Parvovirus B19
| Parvovirus B19 | ||||||||
|---|---|---|---|---|---|---|---|---|
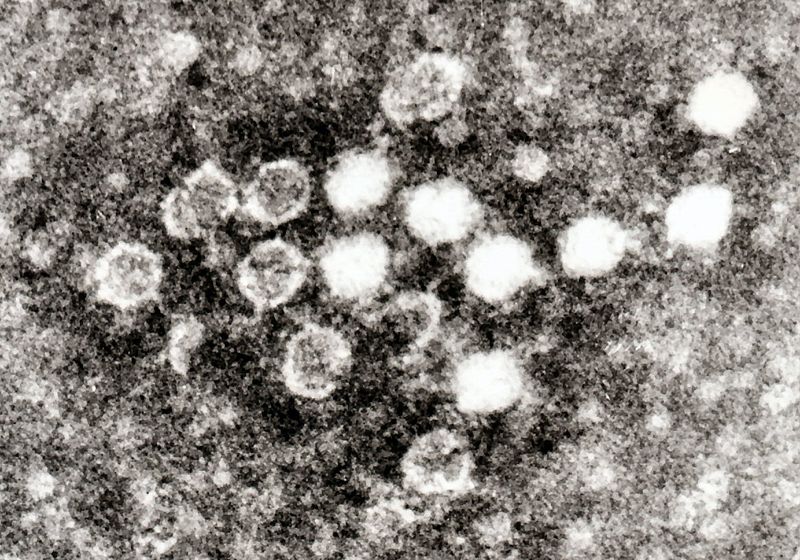 Electron micrograph of Parvoviruses in blood
| ||||||||
| Virus classification | ||||||||
|
|
Fifth disease Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Parvovirus B19 On the Web |
|
American Roentgen Ray Society Images of Parvovirus B19 |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Parvovirus B19 (B19 virus) was the first (and, until 2005, only) human parvovirus to be discovered, by chance in 1975 by the Australian virologist Yvonne Cossart.[1] It gained its name because it was discovered in well B19 of a large series of petri dishes apparently numbered in this way.[2]

Parvovirus B19 is best known for causing a childhood exanthem called fifth disease or erythema infectiosum.[3]
Virology
The B19 virus belongs to the Parvoviridae family of small DNA viruses.[4] It is classified as Erythrovirus because of its capability to invade red blood cell precursors in the bone marrow.
Transmission
The virus is primarily spread by infected respiratory droplets; blood-borne transmission, however, has been reported.[5] The secondary attack risk for exposed household persons is about 50%, and about half of that for classroom contacts.[6]
Infectivity
B19 symptoms begins some six days after exposure and last about a week. Infected patients with normal immune systems are contagious before becoming symptomatic, but probably not after then.[7] Individuals with B19 IgG antibodies are generally considered immune to recurrent infection, but reinfection is possible in a minority of cases.[8] About half of adults are B19-immune due to a past infection.
Epidemiology
A significant increase in the number of cases is seen every three to four years; the last epidemic year was 1998. Outbreaks can arise especially in nurseries and schools.
Parvovirus B19 causes an infection in humans only; cat and dog parvoviruses do not infect humans. In contrast with small animals, there is no vaccine available for human parvovirus B19.
Role in disease
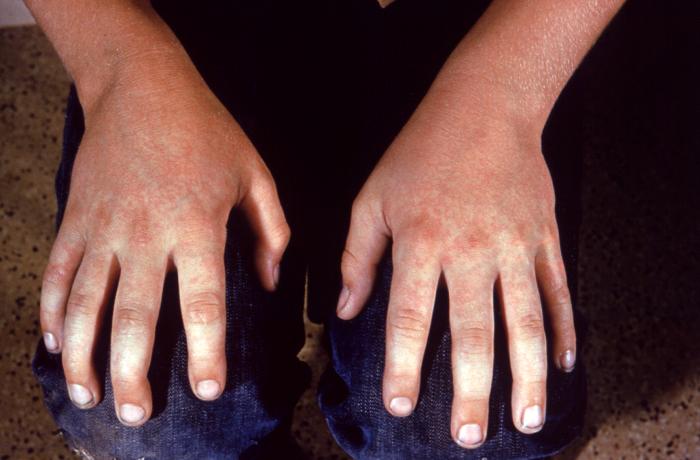
Fifth disease
Fifth disease or erythema infectiosium is only one of several expressions of Parvovirus B19. Any age may be affected, although it is most common in children aged six to ten years.
After being infected, patients usually develop the illness after an incubation period of four to fourteen days. The disease commences with fever and malaise while the virus is most abundant in the bloodstream, and patients are usually no longer infectious once the characteristic rash of this disease has appeared.
Teenagers or young adults tend to develop the so called 'Papular Pupuric Gloves and Socks Syndrome.' Unlike young children, these patients may be infectious with this rash.
Rash
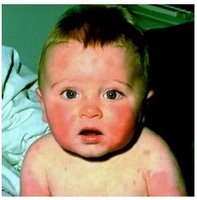
The rash of fifth disease is typically described as "slapped cheeks," with erythema across the cheeks and sparing the nasolabial folds, forehead, and mouth. Because of this rash, fifth disease is sometimes called slapped cheek syndrome. Fifth disease is also known for "lace-like" rashes on the arms, legs, torso, and back. These rashes can last for up to 5 weeks and are worse after sun exposure, exercise, or hot baths.
Arthritis
In adults (and perhaps some children), parvovirus B19 can lead to a seronegative arthritis which is usually easily controlled with analgesics. Women are approximately twice as likely as men to experience arthritis after parvo virus infection. Possibly up to 15% of all new cases of arthritis are due to parvovirus, and a history of recent contact with a patient and positive serology generally confirms the diagnosis.[7] This arthritis does not progress to other forms of arthritis. Typically joint symptoms last 1-3 weeks, but in 10-20% of those affected, it may last weeks to months.
Aplastic crisis
Although most patients have an arrest of erythropoiesis (production of red blood cells) during parvovirus infection, it causes worse problems in patients with sickle cell anemia, or with hereditary spherocytosis, who are heavily dependent on erythropoeisis due to the reduced lifespan of the red cells. This is termed "aplastic crisis" (also called reticulocytopenia). It is treated with blood transfusion. Sickle-cell patients will probably be the first candidates for a parvovirus B19 vaccine when it is developed.
Hydrops fetalis
Parvovirus infection in pregnant women is associated with hydrops fetalis due to severe fetal anemia, sometimes leading to miscarriage or stillbirth. The risk of fetal loss is about 10% if infection occurs before pregnancy week 20 (esp. between weeks 14-20), but minimal after then. Routine screening of the antinatal sample would enable the pregnant mother to determine the risk of infection. Knowledge of her status would allow the mother to avoid the risk of infection.The risk to the fetus will be reduced with correct diagnosis of the anemia (by ultrasound scans) and treatment (by blood transfusions). Once the baby is born, there is evidence to suggest that no developmental abnormalities are due to B19 infection during pregnancy.
Differentiating Parvovirus B19 Rash from other diseases
Different rash-like conditions can be confused with Parvovirus B19 and are thus included in its differential diagnosis. The various conditions that should be differentiated from Parvovirus B19 include:[9][10][11][12][13][14][15]
| Disease | Features |
|---|---|
| Impetigo | |
| Insect bites |
|
| Kawasaki disease |
|
| Measles |
|
| Monkeypox |
|
| Rubella |
|
| Atypical measles |
|
| Coxsackievirus |
|
| Acne |
|
| Syphilis | It commonly presents with gneralized systemic symptoms such as malaise, fatigue, headache and fever. Skin eruptions may be subtle and asymptomatic It is classically described as:
|
| Molluscum contagiosum |
|
| Mononucleosis |
|
| Toxic erythema | |
| Rat-bite fever | |
| Parvovirus B19 | |
| Cytomegalovirus |
|
| Scarlet fever |
|
| Rocky Mountain spotted fever |
|
| Stevens-Johnson syndrome |
|
| Varicella-zoster virus | |
| Chickenpox |
|
| Meningococcemia | |
| Rickettsial pox | |
| Meningitis |
|
References
- ↑ Heegaard ED, Brown KE (2002). "Human parvovirus B19". Clin Microbiol Rev. 15 (3): 485–505. PMID 12097253.
- ↑ Cossart YE, Field AM, Cant B, Widdows D (1975). "Parvovirus-like particles in human sera". Lancet. 1 (7898): 72–3. PMID 46024.
- ↑ Vafaie J, Schwartz RA (2004). "Parvovirus B19 infections". Int J Dermatol. 43 (10): 747–9. PMID 15485533.
- ↑ Brown KE (2004). "Variants of B19". Dev Biol (Basel). 118: 71–7. PMID 15645675.
- ↑ Pattison JR, Patou G (1996). Parvoviruses. In: Barron's Medical Microbiology (Barron S et al, eds.) (4th ed. ed.). Univ of Texas Medical Branch. ISBN 0-9631172-1-1.
- ↑ Young NS, Brown KE (2004). "Parvovirus B19". N Engl J Med. 350 (6): 586–97. PMID 14762186.
- ↑ 7.0 7.1 Corcoran A, Doyle S (2004). "Advances in the biology, diagnosis and host-pathogen interactions of parvovirus B19". J Med Microbiol. 53 (Pt 6): 459–75. PMID 15150324.
- ↑ Lehmann HW, von Landenberg P, Modrow S (2003). "Parvovirus B19 infection and autoimmune disease". Autoimmun Rev. 2 (4): 218–23. PMID 12848949.
- ↑ Hartman-Adams H, Banvard C, Juckett G (2014). "Impetigo: diagnosis and treatment". Am Fam Physician. 90 (4): 229–35. PMID 25250996.
- ↑ Mehta N, Chen KK, Kroumpouzos G (2016). "Skin disease in pregnancy: The approach of the obstetric medicine physician". Clin Dermatol. 34 (3): 320–6. doi:10.1016/j.clindermatol.2016.02.003. PMID 27265069.
- ↑ Moore, Zack S; Seward, Jane F; Lane, J Michael (2006). "Smallpox". The Lancet. 367 (9508): 425–435. doi:10.1016/S0140-6736(06)68143-9. ISSN 0140-6736.
- ↑ Ibrahim F, Khan T, Pujalte GG (2015). "Bacterial Skin Infections". Prim Care. 42 (4): 485–99. doi:10.1016/j.pop.2015.08.001. PMID 26612370.
- ↑ Ramoni S, Boneschi V, Cusini M (2016). "Syphilis as "the great imitator": a case of impetiginoid syphiloderm". Int J Dermatol. 55 (3): e162–3. doi:10.1111/ijd.13072. PMID 26566601.
- ↑ Kimura U, Yokoyama K, Hiruma M, Kano R, Takamori K, Suga Y (2015). "Tinea faciei caused by Trichophyton mentagrophytes (molecular type Arthroderma benhamiae ) mimics impetigo : a case report and literature review of cases in Japan". Med Mycol J. 56 (1): E1–5. doi:10.3314/mmj.56.E1. PMID 25855021.
- ↑ CEDEF (2012). "[Item 87--Mucocutaneous bacterial infections]". Ann Dermatol Venereol. 139 (11 Suppl): A32–9. doi:10.1016/j.annder.2012.01.002. PMID 23176858.