, Kim A.Multicentre, parallelgroup, randomised, single-blind study of the safety and efficacy of atosibanversus ritodrine in the treatment of acute preterm labour in Korean women.BJOG. 2006Nov;113(11):1228-34. Epub 2006 Sep 15.
- ↑ Coomarasamy A, Knox EM, Gee H, Khan KS. Oxytocin antagonists for tocolysis in preterm labour -- a systematic review. Med Sci Monit. 2002 Nov;8(11):RA268-73.
- ↑ Papatsonis D, Flenady V, Cole S, Liley H (2005). Papatsonis, Dimitri, ed. "Oxytocin receptor antagonists for inhibiting preterm labour". Cochrane database of systematic reviews (Online) (3): CD004452. doi:10.1002/14651858.CD004452.pub2. PMID 16034931.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- Template:drugs.com link with non-standard subpage
- Articles with changed CASNo identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical pages without DrugBank identifier
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drug
- Tocolytics
- Peptides
- Vasopressin receptor antagonists
- Oxytocin receptor antagonists