Riluzole
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Riluzole is a Glutamate antagonist that is FDA approved for the treatment of amyotrophic lateral sclerosis (ALS). Common adverse reactions include headache, abdominal pain, back pain, vomiting, dyspepsia, diarrhea, dizziness.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Riluzole tablets, USP are indicated for the treatment of patients with amyotrophic lateral sclerosis (ALS).
- Riluzole extends survival and/or time to tracheostomy.
Dosage
- The recommended dose for riluzole tablets is 50 mg every 12 hours. No increased benefit can be expected from higher daily doses, but adverse events are increased.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Riluzole in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Riluzole in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Riluzole FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Riluzole in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Riluzole in pediatric patients.
Contraindications
- Riluzole tablets are contraindicated in patients who have a history of severe hypersensitivity reactions to riluzole or any of the tablet components.
Warnings
Liver Injury / Monitoring Liver Chemistries
- Riluzole tablets should be prescribed with care in patients with current evidence or history of abnormal liver function indicated by significant abnormalities in serum transaminase (ALT/SGPT; AST/SGOT), bilirubin, and/or gamma-glutamate transferase (GGT) levels. Baseline elevations of several LFTs (especially elevated bilirubin) should preclude the use of riluzole.
- Riluzole, even in patients without a prior history of liver disease, causes serum aminotransferase elevations. Treatment should be discontinued if ALT levels are ≥ 5 X ULN or if clinical jaundice develops.
- Experience in almost 800 ALS patients indicates that about 50% of riluzole-treated patients will experience at least one ALT/SGPT level above the upper limit of normal, about 8% will have elevations > 3 × ULN, and about 2% of patients will have elevations > 5 × ULN. A single non-ALS patient with epilepsy treated with concomitant carbamazepine and phenobarbital experienced marked, rapid elevations of liver enzymes with jaundice (ALT 26 × ULN, AST 17 × ULN, and bilirubin 11 × ULN) four months after starting riluzole; these returned to normal 7 weeks after treatment discontinuation.
- Maximum increases in serum ALT usually occurred within 3 months after the start of riluzole therapy and were usually transient when < 5 times ULN. In trials, if ALT levels were < 5 times ULN, treatment continued and ALT levels usually returned to below 2 times ULN within 2 to 6 months. Treatment in studies was discontinued, however, if ALT levels exceeded 5 × ULN, so that there is no experience with continued treatment of ALS patients once ALT values exceed 5 times ULN. There were rare instances of jaundice. There is limited experience with rechallenge of patients who have had riluzole discontinued for ALT > 5 X ULN, but there is the possibility of increased ALT values reoccurring: Laboratory Tests). Therefore, rechallenge is not recommended.
- In postmarketing experience, cases of clinical hepatitis associated with riluzole have been reported, including with fatal outcome.
Neutropenia
- Among approximately 4000 patients given riluzole for ALS, there were three cases of marked neutropenia (absolute neutrophil count less than 500/mm3), all seen within the first 2 months of riluzole treatment. In one case, neutrophil counts rose on continued treatment. In a second case, counts rose after therapy was stopped. A third case was more complex, with marked anemia as well as neutropenia and the etiology of both is uncertain. Patients should be warned to report any febrile illness to their physicians. The report of a febrile illness should prompt treating physicians to check white blood cell counts.
Interstitial Lung Disease
- Cases of interstitial lung disease have been reported in patients treated with riluzole, some of them severe; upon further investigation, many of these cases were hypersensitivity pneumonitis. If respiratory symptoms develop such as dry cough and/or dyspnea, chest radiography should be performed, and in case of findings suggestive of interstitial lung disease or hypersensitivity pneumonitis (e.g., bilateral diffuse lung opacities), riluzole should be discontinued immediately. In the majority of the reported cases, symptoms resolved after drug discontinuation and symptomatic treatment.
PRECAUTIONS
Use in Patients with Concomitant Disease
- Riluzole should be used with caution in patients with concomitant liver insufficiency. In particular, in cases of riluzole-induced hepatic injury manifested by elevated liver enzymes, the effect of the hepatic injury on riluzole metabolism is unknown.
Special Populations
- Riluzole should be used with caution in elderly patients whose hepatic function may be compromised due to age. Also, female patients may possess a lower metabolic capacity to eliminate riluzole compared to males.
Information for the Patients
- Patients should be advised to report any febrile illness to their physicians: Neutropenia.
- Patients should be advised to report any cough or difficulties in breathing to their physicians: Interstitial Lung Disease.
- Patients and caregivers should be advised that riluzole tablets should be taken on a regular basis and at the same time of the day (e.g., in the morning and evening) each day. If a dose is missed, take the next tablet as originally planned.
- Patients should be warned about the potential for dizziness, vertigo, or somnolence and advised not to drive or operate machinery until they have gained sufficient experience on riluzole to gauge whether or not it affects their mental and/or motor performance adversely.
- Whether alcohol increases the risk of serious hepatotoxicity with riluzole is unknown; therefore, patients being treated with riluzole should be discouraged from drinking excessive amounts of alcohol.
- Patients should also be made aware that riluzole tablets should be stored at temperatures 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F) and protected from bright light.
- Riluzole tablets must be kept out of the reach of children.
Adverse Reactions
Clinical Trials Experience
- The most commonly observed AEs associated with the use of riluzole more frequently than placebo treated patients were: asthenia, nausea, dizziness, decreased lung function, diarrhea, abdominal pain, pneumonia, vomiting, vertigo, circumoral paresthesia, anorexia, and somnolence. Asthenia, nausea, dizziness, diarrhea, anorexia, vertigo, somnolence, and circumoral paresthesia were dose related.
- Approximately 14% (n = 141) of the 982 individuals with ALS who received riluzole in pre-marketing clinical trials discontinued treatment because of an adverse experience. Of those patients who discontinued due to adverse events, the most commonly reported were: nausea, abdominal pain, constipation, and ALT elevations. In a dose response study in ALS patients, the rates of discontinuation of riluzole for asthenia, nausea, abdominal pain, and ALT elevation were dose related.
Incidence in Controlled ALS Clinical Studies
- Table 1 lists treatment-emergent signs and symptoms that occurred in at least 2% of patients with ALS treated with riluzole (n=794) participating in placebo-controlled trials and were numerically greater in the patients treated with riluzole 100 mg/day than with placebo or for which a dose response relationship is suggested.
- The prescriber should be aware that these figures cannot be used to predict the frequency of adverse experiences in the course of usual medical practice where patient characteristics and other factors may differ from those prevailing during clinical studies. Inspection of these frequencies, however, does provide the prescriber with one basis to estimate the relative contribution of drug and non-drug factors to the AE incidences in the population studied.

Other Adverse Events Observed
- Other events which occurred in more than 2% of patients treated with riluzole 100 mg/day but equally or more frequently in the placebo group included: accidental injury, apnea, bronchitis, constipation, death, dysphagia, dyspnea, flu syndrome, heart arrest, increased sputum, pneumonia, and respiratory disorder.
- The overall adverse event profile for riluzole was similar between females and males, and was independent of age. Because the largest non-white racial subgroup was only 2% of patients exposed to riluzole (18/794) in placebo-controlled trials, there are insufficient data to support a statement regarding the distribution of adverse experience reports by race. In ALS studies, dizziness did occur more commonly in females (11%) than in males (4%). There was not a difference between females and males in the rates of discontinuation of riluzole for individual adverse experiences.
Other Adverse Events Observed During All Clinical Trials
- Riluzole has been administered to 1713 individuals during all clinical trials, some of which were placebo-controlled. During these trials, all adverse events were recorded by the clinical investigators using terminology of their own choosing. To provide a meaningful estimate of the proportion of individuals having adverse events, similar types of events were grouped into a smaller number of standardized categories using modified COSTART dictionary terminology. The frequencies presented represent the proportion of the 1713 individuals exposed to riluzole who experienced an event of the type cited on at least one occasion while receiving riluzole. All reported events are included except those already listed in the previous table, those too general to be informative, and those not reasonably associated with the use of the drug.
- Events are further classified within body system categories and enumerated in order of decreasing frequency using the following definitions: frequent adverse events are defined as those occurring in at least 1/100 patients; infrequent adverse events are those occurring in 1/100 to 1/1000 patients; rare adverse events are those occurring in fewer than 1/1000 patients.
Body as a Whole: Frequent: Hostility. Infrequent: Abscess, sepsis, photosensitivity reaction, cellulitis, face edema, hernia, peritonitis, attempted suicide, injection site reaction, chills, flu syndrome, intentional injury, enlarged abdomen, neoplasm. Rare: Acrodynia, hypothermia, moniliasis, rheumatoid arthritis.
Digestive System: Infrequent: Increased appetite, intestinal obstruction, fecal impaction, gastrointestinal hemorrhage, gastrointestinal ulceration, gastritis, fecal incontinence, jaundice, hepatitis, glossitis, gum hemorrhage, pancreatitis, tenesmus, esophageal stenosis. Rare: Cheilitis, cholecystitis, hematemesis, melena, biliary pain, proctitis, pseudomembranous enterocolitis, enlarged salivary gland, tongue discoloration, tooth caries.
Immune System Disorders: Infrequent: Anaphylactoid reaction and anaphylaxis.
Nervous System: Frequent: Agitation, tremor. Infrequent: Hallucinations, personality disorder, abnormal thinking, coma, paranoid reaction, manic reaction, ataxia, extrapyramidal syndrome, hypokinesia, urinary retention, emotional lability, delusions, apathy, hypesthesia, incoordination, confusion, convulsion, leg cramps, amnesia, dysarthria, increased libido, stupor, subdural hematoma, abnormal gait, delirium, depersonalization, facial paralysis, hemiplegia, decreased libido, myoclonus. Rare: Abnormal dreams, acute brain syndrome, CNS depression, dementia, cerebral embolism, euphoria, hypotonia, ileus, peripheral neuritis, psychosis, psychotic depression, schizophrenic reaction, trismus, wristdrop.
Skin and Appendages: Infrequent: Skin ulceration, urticaria, psoriasis, seborrhea1, skin disorder, fungal dermatitis. Rare: Angioedema, contact dermatitis, erythema multiforme, furunculosis, skin moniliasis, skin granuloma, skin nodule.
Respiratory System: Infrequent: Hiccup, pleural disorder, asthma, epistaxis, hemoptysis, yawn, hyperventilation, lung edema, hypoventilation, lung carcinoma, hypoxia, laryngitis, pleural effusion, pneumothorax, respiratory moniliasis, stridor, interstitial lung disease, hypersensitivity pneumonitis.
Cardiovascular System: Infrequent: Syncope, hypotension, heart failure, migraine, peripheral vascular disease, angina pectoris, myocardial infarction, ventricular extrasystoles, cerebral hemorrhage, atrial fibrillation, bundle branch block, congestive heart failure, pericarditis, lower extremity embolus, myocardial ischemia, shock. Rare: Bradycardia, cerebral ischemia, hemorrhage, mesenteric artery occlusion, subarachnoid hemorrhage, supraventricular tachycardia, thrombosis, ventricular fibrillation, ventricular tachycardia.
Metabolic and Nutritional Disorders: Infrequent: Gout, respiratory acidosis, edema, thirst, hypokalemia, hyponatremia, weight gain. Rare: Generalized edema, hypercalcemia, hypercholesteremia.
Endocrine System: Infrequent: Diabetes mellitus, thyroid neoplasia. Rare: Diabetes insipidus, parathyroid disorder.
Hemic and Lymphatic System: Infrequent: Anemia, leukocytosis, leukopenia, ecchymosis. Rare: Neutropenia, aplastic anemia, cyanosis, hypochromic anemia, iron deficiency anemia, lymphadenopathy, petechiae1, purpura.
Musculoskeletal System: Infrequent: Arthrosis, myasthenia1, bone neoplasm. Rare: Bone necrosis, osteoporosis, tetany.
Special Senses: Infrequent: Amblyopia, ophthalmitis. Rare: Blepharitis, cataract, deafness, diplopia, ear pain, glaucoma, hyperacusis, photophobia, taste loss, vestibular disorder.
Urogenital System: Infrequent: Urinary urgency, urine abnormality, urinary incontinence, kidney calculus, hematuria, impotence, prostate carcinoma, kidney pain, metrorrhagia, priapism. Rare: Amenorrhea, breast abscess, breast pain, nephritis, nocturia, pyelonephritis, enlarged uterine fibroids, uterine hemorrhage, vaginal moniliasis.
Postmarketing Experience
There is limited information regarding Riluzole Postmarketing Experience in the drug label.
Drug Interactions
- There have been no clinical studies designed to evaluate the interaction of riluzole with other drugs.
- As with all drugs, the potential for interaction by a variety of mechanisms is a possibility.
Hepatotoxic Drugs The clinical trials in ALS excluded patients on concomitant medications which were potentially hepatotoxic, (e.g., allopurinol, methyldopa, sulfasalazine). Accordingly, there is no information about the safety of administering riluzole in conjunction with such medications. If the practitioner chooses to prescribe such a combination, caution should be exercised.
Drugs Highly Bound To Plasma Proteins Riluzole is highly bound (96%) to plasma proteins, binding mainly to serum albumin and to lipoproteins. The effect of riluzole (up to 5 mcg/mL) on warfarin (5 mcg/mL) binding did not show any displacement of warfarin. Conversely, riluzole binding was unaffected by the addition of warfarin, digoxin, imipramine and quinine at high therapeutic concentrations.
Effect of Other Drugs On Riluzole Metabolism In vitro studies using human liver microsomal preparations suggest that CYP 1A2 is the principal isozyme involved in the initial oxidative metabolism of riluzole and, therefore, potential interactions may occur when riluzole is given concurrently with agents that affect CYP 1A2 activity. Potential inhibitors of CYP 1A2 (e.g., caffeine, phenacetin, theophylline, amitriptyline, and quinolones) could decrease the rate of riluzole elimination, while inducers of CYP 1A2 (e.g., cigarette smoke, charcoal-broiled food, rifampicin, and omeprazole) could increase the rate of riluzole elimination.
Effect of Riluzole On the Metabolism of Other Drugs CYP 1A2 is the principal isoenzyme involved in the initial oxidative metabolism of riluzole; potential interactions may occur when riluzole is given concurrently with other agents which are also metabolized primarily by CYP 1A2 (e.g., theophylline, caffeine, and tacrine). Currently, it is not known whether riluzole has any potential for enzyme induction in humans.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Pregnancy category C
- Oral administration of riluzole to pregnant animals during the period of organogenesis caused embryotoxicity in rats and rabbits at doses of 27 mg/kg and 60 mg/kg, respectively, or 2.6 and 11.5 times, respectively, the recommended maximum human daily dose on a mg/m2 basis. Evidence of maternal toxicity was also observed at these doses.
- When administered to rats prior to and during mating (males and females) and throughout gestation and lactation (females), riluzole produced adverse effects on pregnancy (decreased implantations, increased intrauterine death) and offspring viability and growth at an oral dose of 15 mg/kg or 1.5 times the maximum daily dose on a mg/m2 basis.
- There are no adequate and well-controlled studies in pregnant women. Riluzole should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Riluzole in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Riluzole during labor and delivery.
Nursing Mothers
- In rat studies, 14C-riluzole was detected in maternal milk. It is not known whether riluzole is excreted in human breast milk. Because many drugs are excreted in human milk, and because the potential for serious adverse reactions in nursing infants from riluzole is unknown, women should be advised not to breast-feed during treatment with riluzole.
Pediatric Use
- The safety and the effectiveness of riluzole in pediatric patients have not been established.
Geriatic Use
- Age-related compromised renal and hepatic function may cause a decrease in clearance of riluzole. In controlled clinical trials, about 30% of patients were over 65. There were no differences in adverse effects between younger and older patients.
Gender
There is no FDA guidance on the use of Riluzole with respect to specific gender populations.
Race
There is no FDA guidance on the use of Riluzole with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Riluzole in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Riluzole in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Riluzole in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Riluzole in patients who are immunocompromised.
Others
(Description)
Administration and Monitoring
Administration
- Oral
- Riluzole tablets should be taken at least an hour before, or two hours after, a meal to avoid a food-related decrease in bioavailability.
Monitoring
There is limited information regarding Riluzole Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Riluzole and IV administrations.
Overdosage
- No specific antidote or information on treatment of overdosage with RILUTEK is available. In the event of overdose, RILUTEK therapy should be discontinued immediately. Experience with riluzole overdose in humans is limited. Neurological and psychiatric symptoms, acute toxic encephalopathy with stupor, coma, and methemoglobinemia have been observed in isolated cases. Treatment should be supportive and directed toward alleviating symptoms.
- Severe methemoglobinemia may be rapidly reversible after treatment with methylene blue.
- The estimated oral median lethal dose is 94 mg/kg and 39 mg/kg for male mice and rats, respectively.
Pharmacology
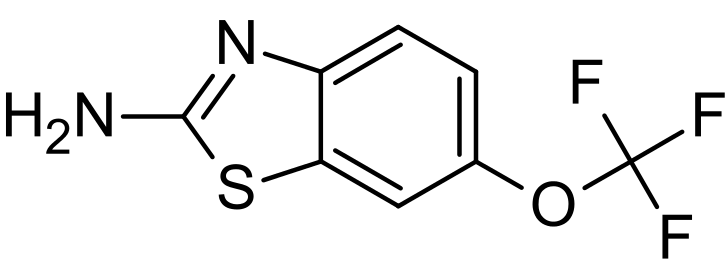
| |

| |
Riluzole
| |
| Systematic (IUPAC) name | |
| 6-(trifluoromethoxy)benzothiazol-2-amine | |
| Identifiers | |
| CAS number | |
| ATC code | N07 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 234.199 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | 60±18%[1] |
| Protein binding | 97%[1] |
| Metabolism | Hepatic (CYP1A2)[1] |
| Half life | 9-15 hours[1] |
| Excretion | Urine (90%)[1] |
| Therapeutic considerations | |
| Licence data |
|
| Pregnancy cat. | |
| Legal status | |
| Routes | Oral |
Mechanism of Action
There is limited information regarding Riluzole Mechanism of Action in the drug label.
Structure
There is limited information regarding Riluzole Structure in the drug label.
Pharmacodynamics
There is limited information regarding Riluzole Pharmacodynamics in the drug label.
Pharmacokinetics
There is limited information regarding Riluzole Pharmacokinetics in the drug label.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment Of Fertility
- Riluzole was not carcinogenic in mice or rats when administered for 2 years at daily oral doses up to 20 mg/kg and 10 mg/kg, respectively, which are approximately equivalent to the maximum human dose on a mg/m2 basis.
- The genotoxic potential of riluzole was evaluated in the bacterial mutagenicity (Ames) test, the mouse lymphoma mutation assay in L5178Y cells, the in vitro chromosomal aberration assay in human lymphocytes and the in vivo rat cytogenetic assay and in vivo mouse micronucleus assay in bone marrow. There was no evidence of mutagenic or clastogenic potential in the Ames test, the mouse lymphoma assay, or the in vivo assays in the mouse and rat. There was an equivocal clastogenic response in the in vitro human lymphocyte chromosomal aberration assay, which was not reproduced in a second assay performed at equal or higher concentrations; riluzole was therefore considered non-clastogenic in the human lymphocyte assay.
- N-hydroxyriluzole, the major active metabolite of riluzole, caused chromosomal damage in the in vitro mammalian mouse lymphoma assay and in the in vitro micronucleus assay that used the same mouse lymphoma cell line, L5178Y. N-hydroxyriluzole was not mutagenic in this cell line when tested in the HPRT gene mutation assay, and was negative in the Ames bacterial gene mutation assay (with and without rat or hamster S9), the in vitro UDS assay in rat hepatocytes, the chromosomal aberration test in human lymphocytes, and the in vivo mouse bone marrow micronucleus test.
- Riluzole impaired fertility when administered to male and female rats prior to and during mating at an oral dose of 15 mg/kg or 1.5 times the maximum daily dose on a mg/m2 basis.
Clinical Studies
There is limited information regarding Riluzole Clinical Studies in the drug label.
How Supplied
- Riluzole tablets, USP 50 mg are white to off white colored, round-shaped, biconvex film-coated tablets and debossed with “538” on one side and plain on other side.
They are supplied as follows:
- Unit Dose Box of 30’s ………………...NDC 0179-0150-70
Storage
Store at 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F)and protect from bright light.
Images
Drug Images
{{#ask: Page Name::Riluzole |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Riluzole |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patients should be advised to report any febrile illness to their physicians.
- Patients should be advised to report any cough or difficulties in breathing to their physicians.
- Patients and caregivers should be advised that riluzole tablets should be taken on a regular basis and at the same time of the day (e.g., in the morning and evening) each day. If a dose is missed, take the next tablet as originally planned.
- Patients should be warned about the potential for dizziness, vertigo, or somnolence and advised not to drive or operate machinery until they have gained sufficient experience on riluzole to gauge whether or not it affects their mental and/or motor performance adversely.
- Whether alcohol increases the risk of serious hepatotoxicity with riluzole is unknown; therefore, patients being treated with riluzole should be discouraged from drinking excessive amounts of alcohol.
- Patients should also be made aware that riluzole tablets should be stored at temperatures 20° to 25°C (68° to 77°F); excursions permitted between 15° and 30°C (59° and 86°F) and protected from bright light.
- Riluzole tablets must be kept out of the reach of children.
Precautions with Alcohol
Alcohol-Riluzole interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Rilutek
Look-Alike Drug Names
There is limited information regarding Riluzole Look-Alike Drug Names in the drug label.
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Riluzole |Label Name=Riluzole image.jpg
}}
{{#subobject:
|Label Page=Riluzole |Label Name=Riluzole ingredients and appearance.png
}}