Insulin glulisine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Insulin glulisine is a human insulin analog that is FDA approved for the {{{indicationType}}} of diabetes mellitus. Common adverse reactions include hypoglycemia, allergic reactions, injection site reactions, lipodystrophy, pruritus, and rash.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Diabetes Mellitus
- Dosing Information
- APIDRA is a recombinant insulin analog that is equipotent to human insulin (i.e. one unit of APIDRA has the same glucose-lowering effect as one unit of regular human insulin) when given intravenously. When given subcutaneously, APIDRA has a more rapid onset of action and a shorter duration of action than regular human insulin.
- The dosage of APIDRA must be individualized. Blood glucose monitoring is essential in all patients receiving insulin therapy.
- The total daily insulin requirement may vary and is usually between 0.5 to 1 Unit/kg/day. Insulin requirements may be altered during stress, major illness, or with changes in exercise, meal patterns, or coadministered drugs.
- Subcutaneous administration
- APIDRA should be given within 15 minutes before a meal or within 20 minutes after starting a meal.
- APIDRA given by subcutaneous injection should generally be used in regimens with an intermediate or long-acting insulin.
- APIDRA should be administered by subcutaneous injection in the abdominal wall, thigh, or upper arm. Injection sites should be rotated within the same region (abdomen, thigh or upper arm) from one injection to the next to reduce the risk of lipodystrophy.
- Continuous subcutaneous infusion (insulin pump)
- APIDRA may be administered by continuous subcutaneous infusion in the abdominal wall. Do not use diluted or mixed insulins in external insulin pumps. Infusion sites should be rotated within the same region to reduce the risk of lipodystrophy. The initial programming of the external insulin infusion pump should be based on the total daily insulin dose of the previous regimen.
- The following insulin pumps1 have been used in APIDRA clinical trials conducted by sanofi-aventis, the manufacturer of APIDRA:
- Disetronic® H-Tron® plus V100 and D-Tron® with Disetronic catheters (Rapid™, Rapid C™, Rapid D™, and Tender™)
- MiniMed® Models 506, 507, 507c and 508 with MiniMed catheters (Sof-set Ultimate QR™, and Quick-set™).
- Before using a different insulin pump with APIDRA, read the pump label to make sure the pump has been evaluated with APIDRA.
- Physicians and patients should carefully evaluate information on pump use in the APIDRA prescribing information, Patient Information Leaflet, and the pump manufacturer's manual. APIDRA-specific information should be followed for in-use time, frequency of changing infusion sets, or other details specific to APIDRA usage, because APIDRA-specific information may differ from general pump manual instructions. Failure to follow APIDRA-specific instructions may lead to serious adverse events.
- Patients administering APIDRA by continuous subcutaneous infusion must have an alternative insulin delivery system in case of pump system failure.
- Based on in vitro studies which have shown loss of the preservative, metacresol and insulin degradation, APIDRA in the reservoir should be changed at least every 48 hours. APIDRA should not be exposed to temperatures greater than 98.6°F (37°C).
- In clinical use, the infusion sets and the APIDRA in the reservoir must be changed at least every 48 hours.
- Intravenous administration
- APIDRA can be administered intravenously under medical supervision for glycemic control with close monitoring of blood glucose and serum potassium to avoid hypoglycemia and hypokalemia. For intravenous use, APIDRA should be used at concentrations of 0.05 Units/mL to 1 Unit/mL insulin glulisine in infusion systems using polyvinyl chloride (PVC) bags. APIDRA has been shown to be stable only in normal saline solution (0.9% sodium chloride). Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer insulin mixtures intravenously.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Insulin glulisine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Insulin glulisine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Diabetes Mellitus
- Dosing Information
- APIDRA is a recombinant insulin analog that is equipotent to human insulin (i.e. one unit of APIDRA has the same glucose-lowering effect as one unit of regular human insulin) when given intravenously. When given subcutaneously, APIDRA has a more rapid onset of action and a shorter duration of action than regular human insulin.
- The dosage of APIDRA must be individualized. Blood glucose monitoring is essential in all patients receiving insulin therapy.
- The total daily insulin requirement may vary and is usually between 0.5 to 1 Unit/kg/day. Insulin requirements may be altered during stress, major illness, or with changes in exercise, meal patterns, or coadministered drugs.
- Subcutaneous administration
- APIDRA should be given within 15 minutes before a meal or within 20 minutes after starting a meal.
- APIDRA given by subcutaneous injection should generally be used in regimens with an intermediate or long-acting insulin.
- APIDRA should be administered by subcutaneous injection in the abdominal wall, thigh, or upper arm. Injection sites should be rotated within the same region (abdomen, thigh or upper arm) from one injection to the next to reduce the risk of lipodystrophy.
- Continuous subcutaneous infusion (insulin pump)
- APIDRA may be administered by continuous subcutaneous infusion in the abdominal wall. Do not use diluted or mixed insulins in external insulin pumps. Infusion sites should be rotated within the same region to reduce the risk of lipodystrophy. The initial programming of the external insulin infusion pump should be based on the total daily insulin dose of the previous regimen.
- The following insulin pumps1 have been used in APIDRA clinical trials conducted by sanofi-aventis, the manufacturer of APIDRA:
- Disetronic® H-Tron® plus V100 and D-Tron® with Disetronic catheters (Rapid™, Rapid C™, Rapid D™, and Tender™)
- MiniMed® Models 506, 507, 507c and 508 with MiniMed catheters (Sof-set Ultimate QR™, and Quick-set™).
- Before using a different insulin pump with APIDRA, read the pump label to make sure the pump has been evaluated with APIDRA.
- Physicians and patients should carefully evaluate information on pump use in the APIDRA prescribing information, Patient Information Leaflet, and the pump manufacturer's manual. APIDRA-specific information should be followed for in-use time, frequency of changing infusion sets, or other details specific to APIDRA usage, because APIDRA-specific information may differ from general pump manual instructions. Failure to follow APIDRA-specific instructions may lead to serious adverse events.
- Patients administering APIDRA by continuous subcutaneous infusion must have an alternative insulin delivery system in case of pump system failure.
- Based on in vitro studies which have shown loss of the preservative, metacresol and insulin degradation, APIDRA in the reservoir should be changed at least every 48 hours. APIDRA should not be exposed to temperatures greater than 98.6°F (37°C).
- In clinical use, the infusion sets and the APIDRA in the reservoir must be changed at least every 48 hours.
- Intravenous administration
- APIDRA can be administered intravenously under medical supervision for glycemic control with close monitoring of blood glucose and serum potassium to avoid hypoglycemia and hypokalemia. For intravenous use, APIDRA should be used at concentrations of 0.05 Units/mL to 1 Unit/mL insulin glulisine in infusion systems using polyvinyl chloride (PVC) bags. APIDRA has been shown to be stable only in normal saline solution (0.9% sodium chloride). Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit. Do not administer insulin mixtures intravenously.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Insulin glulisine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Insulin glulisine in pediatric patients.
Contraindications
- APIDRA is contraindicated:
- during episodes of hypoglycemia
- in patients who are hypersensitive to APIDRA or to any of its excipients
- When used in patients with known hypersensitivity to APIDRA or its excipients, patients may develop localized or generalized hypersensitivity reactions.
Warnings
Precautions
- Dosage adjustment and monitoring
- Glucose monitoring is essential for patients receiving insulin therapy. Changes to an insulin regimen should be made cautiously and only under medical supervision. Changes in insulin strength, manufacturer, type, or method of administration may result in the need for a change in insulin dose. Concomitant oral antidiabetic treatment may need to be adjusted.
- As with all insulin preparations, the time course of action for APIDRA may vary in different individuals or at different times in the same individual and is dependent on many conditions, including the site of injection, local blood supply, or local temperature. Patients who change their level of physical activity or meal plan may require adjustment of insulin dosages.
- Hypoglycemia
- Hypoglycemia is the most common adverse reaction of insulin therapy, including APIDRA. The risk of hypoglycemia increases with tighter glycemic control. Patients must be educated to recognize and manage hypoglycemia. Severe hypoglycemia may lead to unconsciousness and/or convulsions and may result in temporary or permanent impairment of brain function or death. Severe hypoglycemia requiring the assistance of another person and/or parenteral glucose infusion or glucagon administration has been observed in clinical trials with insulin, including trials with APIDRA.
- The timing of hypoglycemia usually reflects the time-action profile of the administered insulin formulations. Other factors such as changes in food intake (e.g., amount of food or timing of meals), injection site, exercise, and concomitant medications may also alter the risk of hypoglycemia.
- As with all insulins, use caution in patients with hypoglycemia unawareness and in patients who may be predisposed to hypoglycemia (e.g., the pediatric population and patients who fast or have erratic food intake). The patient's ability to concentrate and react may be impaired as a result of hypoglycemia. This may present a risk in situations where these abilities are especially important, such as driving or operating other machinery.
- Rapid changes in serum glucose levels may induce symptoms similar to hypoglycemia in persons with diabetes, regardless of the glucose value. Early warning symptoms of hypoglycemia may be different or less pronounced under certain conditions, such as longstanding diabetes, diabetic nerve disease, use of medications such as beta-blockers, or intensified diabetes control. These situations may result in severe hypoglycemia (and, possibly, loss of consciousness) prior to the patient's awareness of hypoglycemia.
- Intravenously administered insulin has a more rapid onset of action than subcutaneously administered insulin, requiring closer monitoring for hypoglycemia.
- Hypersensitivity and allergic reactions
- Severe, life-threatening, generalized allergy, including anaphylaxis, can occur with insulin products, including APIDRA.
- Hypokalemia
- All insulin products, including APIDRA, cause a shift in potassium from the extracellular to intracellular space, possibly leading to hypokalemia. Untreated hypokalemia may cause respiratory paralysis, ventricular arrhythmia, and death. Use caution in patients who may be at risk for hypokalemia (e.g., patients using potassium-lowering medications, patients taking medications sensitive to serum potassium concentrations). Monitor glucose and potassium frequently when APIDRA is administered intravenously.
- Renal or hepatic impairment
- Frequent glucose monitoring and insulin dose reduction may be required in patients with renal or hepatic impairment.
- Mixing of insulins
- APIDRA for subcutaneous injection should not be mixed with insulin preparations other than NPH insulin. If APIDRA is mixed with NPH insulin, APIDRA should be drawn into the syringe first. Injection should occur immediately after mixing.
- Do not mix APIDRA with other insulins for intravenous administration or for use in a continuous subcutaneous infusion pump.
- APIDRA for intravenous administration should not be diluted with solutions other than 0.9% sodium chloride (normal saline). The efficacy and safety of mixing APIDRA with diluents or other insulins for use in external subcutaneous infusion pumps have not been established.
- Subcutaneous insulin infusion pumps
- When used in an external insulin pump for subcutaneous infusion, APIDRA should not be diluted or mixed with any other insulin. APIDRA in the reservoir must be changed at least every 48 hours. APIDRA should not be exposed to temperatures greater than 98.6°F (37°C).
- Malfunction of the insulin pump or infusion set or handling errors or insulin degradation can rapidly lead to hyperglycemia, ketosis and diabetic ketoacidosis. Prompt identification and correction of the cause of hyperglycemia or ketosis or diabetic ketoacidosis is necessary. Interim subcutaneous injections with APIDRA may be required. Patients using continuous subcutaneous insulin infusion pump therapy must be trained to administer insulin by injection and have alternate insulin therapy available.
- Intravenous administration
- When APIDRA is administered intravenously, glucose and potassium levels must be closely monitored to avoid potentially fatal hypoglycemia and hypokalemia.
- Do not mix APIDRA with other insulins for intravenous administration. APIDRA may be diluted only in normal saline solution.
- Drug interactions
- Some medications may alter insulin requirements and the risk for hypoglycemia or hyperglycemia.
- Fluid retention and heart failure with concomitant use of PPAR-gamma agonists
- Thiazolidinediones (TZDs), which are peroxisome proliferator-activated receptor (PPAR)gamma agonists, can cause dose-related fluid retention, particularly when used in combination with insulin. Fluid retention may lead to or exacerbate heart failure. Patients treated with insulin, including APIDRA and a PPAR-gamma agonist should be observed for signs and symptoms of heart failure. If heart failure develops, it should be managed according to current standards of care, and discontinuation or dose reduction of the PPAR-gamma agonist must be considered.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying designs, the adverse reaction rates reported in one clinical trial may not be easily compared to those rates reported in another clinical trial, and may not reflect the rates actually observed in clinical practice.
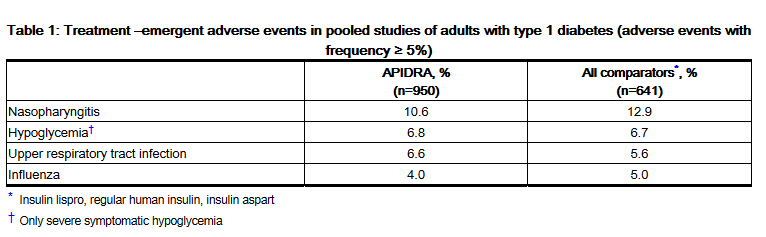
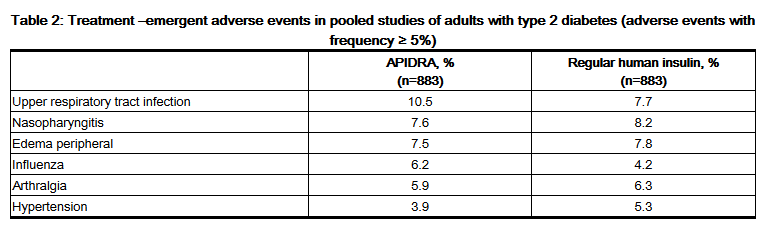
- The frequencies of adverse drug reactions during APIDRA clinical trials in patients with type 1 diabetes mellitus and type 2 diabetes mellitus are listed in the tables below.
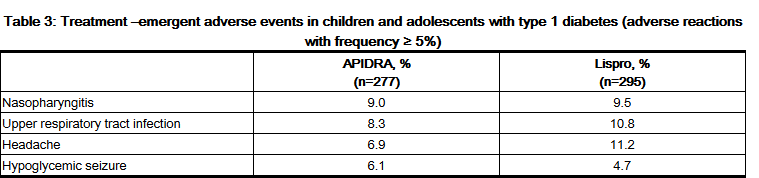
- Pediatrics
- Table 3 summarizes the adverse reactions occurring with frequency higher than 5% in a clinical study in children and adolescents with type 1 diabetes treated with APIDRA (n=277) or insulin lispro (n=295).
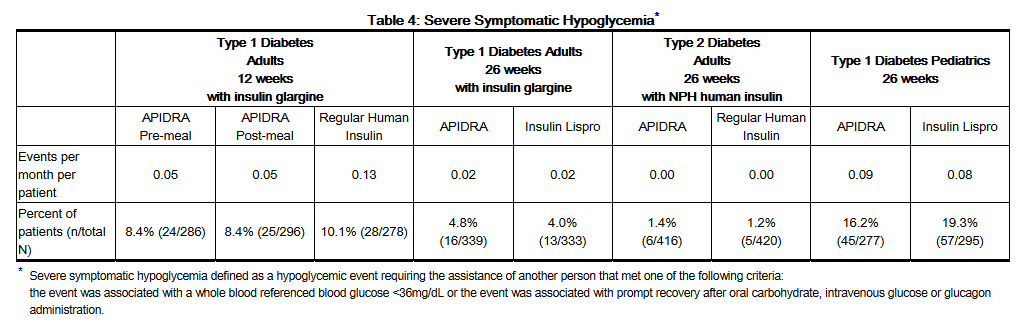
- Severe symptomatic hypoglycemia
- Hypoglycemia is the most commonly observed adverse reaction in patients using insulin, including APIDRA. The rates and incidence of severe symptomatic hypoglycemia, defined as hypoglycemia requiring intervention from a third party, were comparable for all treatment regimens (see Table 4). In the phase 3 clinical trial, children and adolescents with type 1 diabetes had a higher incidence of severe symptomatic hypoglycemia in the two treatment groups compared to adults with type 1 diabetes. (see Table 4).
- Insulin initiation and intensification of glucose control
- Intensification or rapid improvement in glucose control has been associated with a transitory, reversible ophthalmologic refraction disorder, worsening of diabetic retinopathy, and acute painful peripheral neuropathy. However, long-term glycemic control decreases the risk of diabetic retinopathy and neuropathy.
- Lipodystrophy
- Long-term use of insulin, including APIDRA, can cause lipodystrophy at the site of repeated insulin injections or infusion. Lipodystrophy includes lipohypertrophy (thickening of adipose tissue) and lipoatrophy (thinning of adipose tissue), and may affect insulin absorption. Rotate insulin injection or infusion sites within the same region to reduce the risk of lipodystrophy.
- Weight gain
- Weight gain can occur with insulin therapy, including APIDRA, and has been attributed to the anabolic effects of insulin and the decrease in glucosuria.
- Peripheral Edema
- Insulin, including APIDRA, may cause sodium retention and edema, particularly if previously poor metabolic control is improved by intensified insulin therapy.
- Adverse Reactions with Continuous Subcutaneous Insulin Infusion (CSII)
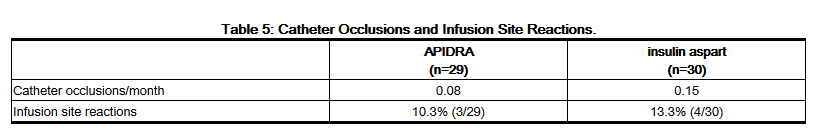
- In a 12-week randomized study in patients with type 1 diabetes (n=59), the rates of catheter occlusions and infusion site reactions were similar for APIDRA and insulin aspart treated patients (Table 5).
- Allergic Reactions
- Local Allergy
- As with any insulin therapy, patients taking APIDRA may experience redness, swelling, or itching at the site of injection. These minor reactions usually resolve in a few days to a few weeks, but in some occasions may require discontinuation of APIDRA. In some instances, these reactions may be related to factors other than insulin, such as irritants in a skin cleansing agent or poor injection technique.
- Systemic Allergy
- Severe, life-threatening, generalized allergy, including anaphylaxis, may occur with any insulin, including APIDRA. Generalized allergy to insulin may cause whole body rash (including pruritus), dyspnea, wheezing, hypotension, tachycardia, or diaphoresis.
- In controlled clinical trials up to 12 months duration, potential systemic allergic reactions were reported in 79 of 1833 patients (4.3%) who received APIDRA and 58 of 1524 patients (3.8%) who received the comparator short-acting insulins. During these trials treatment with APIDRA was permanently discontinued in 1 of 1833 patients due to a potential systemic allergic reaction.
- Localized reactions and generalized myalgias have been reported with the use of metacresol, which is an excipient of APIDRA.
- Antibody Production
- In a study in patients with type 1 diabetes (n=333), the concentrations of insulin antibodies that react with both human insulin and insulin glulisine (cross-reactive insulin antibodies) remained near baseline during the first 6 months of the study in the patients treated with APIDRA. A decrease in antibody concentration was observed during the following 6 months of the study. In a study in patients with type 2 diabetes (n=411), a similar increase in cross-reactive insulin antibody concentration was observed in the patients treated with APIDRA and in the patients treated with human insulin during the first 9 months of the study. Thereafter the concentration of antibodies decreased in the APIDRA patients and remained stable in the human insulin patients. There was no correlation between cross-reactive insulin antibody concentration and changes in HbA1c, insulin doses, or incidence of hypoglycemia. The clinical significance of these antibodies is not known.
- APIDRA did not elicit a significant antibody response in a study of children and adolescents with type 1 diabetes.
Postmarketing Experience
- The following adverse reactions have been identified during post-approval use of APIDRA.
- Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to estimate reliably their frequency or establish a causal relationship to drug exposure.
- Medication errors have been reported in which other insulins, particularly long-acting insulins, have been accidentally administered instead of APIDRA.
Drug Interactions
- A number of drugs affect glucose metabolism and may necessitate insulin dose adjustment and particularly close monitoring.
- Drugs that may increase the blood glucose-lowering effect of insulins including APIDRA, and therefore increase the risk of hypoglycemia, include oral antidiabetic products, pramlintide, ACE inhibitors, disopyramide, fibrates, fluoxetine, monoamine oxidase inhibitors, propoxyphene, pentoxifylline, salicylates, somatostatin analogs, and sulfonamide antibiotics.
- Drugs that may reduce the blood-glucose-lowering effect of APIDRA include corticosteroids, niacin, danazol, diuretics, sympathomimetic agents (e.g., epinephrine, albuterol, terbutaline), glucagon, isoniazid, phenothiazine derivatives, somatropin, thyroid hormones, estrogens, progestogens (e.g., in oral contraceptives), protease inhibitors, and atypical antipsychotics.
- Beta-blockers, clonidine, lithium salts, and alcohol may either increase or decrease the blood-glucose-lowering effect of insulin.
- Pentamidine may cause hypoglycemia, which may sometimes be followed by hyperglycemia.
- The signs of hypoglycemia may be reduced or absent in patients taking anti-adrenergic drugs such as beta-blockers, clonidine, guanethidine, and reserpine.
Use in Specific Populations
Pregnancy
- Pregnancy Category C
- Reproduction and teratology studies have been performed with insulin glulisine in rats and rabbits using regular human insulin as a comparator. Insulin glulisine was given to female rats throughout pregnancy at subcutaneous doses up to 10 Units/kg once daily (dose resulting in an exposure 2 times the average human dose, based on body surface area comparison) and did not have any remarkable toxic effects on embryo-fetal development.
- Insulin glulisine was given to female rabbits throughout pregnancy at subcutaneous doses up to 1.5 Units/kg/day (dose resulting in an exposure 0.5 times the average human dose, based on body surface area comparison). Adverse effects on embryo-fetal development were only seen at maternal toxic dose levels inducing hypoglycemia. Increased incidence of post-implantation losses and skeletal defects were observed at a dose level of 1.5 Units/kg once daily (dose resulting in an exposure 0.5 times the average human dose, based on body surface area comparison) that also caused mortality in dams. A slight increased incidence of post-implantation losses was seen at the next lower dose level of 0.5 Units/kg once daily (dose resulting in an exposure 0.2 times the average human dose, based on body surface area comparison) which was also associated with severe hypoglycemia but there were no defects at that dose. No effects were observed in rabbits at a dose of 0.25 Units/kg once daily (dose resulting in an exposure 0.1 times the average human dose, based on body surface area comparison). The effects of insulin glulisine did not differ from those observed with subcutaneous regular human insulin at the same doses and were attributed to secondary effects of maternal hypoglycemia.
- There are no well-controlled clinical studies of the use of APIDRA in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. It is essential for patients with diabetes or a history of gestational diabetes to maintain good metabolic control before conception and throughout pregnancy. Insulin requirements may decrease during the first trimester, generally increase during the second and third trimesters, and rapidly decline after delivery. Careful monitoring of glucose control is essential in these patients.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Insulin glulisine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Insulin glulisine during labor and delivery.
Nursing Mothers
- It is unknown whether insulin glulisine is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when APIDRA is administered to a nursing woman. Use of APIDRA is compatible with breastfeeding, but women with diabetes who are lactating may require adjustments of their insulin doses.
Pediatric Use
- The safety and effectiveness of subcutaneous injections of APIDRA have been established in pediatric patients (age 4 to 17 years) with type 1 diabetes. APIDRA has not been studied in pediatric patients with type 1 diabetes younger than 4 years of age and in pediatric patients with type 2 diabetes.
- As in adults, the dosage of APIDRA must be individualized in pediatric patients based on metabolic needs and frequent monitoring of blood glucose.
Geriatic Use
- In clinical trials (n=2408), APIDRA was administered to 147 patients ≥65 years of age and 27 patients ≥75 years of age. The majority of this small subset of elderly patients had type 2 diabetes. The change in HbA1c values and hypoglycemia frequencies did not differ by age. Nevertheless, caution should be exercised when APIDRA is administered to geriatric patients.
Gender
There is no FDA guidance on the use of Insulin glulisine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Insulin glulisine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Insulin glulisine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Insulin glulisine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Insulin glulisine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Insulin glulisine in patients who are immunocompromised.
Administration and Monitoring
Administration
- Subcutaneous
Monitoring
There is limited information regarding Monitoring of Insulin glulisine in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Insulin glulisine in the drug label.
Overdosage
Acute Overdose
Signs and Symptoms
- Excess insulin may cause hypoglycemia and, particularly when given intravenously, hypokalemia. Sustained carbohydrate intake and observation may be necessary because hypoglycemia may recur after apparent clinical recovery. Hypokalemia must be corrected appropriately.
Management
- Mild episodes of hypoglycemia usually can be treated with oral glucose. Adjustments in drug dosage, meal patterns, or exercise may be needed. More severe episodes of hypoglycemia with coma, seizure, or neurologic impairment may be treated with intramuscular/subcutaneous glucagon or concentrated intravenous glucose.
Chronic Overdose
There is limited information regarding Chronic Overdose of Insulin glulisine in the drug label.
Pharmacology
Insulin glulisine
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | |
| ATC code | A10 |
| PubChem | ? |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 5823 g/mol |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
C(US) |
| Legal status |
POM(UK) [[Prescription drug|Template:Unicode-only]](US) |
| Routes | Subcutaneous |
Mechanism of Action
- Regulation of glucose metabolism is the primary activity of insulins and insulin analogs, including insulin glulisine. Insulins lower blood glucose by stimulating peripheral glucose uptake by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulins inhibit lipolysis and proteolysis, and enhance protein synthesis.
- The glucose lowering activities of APIDRA and of regular human insulin are equipotent when administered by the intravenous route. After subcutaneous administration, the effect of APIDRA is more rapid in onset and of shorter duration compared to regular human insulin.
Structure
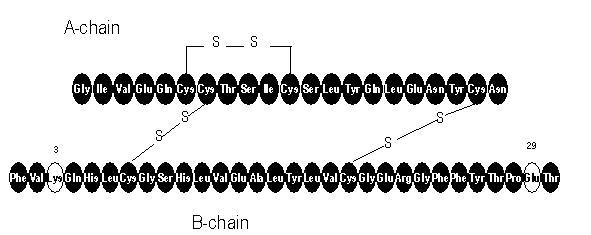
- APIDRA® (insulin glulisine [rDNA origin] injection) is a rapid-acting human insulin analog used to lower blood glucose. Insulin glulisine is produced by recombinant DNA technology utilizing a non-pathogenic laboratory strain of Escherichia coli (K12). Insulin glulisine differs from human insulin in that the amino acid asparagine at position B3 is replaced by lysine and the lysine in position B29 is replaced by glutamic acid. Chemically, insulin glulisine is 3B-lysine-29B-glutamic acid-human insulin, has the empirical formula C258H384N64O78S6 and a molecular weight of 5823 and has the following structural formula:
- APIDRA is a sterile, aqueous, clear, and colorless solution. Each milliliter of APIDRA contains 100 units (3.49 mg) insulin glulisine, 3.15 mg metacresol, 6 mg tromethamine, 5 mg sodium chloride, 0.01 mg polysorbate 20, and water for injection. APIDRA has a pH of approximately 7.3. The pH is adjusted by addition of aqueous solutions of hydrochloric acid and/or sodium hydroxide.
Pharmacodynamics
- Studies in healthy volunteers and patients with diabetes demonstrated that APIDRA has a more rapid onset of action and a shorter duration of activity than regular human insulin when given subcutaneously.
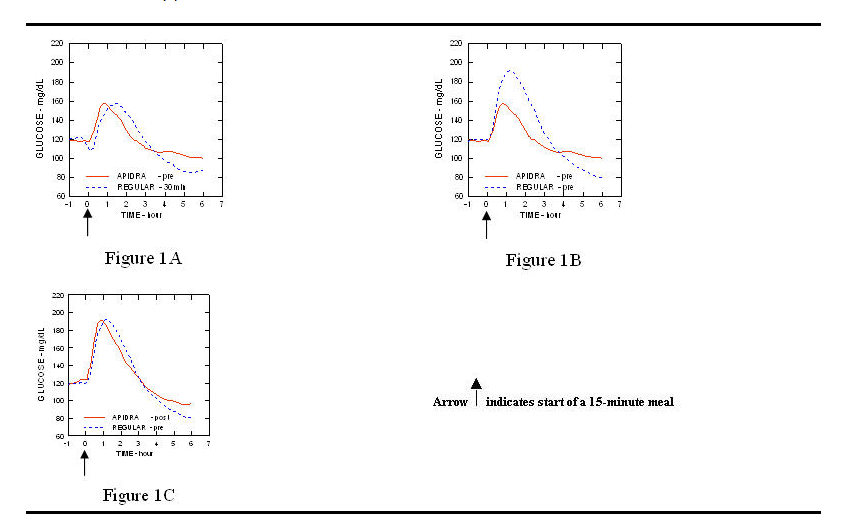
- In a study in patients with type 1 diabetes (n= 20), the glucose-lowering profiles of APIDRA and regular human insulin were assessed at various times in relation to a standard meal at a dose of 0.15 Units/kg. (Figure 1.)
- The maximum blood glucose excursion (ΔGLUmax; baseline subtracted glucose concentration) for APIDRA injected 2 minutes before a meal was 65 mg/dL compared to 64 mg/dL for regular human insulin injected 30 minutes before a meal (see Figure 1A), and 84 mg/dL for regular human insulin injected 2 minutes before a meal (see Figure 1B). The maximum blood glucose excursion for APIDRA injected 15 minutes after the start of a meal was 85 mg/dL compared to 84 mg/dL for regular human insulin injected 2 minutes before a meal (see Figure 1C).
- Figure 1. Serial mean blood glucose collected up to 6 hours following a single dose of APIDRA and regular human insulin. APIDRA given 2 minutes (APIDRA - pre) before the start of a meal compared to regular human insulin given 30 minutes (Regular - 30 min) before start of the meal (Figure 1A) and compared to regular human insulin (Regular - pre) given 2 minutes before a meal (Figure 1B). APIDRA given 15 minutes (APIDRA - post) after start of a meal compared to regular human insulin (Regular - pre) given 2 minutes before a meal (Figure 1C). On the x-axis zero (0) is the start of a 15-minute meal.
- In a randomized, open-label, two-way crossover study, 16 healthy male subjects received an intravenous infusion of APIDRA or regular human insulin with saline diluent at a rate of 0.8 milliUnits/kg/min for two hours. Infusion of the same dose of APIDRA or regular human insulin produced equivalent glucose disposal at steady state.
Pharmacokinetics
- Absorption and bioavailability
- Pharmacokinetic profiles in healthy volunteers and patients with diabetes (type 1 or type 2) demonstrated that absorption of insulin glulisine was faster than that of regular human insulin.
- In a study in patients with type 1 diabetes (n=20) after subcutaneous administration of 0.15 Units/kg, the median time to maximum concentration (Tmax) was 60 minutes (range 40 to 120 minutes) and the peak concentration (Cmax) was 83 microUnits/mL (range 40 to 131 microUnits/mL) for insulin glulisine compared to a median Tmax of 120 minutes (range 60 to 239 minutes) and a Cmax of 50 microUnits/mL (range 35 to 71 microUnits/mL) for regular human insulin. (Figure 2)
- Figure 2. Pharmacokinetic profiles of insulin glulisine and regular human insulin in patients with type 1 diabetes after a dose of 0.15 Units/kg.
- Insulin glulisine and regular human insulin were administered subcutaneously at a dose of 0.2 Units/kg in an euglycemic clamp study in patients with type 2 diabetes (n=24) and a body mass index (BMI) between 20 and 36 kg/m2. The median time to maximum concentration (Tmax) was 100 minutes (range 40 to 120 minutes) and the median peak concentration (Cmax) was 84 microUnits/mL (range 53 to 165 microUnits/mL) for insulin glulisine compared to a median Tmax of 240 minutes (range 80 to 360 minutes) and a median Cmax of 41 microUnits/mL (range 33 to 61 microUnits/mL) for regular human insulin. (Figure 3.)
- Figure 3. Pharmacokinetic profiles of insulin glulisine and regular human insulin in patients with type 2 diabetes after a subcutaneous dose of 0.2 Units/kg.
- When APIDRA was injected subcutaneously into different areas of the body, the time-concentration profiles were similar. The absolute bioavailability of insulin glulisine after subcutaneous administration is approximately 70%, regardless of injection area (abdomen 73%, deltoid 71%, thigh 68%).
- In a clinical study in healthy volunteers (n=32) the total insulin glulisine bioavailability was similar after subcutaneous injection of insulin glulisine and NPH insulin (premixed in the syringe) and following separate simultaneous subcutaneous injections. There was 27% attenuation of the maximum concentration (Cmax) of APIDRA after premixing; however, the time to maximum concentration (Tmax) was not affected. No data are available on mixing APIDRA with insulin preparations other than NPH insulin.
- Distribution and elimination
- The distribution and elimination of insulin glulisine and regular human insulin after intravenous administration are similar with volumes of distribution of 13 and 21 L and half-lives of 13 and 17 minutes, respectively. After subcutaneous administration, insulin glulisine is eliminated more rapidly than regular human insulin with an apparent half-life of 42 minutes compared to 86 minutes.
- Pediatric patients
- The pharmacokinetic and pharmacodynamic properties of APIDRA and regular human insulin were assessed in a study conducted in children 7 to 11 years old (n=10) and adolescents 12 to 16 years old (n=10) with type 1 diabetes. The relative differences in pharmacokinetics and pharmacodynamics between APIDRA and regular human insulin in these patients with type 1 diabetes were similar to those in healthy adult subjects and adults with type 1 diabetes.
- Race
- A study in 24 healthy Caucasians and Japanese subjects compared the pharmacokinetics and pharmacodynamics after subcutaneous injection of insulin glulisine, insulin lispro, and regular human insulin. With subcutaneous injection of insulin glulisine, Japanese subjects had a greater initial exposure (33%) for the ratio of AUC(0–1h) to AUC(0–clamp end) than Caucasians (21%) although the total exposures were similar. There were similar findings with insulin lispro and regular human insulin.
- Obesity
- Insulin glulisine and regular human insulin were administered subcutaneously at a dose of 0.3 Units/kg in a euglycemic clamp study in obese, non-diabetic subjects (n=18) with a body mass index (BMI) between 30 and 40 kg/m2. The median time to maximum concentration (Tmax) was 85 minutes (range 49 to 150 minutes) and the median peak concentration (Cmax) was 192 microUnits/mL (range 98 to 380 microUnits/mL) for insulin glulisine compared to a median Tmax of 150 minutes (range 90 to 240 minutes) and a median Cmax of 86 microUnits/mL (range 43 to 175 microUnits/mL) for regular human insulin.
- The more rapid onset of action and shorter duration of activity of APIDRA and insulin lispro compared to regular human insulin were maintained in an obese non-diabetic population (n= 18). (Figure 4.)
- Figure 4. Glucose infusion rates (GIR) in a euglycemic clamp study after subcutaneous injection of 0.3 Units/kg of APIDRA, insulin lispro or regular human insulin in an obese population.
- Renal impairment
- Studies with human insulin have shown increased circulating levels of insulin in patients with renal failure. In a study performed in 24 non-diabetic subjects with normal renal function (ClCr >80 mL/min), moderate renal impairment (30–50 mL/min) and severe renal impairment (<30 mL/min), the subjects with moderate and severe renal impairment had increased exposure to insulin glulisine by 29% to 40% and reduced clearance of insulin glulisine by 20% to 25% compared to subjects with normal renal function.
- Hepatic impairment
- The effect of hepatic impairment on the pharmacokinetics and pharmacodynamics of APIDRA has not been studied. Some studies with human insulin have shown increased circulating levels of insulin in patients with liver failure.
- Gender
- The effect of gender on the pharmacokinetics and pharmacodynamics of APIDRA has not been studied.
- Pregnancy
- The effect of pregnancy on the pharmacokinetics and pharmacodynamics of APIDRA has not been studied.
- Smoking
- The effect of smoking on the pharmacokinetics and pharmacodynamics of APIDRA has not been studied.
Nonclinical Toxicology
Carcinogenesis, mutagenesis, impairment of fertility
- Standard 2-year carcinogenicity studies in animals have not been performed. In Sprague Dawley rats, a 12-month repeat dose toxicity study was conducted with insulin glulisine at subcutaneous doses of 2.5, 5, 20 or 50 Units/kg twice daily (dose resulting in an exposure 1, 2, 8, and 20 times the average human dose, based on body surface area comparison).
- There was a non-dose dependent higher incidence of mammary gland tumors in female rats administered insulin glulisine compared to untreated controls. The incidence of mammary tumors for insulin glulisine and regular human insulin was similar. The relevance of these findings to humans is not known. Insulin glulisine was not mutagenic in the following tests: Ames test, in vitro mammalian chromosome aberration test in V79 Chinese hamster cells, and in vivo mammalian erythrocyte micronucleus test in rats.
- In fertility studies in male and female rats at subcutaneous doses up to 10 Units/kg once daily (dose resulting in an exposure 2 times the average human dose, based on body surface area comparison), no clear adverse effects on male and female fertility, or general reproductive performance of animals were observed.
Clinical Studies
- The safety and efficacy of APIDRA was studied in adult patients with type 1 and type 2 diabetes (n =1833) and in children and adolescent patients (4 to 17 years) with type 1 diabetes (n=572). The primary efficacy parameter in these trials was glycemic control, assessed using glycated hemoglobin (GHb reported as HbA1c equivalent).
- Type 1 Diabetes-Adults
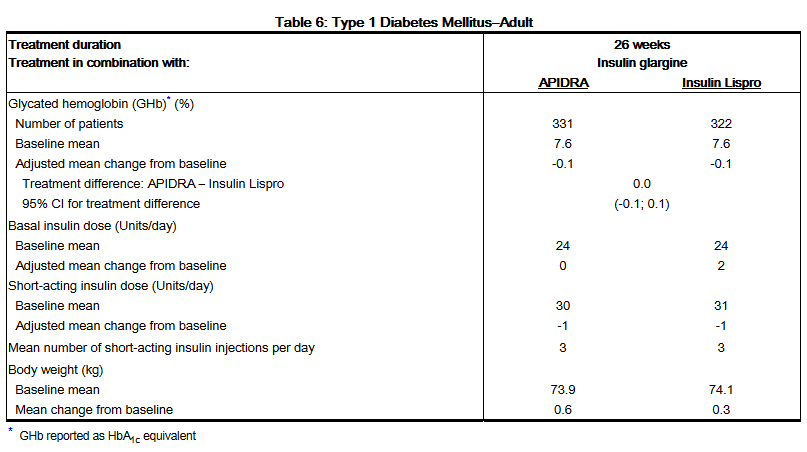
- A 26-week, randomized, open-label, active-controlled, non-inferiority study was conducted in patients with type 1 diabetes to assess the safety and efficacy of APIDRA (n= 339) compared to insulin lispro (n= 333) when administered subcutaneously within 15 minutes before a meal. Insulin glargine was administered once daily in the evening as the basal insulin. There was a 4-week run-in period with insulin lispro and insulin glargine prior to randomization. Most patients were Caucasian (97%). Fifty eight percent of the patients were men. The mean age was 39 years (range 18 to 74 years). Glycemic control, the number of daily short-acting insulin injections and the total daily doses of APIDRA and insulin lispro were similar in the two treatment groups (Table 6).
- Type 2 Diabetes-Adults
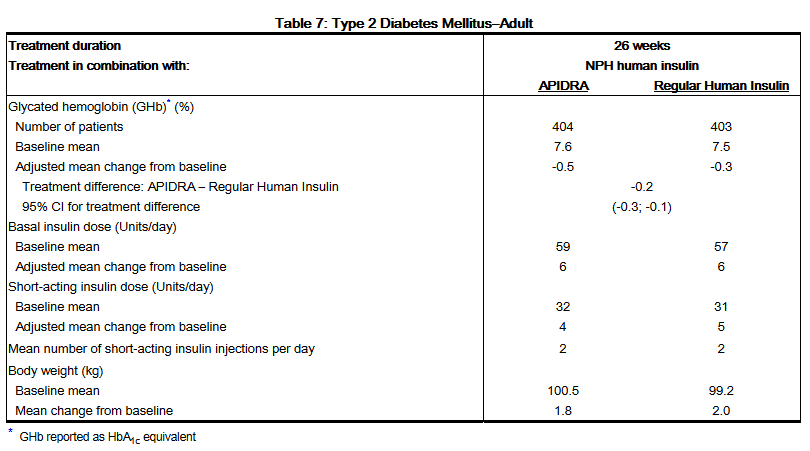
- A 26-week, randomized, open-label, active-controlled, non-inferiority study was conducted in insulin-treated patients with type 2 diabetes to assess the safety and efficacy of APIDRA (n= 435) given within 15 minutes before a meal compared to regular human insulin (n=441) administered 30 to 45 minutes prior to a meal. NPH human insulin was given twice a day as the basal insulin. All patients participated in a 4-week run-in period with regular human insulin and NPH human insulin. Eighty-five percent of patients were Caucasian and 11% were Black. The mean age was 58 years (range 26 to 84 years). The average body mass index (BMI) was 34.6 kg/m2. At randomization, 58% of the patients were taking an oral antidiabetic agent. These patients were instructed to continue use of their oral antidiabetic agent at the same dose throughout the trial. The majority of patients (79%) mixed their short-acting insulin with NPH human insulin immediately prior to injection. The reductions from baseline in GHb were similar between the 2 treatment groups (see Table 7). No differences between APIDRA and regular human insulin groups were seen in the number of daily short-acting insulin injections or basal or short-acting insulin doses. (See Table 7.)
- Type 1 Diabetes-Adults: Pre- and post-meal administration
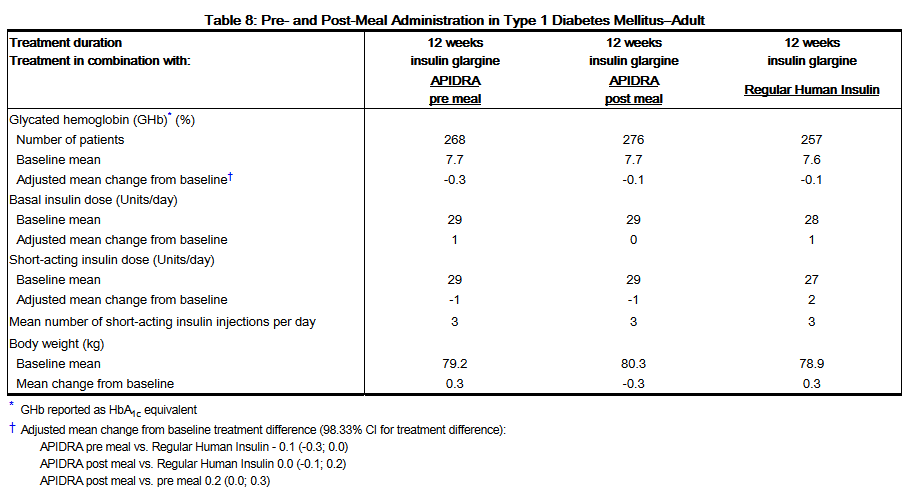
- A 12-week, randomized, open-label, active-controlled, non-inferiority study was conducted in patients with type 1 diabetes to assess the safety and efficacy of APIDRA administered at different times with respect to a meal. APIDRA was administered subcutaneously either within 15 minutes before a meal (n=286) or immediately after a meal (n=296) and regular human insulin (n= 278) was administered subcutaneously 30 to 45 minutes prior to a meal. Insulin glargine was administered once daily at bedtime as the basal insulin. There was a 4-week run-in period with regular human insulin and insulin glargine followed by randomization. Most patients were Caucasian (94%). The mean age was 40 years (range 18 to 73 years). Glycemic control (see Table 8) was comparable for the 3 treatment regimens. No changes from baseline between the treatments were seen in the total daily number of short-acting insulin injections. (See Table 8.)
- Type 1 Diabetes-Pediatric patients
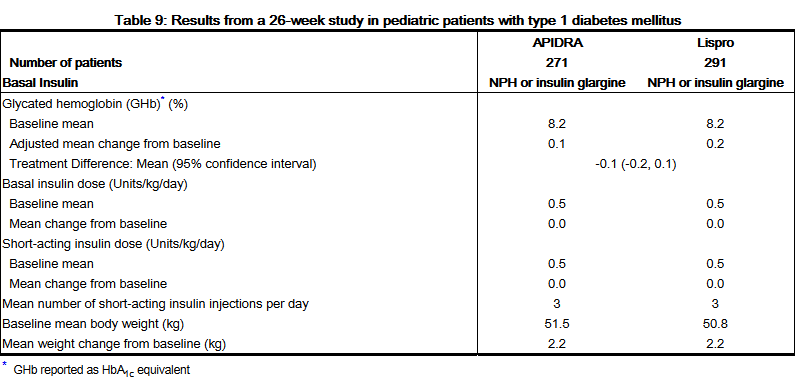
- A 26-week, randomized, open-label, active-controlled, non-inferiority study was conducted in children and adolescents older than 4 years of age with type 1 diabetes mellitus to assess the safety and efficacy of APIDRA (n= 277) compared to insulin lispro (n= 295) when administered subcutaneously within 15 minutes before a meal. Patients also received insulin glargine (administered once daily in the evening) or NPH insulin (administered once in the morning and once in the evening). There was a 4-week run-in period with insulin lispro and insulin glargine or NPH prior to randomization. Most patients were Caucasian (91%). Fifty percent of the patients were male. The mean age was 12.5 years (range 4 to 17 years). Mean BMI was 20.6 kg/m2. Glycemic control (see Table 9) was comparable for the two treatment regimens.
- Type 1 Diabetes-Pediatric patients
- A 26-week, randomized, open-label, active-controlled, non-inferiority study was conducted in children and adolescents older than 4 years of age with type 1 diabetes mellitus to assess the safety and efficacy of APIDRA (n= 277) compared to insulin lispro (n= 295) when administered subcutaneously within 15 minutes before a meal. Patients also received insulin glargine (administered once daily in the evening) or NPH insulin (administered once in the morning and once in the evening). There was a 4-week run-in period with insulin lispro and insulin glargine or NPH prior to randomization. Most patients were Caucasian (91%). Fifty percent of the patients were male. The mean age was 12.5 years (range 4 to 17 years). Mean BMI was 20.6 kg/m2. Glycemic control (see Table 9) was comparable for the two treatment regimens.
How Supplied
- APIDRA 100 units per mL (U-100) is available as:
- 10 mL vials NDC 0088-2500-33
- 3 mL SoloStar prefilled pen, package of 5 NDC 0088-2502-05
- Pen needles are not included in the packs.
- Solostar is compatible with all pen needles from Becton Dickinson and Company, Ypsomed and Owen Mumford.
- Storage
- Do not use after the expiration date.
- Unopened Vial/ SoloStar
- Unopened APIDRA vials and SoloStar should be stored in a refrigerator, 36°F–46°F (2°C–8°C). Protect from light. APIDRA should not be stored in the freezer and it should not be allowed to freeze. Discard if it has been frozen.
- Unopened vials/SoloStar not stored in a refrigerator must be used within 28 days.
- Open (In-Use) Vial:
- Opened vials, whether or not refrigerated, must be used within 28 days. If refrigeration is not possible, the open vial in use can be kept unrefrigerated for up to 28 days away from direct heat and light, as long as the temperature is not greater than 77°F (25°C).
- Open (In-Use) SoloStar prefilled pen:
- The opened (in-use) SoloStar should NOT be refrigerated but should be kept below 77°F (25°C) away from direct heat and light. The opened (in-use) SoloStar kept at room temperature must be discarded after 28 days.
- Infusion sets:
- Infusion sets (reservoirs, tubing, and catheters) and the APIDRA in the reservoir must be discarded after 48 hours of use or after exposure to temperatures that exceed 98.6°F (37°C).
- Intravenous use:
- Infusion bags prepared as indicated under DOSAGE AND ADMINISTRATION (2.4) are stable at room temperature for 48 hours.
- Preparation and handling
- After dilution for intravenous use, the solution should be inspected visually for particulate matter and discoloration prior to administration. Do not use the solution if it has become cloudy or contains particles; use only if it is clear and colorless. APIDRA is not compatible with Dextrose solution and Ringers solution and, therefore, cannot be used with these solution fluids. The use of APIDRA with other solutions has not been studied and is, therefore, not recommended.
Storage
There is limited information regarding Insulin glulisine Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Insulin glulisine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Insulin glulisine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Instructions for all patients
- Patients should be instructed on self-management procedures including glucose monitoring, proper injection technique, and management of hypoglycemia and hyperglycemia.
- Patients must be instructed on handling of special situations such as intercurrent conditions (illness, stress, or emotional disturbances), an inadequate or skipped insulin dose, inadvertent administration of an increased insulin dose, inadequate food intake, and skipped meals.
- Refer patients to the APIDRA Patient Information Leaflet for additional information.
- Women with diabetes should be advised to inform their doctor if they are pregnant or are contemplating pregnancy.
- Accidental mix-ups between APIDRA and other insulins, particularly long-acting insulins, have been reported. To avoid medication errors between APIDRA and other insulins, patients should be instructed to always check the insulin label before each injection.
- For patients using continuous subcutaneous insulin pumps
- Patients using external pump infusion therapy should be trained appropriately.
- The following insulin pumps1 have been used in APIDRA clinical trials conducted by sanofi-aventis, the manufacturer of APIDRA:
- Disetronic® H-Tron® plus V100 and D-Tron® with Disetronic catheters (Rapid™, Rapid C™, Rapid D™, and Tender™)
- MiniMed® Models 506, 507, 507c and 508 with MiniMed catheters (Sof-set Ultimate QR™, and Quick-set™).
- Before using a different insulin pump with APIDRA, read the pump label to make sure the pump has been evaluated with APIDRA.
- To minimize insulin degradation, infusion set occlusion, and loss of the preservative (metacresol), the infusion sets (reservoir, tubing, and catheter) and the APIDRA in the reservoir must be replaced at least every 48 hours and a new infusion site should be selected. The temperature of the insulin may exceed ambient temperature when the pump housing, cover, tubing or sport case is exposed to sunlight or radiant heat. Insulin exposed to temperatures higher than 98.6°F (37°C) should be discarded. Infusion sites that are erythematous, pruritic, or thickened should be reported to the healthcare professional, and a new site selected because continued infusion may increase the skin reaction or alter the absorption of APIDRA.
- Pump or infusion set malfunctions or handling errors or insulin degradation can lead to rapid hyperglycemia, and ketosis and diabetic ketoacidosis. This is especially pertinent for rapid-acting insulin analogs that are more rapidly absorbed through skin and have a shorter duration of action. Prompt identification and correction of the cause of hyperglycemia or ketosis or diabetic ketoacidosis is necessary. Problems include pump malfunction, infusion set occlusion, leakage, disconnection or kinking, handling errors and degraded insulin. Less commonly, hypoglycemia from pump malfunction may occur. If these problems cannot be promptly corrected, patients should resume therapy with subcutaneous insulin injection and contact their healthcare professional. Patients administering APIDRA by continuous subcutaneous infusion must have an alternative insulin delivery system in case of pump system failure.
Precautions with Alcohol
- Alcohol-Insulin glulisine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Apidra®[1]
Look-Alike Drug Names
There is limited information regarding Insulin glulisine Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "APIDRA (insulin glulisine) injection, solution APIDRA SOLOSTAR (insulin glulisine) injection, solution [Sanofi-Aventis U.S. LLC]". line feed character in
|title=at position 103 (help)
{{#subobject:
|Page Name=Insulin glulisine |Pill Name=No image.jpg |Drug Name= |Pill Ingred=|+sep=; |Pill Imprint= |Pill Dosage= |Pill Color=|+sep=; |Pill Shape= |Pill Size (mm)= |Pill Scoring= |Pill Image= |Drug Author= |NDC=
}}
{{#subobject:
|Label Page=Insulin glulisine |Label Name=Insulin glulisine13.png
}}
{{#subobject:
|Label Page=Insulin glulisine |Label Name=Insulin glulisine14.png
}}
{{#subobject:
|Label Page=Insulin glulisine |Label Name=Insulin glulisine15.png
}}
{{#subobject:
|Label Page=Insulin glulisine |Label Name=Insulin glulisine16.png
}}
{{#subobject:
|Label Page=Insulin glulisine |Label Name=Insulin glulisine17.png
}}