Ritodrine
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | Oral, parenteral |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | ~56% |
| Elimination half-life | 1.7-2.6 hours |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
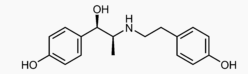
| Formula | C17H21NO3 |
| Molar mass | 287.354 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
|
WikiDoc Resources for Ritodrine |
|
Articles |
|---|
|
Most recent articles on Ritodrine |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Ritodrine at Clinical Trials.gov Clinical Trials on Ritodrine at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Ritodrine
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Ritodrine Discussion groups on Ritodrine Directions to Hospitals Treating Ritodrine Risk calculators and risk factors for Ritodrine
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Ritodrine |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Ritodrine (discontinued preparation: Yutopar) is a tocolytic drug, used to stop premature labor.[1] This drug has been removed from the US market, according to FDA Orange Book. It was available in oral tablets or as an injection and was typically used as the hydrochloride salt, ritodrine hydrochloride.
Mechanism
Ritodrine is a beta-2 adrenergic receptor agonist - a class of medication used for smooth muscle relaxation (other similar drugs are used in asthma or other pulmonary diseases such as salbutamol). Since ritodrine has a bulky N-substituent, it has high β2-selectivity. Also, the 4'-hydroxy on the benzene ring is important for activity as it is needed to form hydrogen bonds. However, the 4'-hydroxy makes it susceptible to metabolism by COMT. Since it is β2-selective it is used for premature labor.[2]
Side effects and potential contraindications
Most side effects of beta-2 agonists result from their concurrent beta-1 activity, and include increase in heart rate, rise in systolic pressure, decrease in diastolic pressure, chest pain secondary to MI, and arrhythmia. Beta agonists may also cause fluid retention secondary to decrease in water clearance, which when added to the tachycardia and increased myocardial work, may result in cardiac failure. In addition, they increase gluconeogenesis in the liver and muscle resulting in hyperglycemia, which increases insulin requirements in diabetic patients. The passage of beta-agonists through the placenta does occur and may be responsible for fetal tachycardia, as well as hypoglycemia or hyperglycemia at birth.
Patients with type 2 diabetes, high blood pressure or migraines should bring this to their doctor's attention before receiving care.
It has also been associated with post-partum hemorrhage.[citation needed]
References
- ↑ Li X, Zhang Y, Shi Z (February 2005). "Ritodrine in the treatment of preterm labour: a meta-analysis" (PDF). The Indian journal of medical research. 121 (2): 120–7. PMID 15756046.
- ↑ Medicinal Chemistry of Adrenergics and Cholinergics
- Pages with script errors
- CS1 maint: Multiple names: authors list
- Template:drugs.com link with non-standard subpage
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Drugs with no legal status
- Drugboxes which contain changes to watched fields
- All articles with unsourced statements
- Articles with unsourced statements from October 2010
- Articles with invalid date parameter in template
- Drug
- Tocolytics
- Phenols
- Phenethylamines