Adenocarcinoma of the lung pathophysiology
|
Adenocarcinoma of the Lung Microchapters |
|
Differentiating Adenocarcinoma of the Lung from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Adenocarcinoma of the lung pathophysiology On the Web |
|
American Roentgen Ray Society Images of Adenocarcinoma of the lung pathophysiology |
|
Risk calculators and risk factors for Adenocarcinoma of the lung pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Trusha Tank, M.D.[2], Shanshan Cen, M.D. [3], Sudarshana Datta, MD [4]
Overview
Adenocarcinoma is the most common type of lung cancer found in non-smokers and is usually seen as a peripheral lesion in the lungs. In past several years many genetic and environmental factors hace been identified as a causative factor for lunng cancer. Individual susceptibility, active smoking, radon exposure, exposure to high pollution levels, asbestos exposure, occupational or environmental exposure to particular agents or carcinogens contribute to the development of adenocarcinoma of the lung. Hydrocarbons cause damage to the DNA and form DNA adducts. Benzo-A-pyrine has effects on inducing p53 mutations and affects molecular signaling pathways such as AKT. Genes involved in the pathogenesis of adenocarcinoma of the lung include EGFR, HER2, KRAS, ALK, and BRAF. On gross pathology, peripheral multifocal lesions are characteristic findings in patients with adenocarcinoma of the lung. On microscopic histopathological analysis, nuclear atypia, eccentrically placed nuclei, abundant cytoplasm, and conspicuous nucleoli are characteristic findings of adenocarcinoma of the lung.
Pathogenesis
- Adenocarcinoma is the most common type of lung cancer found in non-smokers and is usually seen as a peripheral lesion in the lungs, as compared to centrally located tumors such as small cell lung cancer and squamous cell lung cancer.[1][2]
- Individual susceptibility, active smoking, radon exposure, exposure to high pollution levels, asbestos exposure, occupational or environmental exposure to particular agents or carcinogens contribute to the development of adenocarcinoma of the lung. Hydrocarbons cause damage to the DNA and form DNA adducts. Benzo-A-pyrine has effects on inducing p53 mutations and affects molecular signaling pathways such as AKT.
- The “multiple hit theory” for adenocarcinoma of the lung states that genetic reproduction is hindered due to the cumulative effect of several toxic insults. Underlying lung disease such as COPD, idiopathic pulmonary fibrosis and tuberculosis may exacerbate also trigger the process.
- Mutations involving several oncogenes may lead to the development of adenocarcinoma of the lung. These are as follows:
- Mutations of ras (found in thirty percent of cases) affect signal transduction by affecting GTPase activity:
- c-myc
- c-raf
- Tumor suppressor genes retinoblastoma (Rb) and p53
- Mutations of APOBEC protein
Genetics
- Genes involved in the pathogenesis of adenocarcinoma of the lung include:[3][4][5][6]
- EGFR (7p11)
- KRAS (12p12)
- BRAF (7q34)
- PIK3CA (3q26)
- ERBB2 (17q12)
- Translocation EML4/ALK
- Tyrosine kinase fusions
- ALK (2p23), ROS1 (6q22), and RET (10q11)
Gross Pathology
- On gross pathology, peripheral multifocal lesions are characteristic findings in patients with adenocarcinoma of the lung.[7]
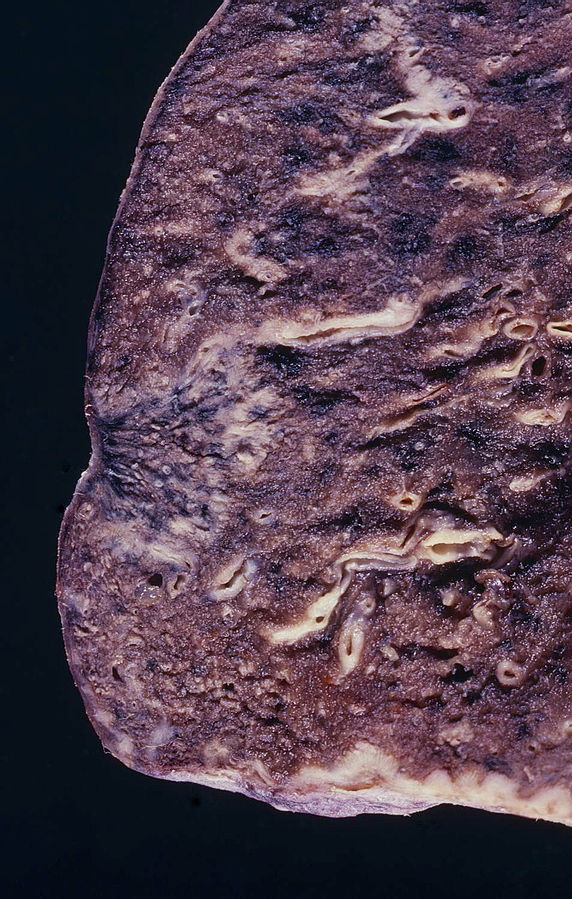
Source: Libre pathology
Microscopic Pathology
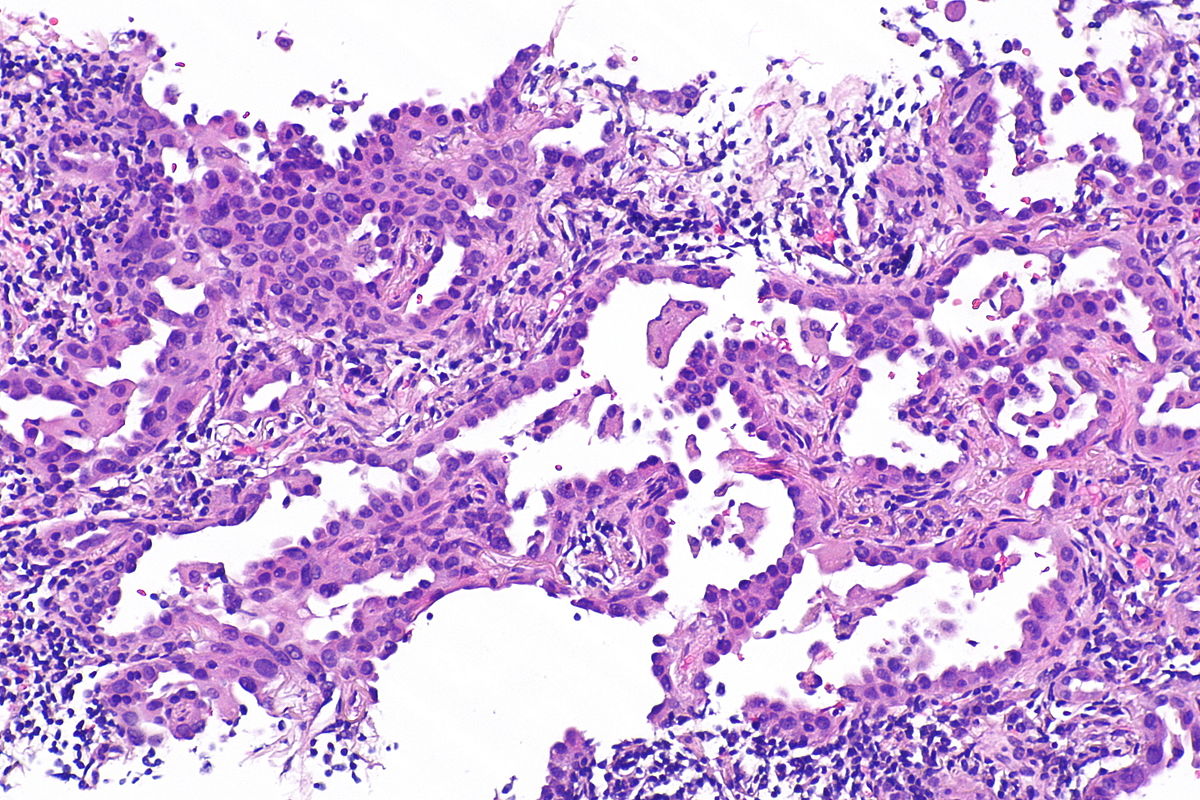
Source: Libre pathology
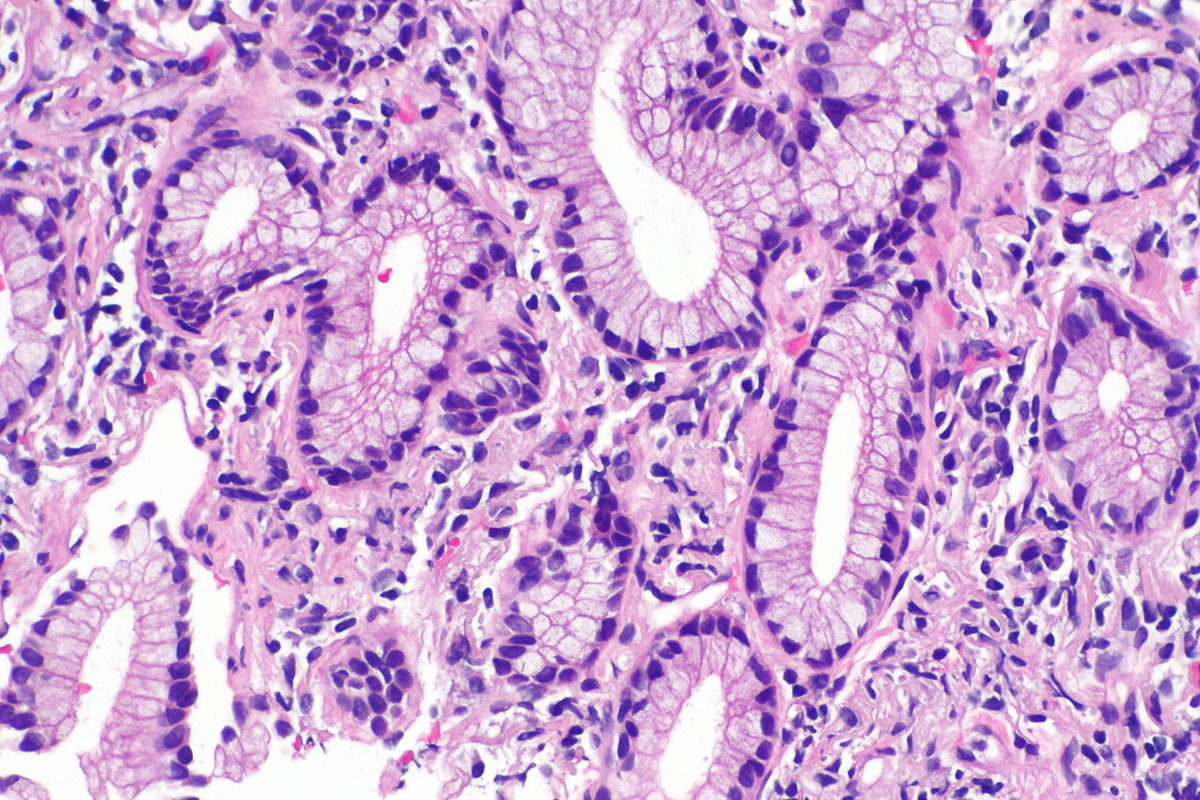
Source: Libre pathology
- On microscopic histopathological analysis, nuclear atypia, eccentrically placed nuclei, abundant cytoplasm, and conspicuous nucleoli are characteristic findings of adenocarcinoma of the lung.
- Atypical adenomatous hyperplasia (AAH): is the precursor of peripheral adenocarcinomas. It consists of well demarcated columnar or cuboidal cells with the following features:[8][9]
- Varying degrees of cytologic atypia
- Hyperchromasia
- Pleomorphism
- Prominent nucleoli
- As adenocarcinoma is a derivative of mucus producing glands in the lungs, it tends to stain mucin positive.
- Based on differentiation, the tumor may be:
- Well differentiated (low grade) : Normal appearance
- Poorly differentiated (high grade): Abnormal glandular appearance with a positive mucin stain
Histological Subtypes
- The IASLC/ATS/ERS lung adenocarcinoma histologic classification system was proposed in the Journal of Thoracic Oncology in 2011.[10]
- According to this new classification, tumor size ≤3 cm with pure lepidic pattern, but without lymphatic, vascular, pleural invasion or tumor necrosis was defined as adenocarcinoma in situ (AIS).
- If tumor size ≤3 cm with a lepidic predominant pattern and contained ≤5 mm stromal invasion it was defined as minimally invasive adenocarcinoma (MIA).
- If tumor had >5 mm stromal invasion it was defined as an invasive adenocarcinoma.
- Histologically adenocarcinoma is divided in to following subtypes:[11][12][13][14][15][16]
- Lepidic adenocarcinoma
- Lepidic growth adenocarcinoma is defined as tumor cells proliferating along the surface of intact alveolar walls without stromal or vascular invasion pathologically.
- Solitary adenocarcinomas with pure lepidic growth, termed “AIS” has 100% disease-specific survival, if the lesion is completely resected.
- Acinar adenocarcinoma:
- Acinar pattern comprises infiltrating round to oval glands lined by tumour cells.
- Irregular-shaped glands.
- Malignant cells: Hyperchromatic nuclei, fibroblastic stroma.
- Sometimes the glandular cells and lumina may contain mucin.
- Papillary adenocarcinoma
- The papillary pattern is composed of glandular tumour cells growing along fibrovascular cores.
- Papillae, necrosis, surrounding invasion, cuboidal to columnar epithelial lining, mucinous or non-mucinous.
- Lung adenocarcinomas with papillary growth show 2 types of papillary architecture:
- True papillary type: Papillae containing a layered glandular epithelium surrounded by fibrovascular core.
- Micropapillary type: The papillary tufts lack a central fibrovascular core and extensively shed within alveolar spaces.
- Micropapillary adenocarcinoma:
- The papillary tufts lack a central fibrovascular core and extensively shed within alveolar spaces.
- Micropapillary growth has been associated with an aggressive clinical course compared with traditional papillary adenocarcinoma.
- Micropapillary adenocarcinoma (MPA) may be often diagnosed at a high stage in nonsmokers, with intralobar satellites.
- Micropapillary adenocarcinoma frequently metastasizes to the contralateral lung, mediastinal lymph nodes, bone, and adrenal glands, with high mortality.
- Solid adenocarcinoma
- Cohesive cell cluster in a nest-like pattern without acinar polarity are the hallmark of the solid growth pattern.
- Solid adenocarcinoma consists of sheets of tumor cells with abundant cytoplasm and mostly vesicular nuclei with several conspicuous nucleoli.
- No acinar, papillary, or lepidic patterns are seen and there was no suggestion of mucin in tumor cell cytoplasm
- Invasive mucinous adenocarcinoma
- Mixed invasive mucinous: Invasive mucinous adenocarcinoma demonstrates areas with lepidic, acinar, and papillary patterns.
- Fibrotic focus that contains invasive tumor with a desmoplastic stroma.
- The tumor consists of columnar cells filled with abundant mucin in the apical cytoplasm and shows small, basally oriented nuclei.
- Nonmucinous adenocarcinoma
- Mixed invasive mucinous: Invasive mucinous adenocarcinoma demonstrates areas with lepidic, acinar, and papillary patterns.
- Colloid adenocarcinoma:
- This tumor consists of abundant pools of mucin growing within and distending airspaces.
- Well differentiated mucinous glandular epithelium along the surface of fibrous septa and within the pools of mucin.
- Tumor cells may be very inconspicuous.
- The surface of the fibrous wall may be lined by well-differentiated cuboidal or columnar mucinous epithelium.
- Fetal adenocarcinoma:
- Fetal adenocarcinoma consists of malignant glandular cells growing in tubules and papillary structures with endometrioid morphology.
- Some tumor cells have prominent clear cytoplasm, and squamoid morules are present
- Enteric adenocarcinoma:
- Consists of an adenocarcinoma that morphologically resembles colonic adenocarcinoma with back-to-back angulated acinar structures.
- The tumor cells are cuboidal to columnar with nuclear pseudostratification.
- The tumor stains strongly for CDX-2.
- Minimally invasive adenocarcinoma (MIA)
- Nonmucinous (MIA):
- This subpleural adenocarcinoma tumor consists primarily of lepidic growth with a small (0.5 cm) central area of invasion.
- It may present as the lepidic pattern and/or acinar invasion.
- Mucinous (MIA):
- Mucinous MIA consists of a tumor showing lepidic growth and a small (0.5 cm) area of invasion.
- The tumor cells consist of mucinous columnar cells and pale cytoplasm resembling goblet cells growing mostly in a lepidic pattern along the surface of alveolar walls.
- The tumor invades the areas of stromal fibrosis in an acinar pattern.
- Low grade differentiation.
- Nonmucinous (MIA):
- Preinvasive lesions
- Atypical adenomatous hyperplasia (AAH): Consists of atypical pneumocytes proliferating along alveolar walls.
- Non invasive.
- The slightly atypical pneumocytes are cuboidal and show gaps between the cells.
- Nuclei are hyperchromatic and may present with nuclear enlargement and multinucleation.
- Adenocarcinoma in situ (AIS)
- Nonmucinous (AIS): Tumor grows purely with a lepidic pattern.
- No foci of invasion or scarring is seen.
- It shows atypical pneumocytes proliferating along the thickened, but preserved, alveolar walls.
- Mucinous AIS: Consists of a nodular proliferation of mucinous columnar cells growing in a purely lepidic pattern.
- Although there is a small central scar, no stromal or vascular invasion is seen.
- The tumor cells consist of cuboidal to columnar cells with abundant apical mucin and small, basally oriented nuclei.
- Nonmucinous (AIS): Tumor grows purely with a lepidic pattern.
- Atypical adenomatous hyperplasia (AAH): Consists of atypical pneumocytes proliferating along alveolar walls.
- Lepidic adenocarcinoma
References
- ↑ Travis WD, Travis LB, Devesa SS (January 1995). "Lung cancer". Cancer. 75 (1 Suppl): 191–202. doi:10.1002/1097-0142(19950101)75:1+<191::AID-CNCR2820751307>3.0.CO;2-Y. PMID 8000996.
- ↑ Mitchell, Richard Sheppard; Kumar, Vinay; Abbas, Abul K.; Fausto, Nelson. "Chapter 13, box on morphology of adenocarcinoma". Robbins Basic Pathology (8th ed.). Philadelphia: Saunders. ISBN 1-4160-2973-7.
- ↑ Stewart, Bernard (2014). World cancer report 2014. Lyon, France Geneva, Switzerland: International Agency for Research on Cancer,Distributed by WHO Press, World Health Organization. ISBN 9283204298.
- ↑ Soda M, Choi YL, Enomoto M, Takada S, Yamashita Y, Ishikawa S; et al. (2007). "Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer". Nature. 448 (7153): 561–6. doi:10.1038/nature05945. PMID 17625570.
- ↑ Davies KD, Le AT, Theodoro MF, Skokan MC, Aisner DL, Berge EM; et al. (2012). "Identifying and targeting ROS1 gene fusions in non-small cell lung cancer". Clin Cancer Res. 18 (17): 4570–9. doi:10.1158/1078-0432.CCR-12-0550. PMC 3703205. PMID 22919003.
- ↑ Stewart, Bernard (2014). World cancer report 2014. Lyon, France Geneva, Switzerland: International Agency for Research on Cancer,Distributed by WHO Press, World Health Organization. ISBN 9283204298.
- ↑ Adenocarcinoma of the lung. Librepathology 2015. http://librepathology.org/wiki/index.php/File:Adenocarcinoma_%283950819000%29.jpg
- ↑ Kumar, Vinay (2007). Robbins basic pathology. Philadelphia, PA: Saunders/Elsevier. ISBN 1416029737.
- ↑ Stewart, Bernard (2014). World cancer report 2014. Lyon, France Geneva, Switzerland: International Agency for Research on Cancer,Distributed by WHO Press, World Health Organization. ISBN 9283204298.
- ↑ . doi:10.3978/j.issn.2072-1439.2014.09.13. Missing or empty
|title=(help) - ↑ Travis, William (2004). Pathology and genetics of tumours of the lung, pleura, thymus, and heart. Lyon: IARC Press. ISBN 9283224183.
- ↑ "www.jto.org".
- ↑ Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger K, Yatabe Y, Ishikawa Y, Wistuba I, Flieder DB, Franklin W, Gazdar A, Hasleton PS, Henderson DW, Kerr KM, Nakatani Y, Petersen I, Roggli V, Thunnissen E, Tsao M (May 2013). "Diagnosis of lung adenocarcinoma in resected specimens: implications of the 2011 International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society classification". Arch. Pathol. Lab. Med. 137 (5): 685–705. doi:10.5858/arpa.2012-0264-RA. PMID 22913371.
- ↑ Iwata H (September 2016). "Adenocarcinoma containing lepidic growth". J Thorac Dis. 8 (9): E1050–E1052. doi:10.21037/jtd.2016.08.78. PMID 27747060.
- ↑ Jones KD (December 2013). "Whence lepidic?: the history of a Canadian neologism". Arch. Pathol. Lab. Med. 137 (12): 1822–4. doi:10.5858/arpa.2013-0144-HP. PMID 23937575.
- ↑ Lin, Gengpeng; Xie, Canmao (2017). "PUB070 Acinar-Predominant Pattern Correlates with Poorer Outcome in Invasive Mucinous Adenocarcinoma of the Lung". Journal of Thoracic Oncology. 12 (1): S1489. doi:10.1016/j.jtho.2016.11.2040. ISSN 1556-0864.