Succinate dehydrogenase
Overview
Succinate-coenzyme Q reductase (EC 1.3.5.1 ; succinate dehydrogenase) is an enzyme complex bound to the inner mitochondrial membrane. It is the only enzyme that participates in both the citric acid cycle and the mitochondrial electron transport chain (in this role it is often called Complex II). It is the only citric acid cycle enzyme that is membrane-bound. It is found in many aerobic and anaerobic organisms including Escherichia coli. The enzyme complex is a heterotetramer divided into three domains: SDHA, the catalytic domain; SDHB, the electron transfer subunit; SDHC/SDHD, the dimeric membrane anchor that contains β -type heme.
Function of the Succinate-coenzyme Q Reductase Complex
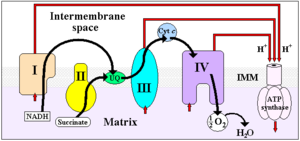
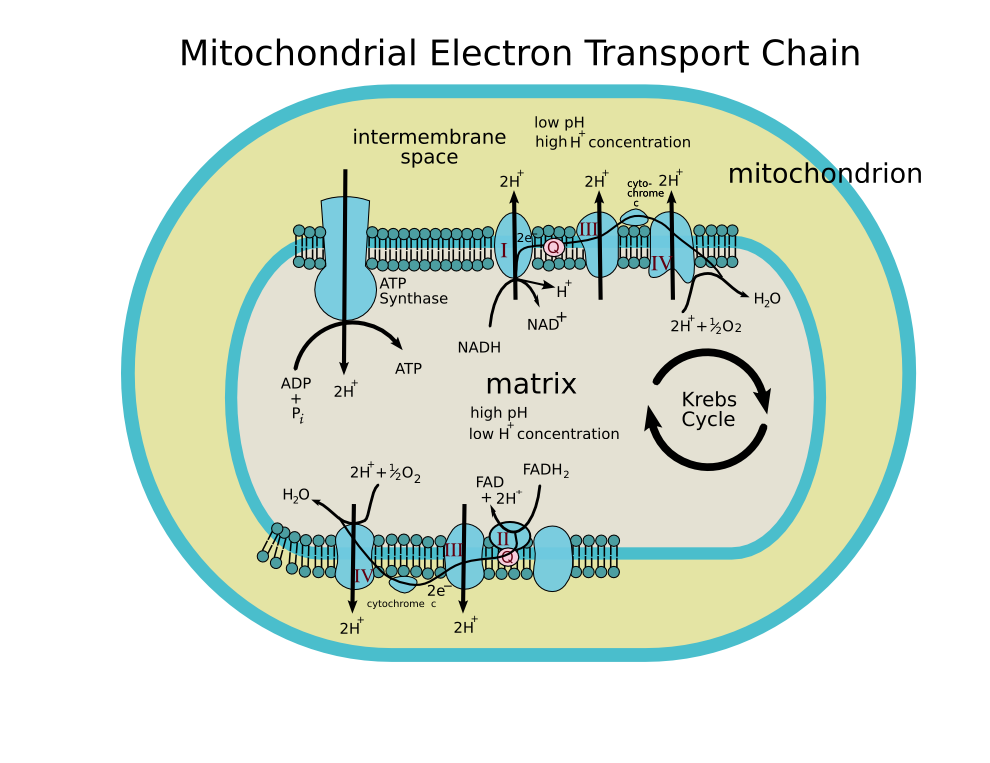
The function of the enzyme is illustrated by following the transfer of electrons from succinate to ubiquinol. The electron path is shown in the diagram by the red arrows.
- Succinate is oxidized to fumarate by the SDHA subunit. SDHA contains a flavin adenine dinucleotide (FAD) cofactor that is covalently linked to a conserved histidine residue. This causes the oxidized FAD to be reduced to FADH2 in a two electron process. This is part of the citric acid cycle.
- The electron transfer subunit (SDHB) contains several iron-sulfur centers which relay electrons from SDHA to the membrane domains: a [2Fe-2S] cluster, a [4Fe-4S] cluster and a [3Fe-4S] cluster.
- These tunnelling electrons are transferred to Coenzyme Q, first to a ubiquinone molecule bound to the SDHC/SDHD dimer, reducing it to ubiquinol (QH2). Quinone reduction is a two-electron process and requires the formation of a ubisemiquinone radical intermediate.
- The resulting ubiquinol molecule is released, free to diffuse through the inner mitochondrial membrane to interact with subsequent enzymes of the mitochondrial respiratory chain (electron transport chain). A heme cofactor is also found within the membrane domain but its function has yet to be discovered.
Role in Disease
The fundamental role of succinate-coenzyme Q reductase in the electron transfer chain of mitochondria makes it vital in most multicellular organisms, removal of this enzyme from the genome has also been shown to be lethal at the embryonic stage in mice.
SDHB, SDHC and SDHD exhibit classic tumor suppressor gene behavior. SDHA has never been reported to be a tumor suppressor. The resulting disease depends on which gene is mutated.
- SDHA mutations can lead to Leigh syndrome.
- SDHB mutations can lead to tumorogenesis in chromaffin cells, causing hereditary paraganglioma and hereditary pheochromocytoma. Tumors tend to be malignant.
- SDHC mutations can lead to hereditary paraganglioma and hereditary pheochromocytoma. Tumors tend to be benign. These mutations are uncommon.
- SDHD mutations can lead to hereditary paraganglioma and hereditary pheochromocytoma. Tumors tend to be benign, and occur often in the head and neck regions.
The precise mechanism for each of these disease pathways is still being determined. See the specific gene for more details. Other diseases linked to succinate dehydrogenase include hereditary paraganglioma, obsessive compulsive disorder, generation of aromatic flatulants, and dwarfism.
Additional images
-
ETC
-
ETC
External links
- Interactive Molecular model of succinate dehydrogenase (Requires MDL Chime)
- Weiner Web page describing research on Complex II Homologs
- UMich Orientation of Proteins in Membranes families/superfamily-3 - Calculated spatial positions of succinate dehydrogenase and related proteins in membranes
- Succinate+dehydrogenase at the US National Library of Medicine Medical Subject Headings (MeSH)
de:Succinat-Dehydrogenase it:Succinato deidrogenasi (ubichinone)