Epithelial sodium channel
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
The epithelial sodium channel (short: ENaC, also: sodium channel non-neuronal 1 (SCNN1) or amiloride sensitive sodium channel (ASSC)) is a membrane-bound ion-channel that is permeable for Li+-ions, protons and especially Na+-ions. It is a constitutively active ion-channel. It is arguably the most selective ion channel.
Structure
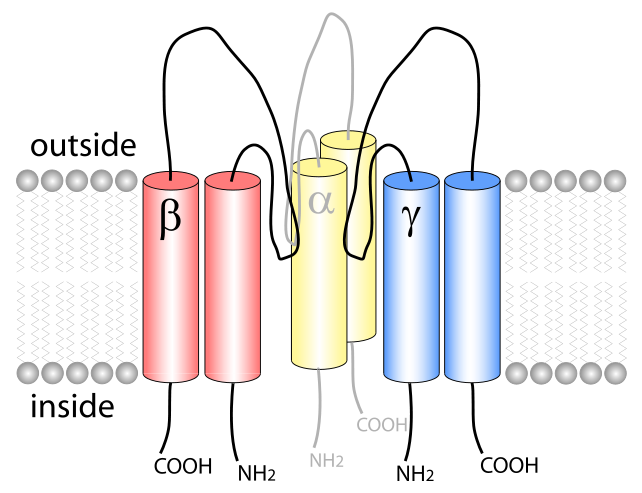
ENaC consists of three different subunits: α, β, γ. The stoichometry of these subunits is still to be verified, but ENaC is very likely to be a heterotrimeric protein like the recently analyzed acid-sensing ion channel 1 (ASIC1), which belongs to the same family.[1] Each of the subunits consists of two transmembrane helices and an extracellular loop. The amino- and carboxy-termini of all polypeptides are located in the cytosol.
Location and function
ENaC is located in the apical membrane of polarized epithelial cells particularly in the kidney, the lung and the colon. It is involved in the transepithelial Na+-ion transport which it accomplishes together with the Na+/K+-ATPase.
It plays a major role in the Na+- and K+-ion homeostasis of blood, epithelia and extraepithelial fluids by resorption of Na+-ions. The activity of ENaC in colon and kidney is modulated by the mineralcorticoid aldosterone. It is blocked by amiloride, which is used in medical science, where amiloride serves as a diuretic.
ENaC can furthermore be found in taste receptor cells, where it plays an important role in salt taste perception. In rodents virtually the entire salt taste is mediated by ENaC, whereas it seems to play a less significant role in humans: about 20 percent can be accredited to the epithelial sodium channel.
δ-subunit
In addition there is a fourth, so-called δ-subunit, that shares considerable sequence similarity with the α-subunit and can form a functional ion-channel together with the β- and γ-subunits. Such δ, β, γ-ENaC appears in pancreas, testes and ovaries. Their function is yet unknown.
Clinical significance
Units β and γ are associated with Liddle's syndrome.[2]
Amiloride and triamterene are potassium-sparing diuretics which act upon the Epithelial sodium channel.
Genes
References
- ↑ Jasti J, Furukawa H, Gonzales EB, Gouaux E (2007). "Structure of acid-sensing ion channel 1 at 1.9 Å resolution and low pH". Nature. 449: 316–322.
- ↑ http://www.neuro.wustl.edu/neuromuscular/mother/chan.html#nachnvg
External links
- Epithelial+sodium+channel at the US National Library of Medicine Medical Subject Headings (MeSH)