Blood donation
|
WikiDoc Resources for Blood donation |
|
Articles |
|---|
|
Most recent articles on Blood donation Most cited articles on Blood donation |
|
Media |
|
Powerpoint slides on Blood donation |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Blood donation at Clinical Trials.gov Trial results on Blood donation Clinical Trials on Blood donation at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Blood donation NICE Guidance on Blood donation
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Blood donation Discussion groups on Blood donation Patient Handouts on Blood donation Directions to Hospitals Treating Blood donation Risk calculators and risk factors for Blood donation
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Blood donation |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Blood donation is a process by which a blood donor voluntarily has blood drawn for storage in a blood bank, generally for subsequent use in a blood transfusion.
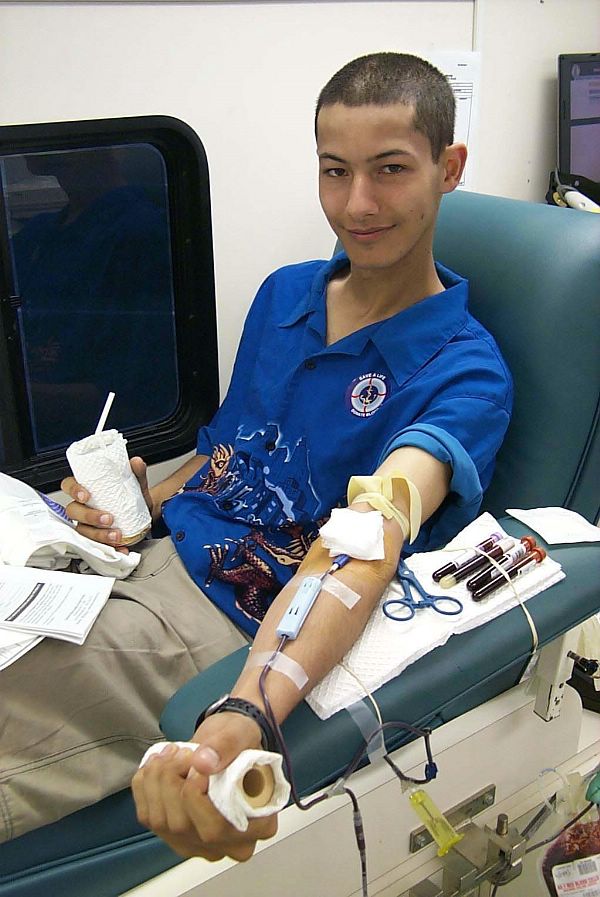
Blood donations may be scheduled at local centres, or at times a "blood drive" will occur. These are events where a blood bank or other blood collecting organization will set up in a convenient location—such as a shopping centre, large employer, university, high school, or a local house of worship—for people to stop in without appointment during their daily routine to donate blood. Sometimes a bloodmobile is used to run a blood drive. Usually a modified recreational vehicle, it is an easy place to run a blood drive because it is already set up for the process of blood donation.
Process
The process of giving blood involves screening the donor, the actual donation, and a brief recovery period. This applies to whole blood donations as well as plasmapheresis (donation of only one's plasma) and apheresis (donation of only one of blood constituents) in general.
Preparation
In the days prior to donating blood, it is important that donors prepare themselves for a process that can temporarily weaken the body. Donors should check their local blood bank's guidelines, as requirements and recommendations vary. Many blood banks recommend that potential donors drink extra water and fluids before donating.[1][2][3] It may be advisable to avoid caffeinated beverages before donation.[3] Eating well is also important, and can reduce the risk of reactions to donation.[1][2] Eating foods high in iron (which has many dietary sources) is also a good idea; low hemoglobin levels may make a donor temporarily ineligible.[1][2][3] This is especially true for women who have menstruated recently, because of iron loss in the menses.[4] Some countries require donors to observe a limited or full fast before donation because of specific screening methods; in particular, some countries will discard blood taken too soon after a fatty meal. High levels of lipemia (essentially fat in the blood) can interfere with infectious disease testing.
Screening
Western countries typically impose screening for blood donor candidates. In the past, it was the practice in America and other countries to separate blood donations on the basis of race, ethnicity, or religion, or to exclude certain groups from the donor pool on those bases. Currently, in the United States, these practices have been eliminated, and donor attributes are considered only in terms of their likelihood to affect the probability of transmission of disease. All blood products in the U.S. are labeled as coming from either "paid" or "volunteer" donors, with paid units being more likely to transmit infection. (Several other countries avoid paid donations altogether for this reason.)
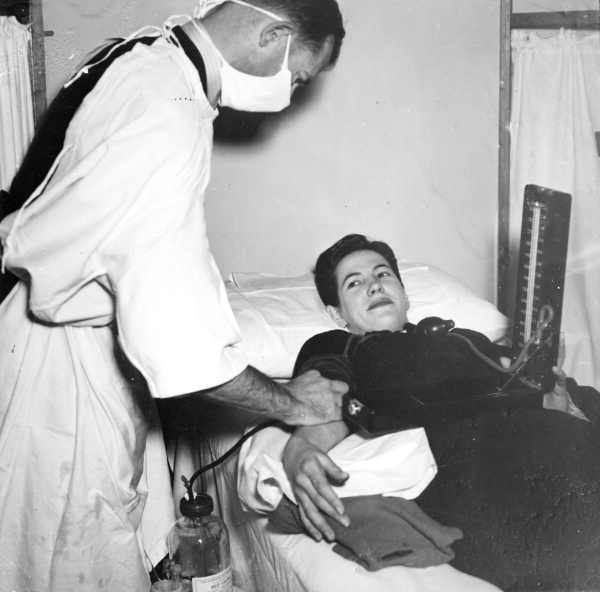
Other donor characteristics are also taken into account: starting in 1985, the American Red Cross and Food and Drug Administration policies prohibit accepting blood donations from gay/bisexual men, specifically from any "male who has had sex with another male since 1977, even once,"[5] or from IV drug users or recent immigrants from certain nations with high rates of HIV infection. The continued inclusion of men who have sex with men on the prohibited list has created some degree of controversy [2]; the FDA cites the public policy need to protect the blood supply from HIV and similar diseases as justification for the continued lifetime ban, while others believe the ban to be discriminatory, since sexually active heterosexuals are not categorically banned and all donated blood is screened. Policies vary in other countries; for instance, Australia formerly had a similar ban, but now only prohibits donating blood within one year after male-male sex (longer than the typical window period for HIV blood screening tests performed on donated blood). In Finland the parliamentary ombudsman launched an investigation on the possible unconstitutionality of the life-time ban in January 2006. The investigation continues as of December 2007.
Similarly important donor eligibility requirement in the US is related to concerns about variant Creutzfeldt-Jakob Disease (vCJD): persons who have spent long periods of time in countries where "mad cow disease" is found[6] are not eligible to donate. As part of the screening interview, blood donors are questioned about past residency in countries on the exclusion list, tracing back as far as 1980. The list of countries of residence that may disqualify a potential donor includes most of Western Europe (with stronger restrictions on those with past residence in the United Kingdom), Turkey, and all of Eastern Europe except Russia, Ukraine, Belarus, and the Baltic countries (those formerly part of the Soviet Union). From 1980 through 1996, if a person visited or resided in country on the exclusion list for a time that adds up to five years or more, the person is not eligible to donate; if a person spent time that adds up to three months or more in the United Kingdom, they are indefinitely deferred. This set of rules affects members of the US Armed Forces and others, who lived at least six months in certain European countries or 3 months in the United Kingdom from 1980 to 1996, who are therefore prohibited from donating blood.[7] A similar policy with regard to vCJD applies in France, Quebec and in the rest of Canada.[8]
When a donor arrives at a donation site, he or she typically fills out a consent form as well as answer an extensive questionnaire to help determine their eligibility. Questions include the donors' age, weight, most recent donation, current health, and various risk factors such as tattooing, illicit use of intravenous drugs (recreational or performance enhancing), residency abroad, recent international travel, and sexual history. In some countries, answers are associated with the donor's blood, but not name, to provide anonymity; in others such as the United States, names are kept to create deferral lists.
Often the blood hemoglobin concentration will be checked next, typically performed by a phlebotomist. While various tests exist, the most common ones are:
- hematocrit: done in some places and requires a centrifuge. 38% is the required level for most blood donors in the US, though autologous donors are accepted with levels as low as 33%. Failing this cutoff is the highest cause of donors not being eligible to donate, as many premenopausal women have lower iron levels.
- colorimetric hemoglobin test using a hemoglobin photometer: a machine-read result from a chemical reaction on a testing strip.
- copper sulfate screening test (aka "float test"): measures the specific gravity of the donor's blood by placing a drop into a copper sulfate solution. The solution is calibrated so that a hemoglobin concentration of in >12.5 g/dl (the cut-off in the U.S. for donation) sinks.
Most blood is later tested for diseases, including STDs. If the test is positive, the donor will be notified and their blood discarded. The tests used are high-sensitivity screening tests and no actual diagnosis is made. Some of the test results are later found to be false positives using more specific testing. Individuals are discouraged from using blood donation for the purpose of anonymous STD screening because a false negative could mean the disease would be passed to someone else. Blood may also be tested for additional infectious diseases such as West Nile Virus, when and where these diseases are prevalent.[9] Some donations, typically autologous blood, are not tested. In the US, these must be clearly labeled as untested.
Donated blood is tested by many methods, and a typical screening panel includes most of the tests below:
- Antibody to Hepatitis B core "anti-HBc"
- Hepatitis B Surface Antigen "HBsAg"
- Nucleic acid testing by Transcription Mediated Amplification (TMA) or Polymerase Chain Reaction (PCR) for Hepatitis B "HBV-NAT"
- Antibody to Hepatitis C "anti-HCV"
- Nucleic acid testing for HCV "HCV-NAT"
- Alanine transaminase "ALT" (this test is used to check for liver problems which may be a sign of hepatitis and has been phased out as tests for hepatitis have improved)
- Antibody to HIV types 1 and 2 "Anti-HIV1/2"
- Nucleic acid for HIV "HIV-NAT"
- Antibody to HIV p24 antigen (this test has been mostly replaced by HIV NAT)
- Antibody to Human T-Lymphotrophic Virus I/II "anti-HTLV"
- Nucleic acid testing for West Nile Virus "WNV-NAT"
- Antibody to Chagas Disease
- Serologic test for syphilis "RPR" or "STS"
- Antibody to Cytomegalovirus "anti-CMV"
- Atypical red cell antigen screening
- Blood typing
Most of the tests used on blood donors are designed to sacrifice specificity for sensitivity, so false positives are not unusual. Blood donors are often told to seek medical care for an actual diagnosis when they test positive for a disease in screening, since these tests are designed for the safety of the recipient, not necessarily for diagnostic purposes. Donors are typically excluded based on the screening test, but may be reinstated depending on the results of more accurate confirmatory testing.
Donation
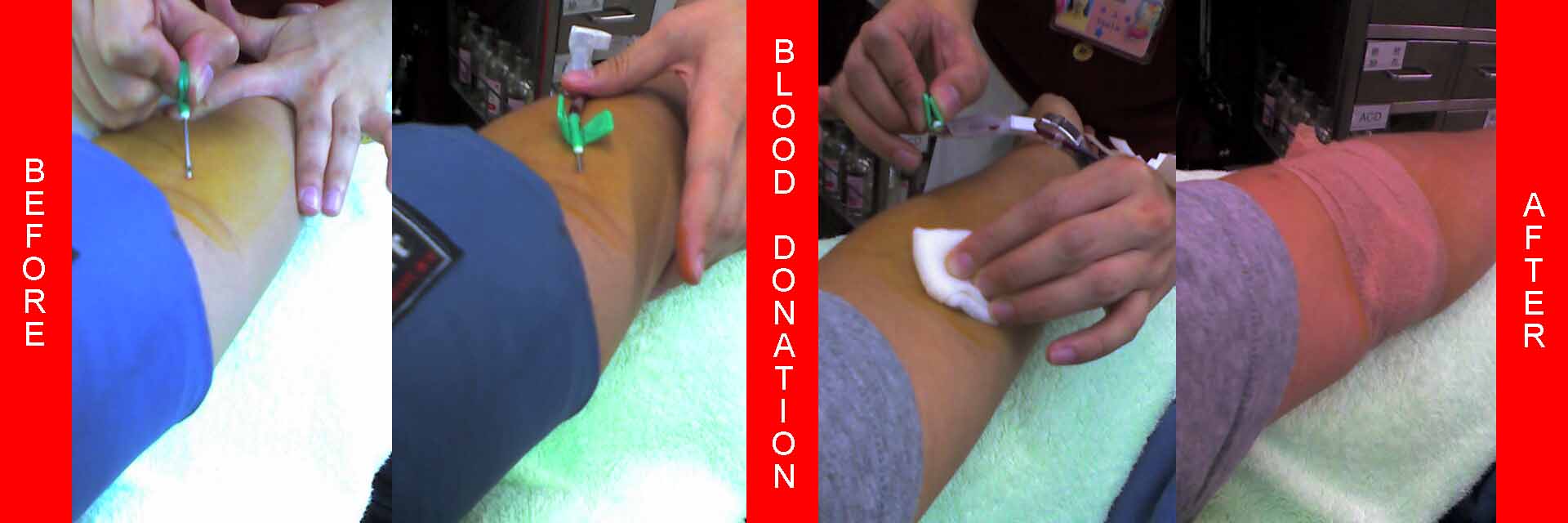
The blood donation itself happens next. The donor lies supine on a cushioned bench and extends an arm. The inner elbow is disinfected, and a cannula is inserted into the median cubital vein. A large bore needle is used to prevent hemolysis. The donor often has a tourniquet wrapped around his or her arm, or may be prompted to squeeze a ball repeatedly, to help speed the process. Blood flows from the vein, through the needle and a tube, and into a special collection bag which is placed on a small scale to measure the amount of blood withdrawn. After the bag is sufficiently filled, several test tubes are drawn from the same needle to be used for disease detection. Typically, around 450 millilitres, about a U.S. pint, is drawn during the blood donation process. The blood is stored in a blood bag which contains acid citrate dextrose, an anticoagulant which prevents blood clotting and preserves the blood for long periods of time.
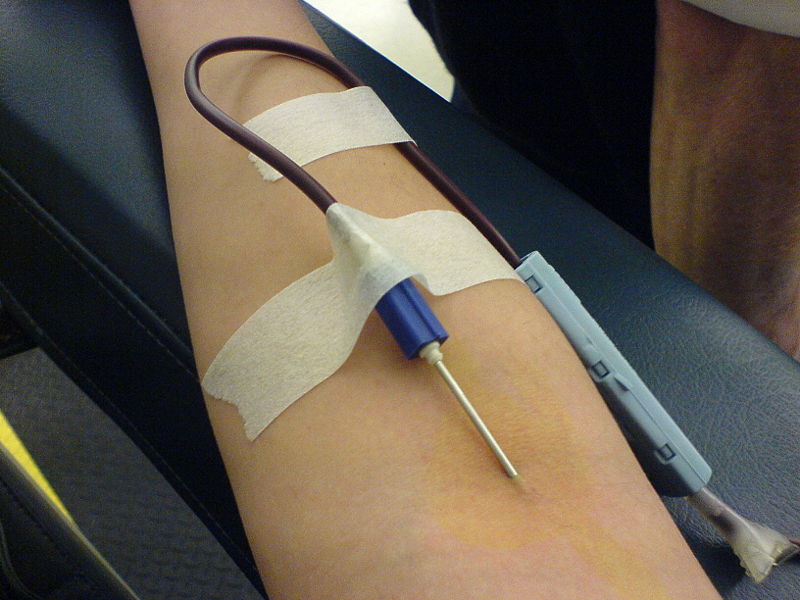

Apheresis
Rather than donating whole blood, a donor sometimes has the option to donate only some blood components while retaining others. This process is known as apheresis, and is more involved, time consuming, and requires more specialized equipment. The benefit is that more of the desired components can be concentrated and removed, and the donor is usually able to donate significantly more frequently than if whole blood had been removed. In some cases, the usefulness of the removed components is not as sensitive to blood type considerations. Additionally, donors with high platelet counts can be divided in what is referred to as a "split" or "double" and a single donation can be used for two future transfusions. A "triple" donation is possible, but requires a very high platelet count in the donor and specific equipment and setup.
The typical method of apheresis is to draw whole blood from the donor, then centrifuge the blood to separate its components (see apheresis for more information). The desired components (e.g. platelets, plasma) are removed and then the remaining components are returned to the donor.
"Double red"
A recent innovation in apheresis is the "double red" donation, which extracts two units of red blood cells instead of the single unit of an ordinary whole-blood donation. This provides several benefits to both the donor and the blood bank. The donor can make the same red-cell contribution with half the visits, and the return of plasma to the body leaves the donor better hydrated. The process takes somewhat longer than a standard donation (about 35-45 minutes), but is much shorter than a regular apheresis visit. The blood bank receives twice the usual red-cell donation in each visit. Patients who require the blood will then not be as susceptible to rejection, as there will be fewer sources and less mixture. Because more red cells are removed from the circulatory system, donors must meet some additional health requirements for a double-red donation.[10][11][12]
Recovery
Once the donation is complete, the donor is given a bandage or gauze to stop further blood flow and is normally allowed to leave. Beverages and snacks may be provided to restore blood volume and blood sugar, and to settle the stomach. Donors are often encouraged to sit down for a few minutes to return to normality, as lightheadedness and faintness may occur. In some countries, stickers are worn by donors to show the donor's pride and to encourage others to give. The entire process, from screening through recovery has a variable time, it can take from some fifteen minutes to about one hour. The actual blood donation takes between four and fifteen minutes for whole blood donation. Donors are discouraged from heavy exercise, such as running, rowing, or lifting for several hours.[13] Plasma volumes will return to normal in around 24 hours, while red blood cells are replaced by bone marrow into the circulatory system within about 3-5 weeks, and lost iron replaced over 6-8 weeks. This recovery process can be accelerated by eating foods high in complex carbohydrates, iron, and other trace minerals.
Post donation information
Blood donors are typically given information about who to contact if he or she experiences medical complications or thinks that the blood donated may not be safe for use. Contacts taken for either reason is rare, but the option is provided to ensure the safety of the blood and donor. Each unit of whole blood has a unique tracking number so that new information can be linked to it.
How long until next eligible to give?
Due to the timeframe required for iron replacement, donors are eligible to donate whole blood approximately 8 to 16 weeks after the previous donation, the exact period varying by country and sex. In the USA, whole blood donations can be taken every 8 weeks, whilst in the UK it is a wait of 16 weeks. The waiting period after "double red" apheresis is 16 weeks in the USA.[11][12] Donors are sometimes given iron supplements to be able to donate more regularly.
Storage
Cryopreservation of red blood cells is done to store special, rare red blood cells for up to 10 years. The ten year outdate is not based on any specific research; it was chosen arbitrarily. Some blood banks maintain truly rare red blood cells past ten years because there are no alternative sources. The cells are first incubated in a 40% glycerol solution which acts as a cryoprotectant ("antifreeze") within the cells. The units are then placed in special sterile containers in a deep freezer at less than -60 °C.
Donors for babies
A tiny percentage of adults may donate blood to small children in emergency rooms, newborn babies, and fetuses. To ensure the safety of blood transfusion to pediatric patients, including those in whom the immune systems are not fully developed, hospitals are taking every precaution to avoid infection and prefer to use specially tested pediatric blood units that are guaranteed negative for cytomegalovirus (CMV),[14] because the consequences of CMV infection for newborns or low weight infants may be severe or even fatal. Additionally, for pediatric patients with certain disorders or in emergency, when there is no time to perform crossmatching, only O/Rh negative blood can be used for neonatal transfusion.[15] Due to these specific restrictions, to be recognized as a baby donor an adult must test negative for cytomegalovirus. Since only 7% of US adults are O-negative and as few as 15% of adults do not carry CMV, only 1% of adults may qualify as baby donors. Other restrictions (body weight, HIV status, vCJD, etc. ) reduce the fraction of potential baby donors to less than 1 in 200. If an adult was not exposed to measles, mumps, rubella, or chicken pox, that amplifies the person's status as a baby donor.
Newborn babies that require multiple surgeries often need up to 20 units of blood in their first few weeks.
Complications
The finger prick and needle insertion typically cause only minor discomfort. Negative medical reactions to blood donation are rare, and blood bank staff are trained to handle them.
Problems can occur, however, and fall into three groups:
- Hypovolemic reactions can occur because of a rapid change in blood pressure. These are far more common in donors that are extremely nervous, dehydrated, or who haven't eaten recently. These are typically mild, and nausea and fainting are generally the worst problems encountered. Donors with significant heart disease may have substantially more serious problems, and they are explicitly excluded from donating because of these risks.
These are typically treated by placing the donor in a Trendelenburg position (on a stretcher tilted "legs up"). Beverages may be given to replenish blood volume further, and the donor may have to remain in a horizontal position for a while under monitoring. Most blood banks encourage donors to rest for several minutes after donation (often while eating and drinking) to let them adjust to the loss of blood and reduce the risk of these problems.
- Physical trauma can occur at the needle site, generally due to poor needle placement. Serious problems can occur if the needle strikes an artery (Arterial stick) or damage the nerve, but both are extremely rare. Infections at the needle site (phlebitis) are unlikely because of the aggressive site preparation, but are theoretically possible.
Bruising of the arm (Hematoma) in the area of the needle insertion is not uncommon. It is almost always harmless and fades away after a week or two. One study found the incidence of bruising to be less than 1%;[16] anecdotally, donors have reported widely varying incidences. Donors who have had clotting problems or are on high doses of anticoagulants are typically deferred because of their higher risks for these complications.
Phlebotomists sometimes experience difficulty in obtaining a full unit of blood from some donors. This is not a problem for the donor, because the session simply ends with an incomplete unit.
- Donors sometimes have adverse reactions to the sodium citrate used in apheresis collection procedures to keep the blood from clotting. Since the anticoagulant is returned to the donor along with blood components that are not being collected, it can bind the calcium in the donor's circulatory system. Citrate reactions tend to cause tingling in the lips, but may cause convulsions or more serious reactions. Donors are sometimes given calcium supplements (e.g. Tums) to chew during the donation to prevent these side effects.
A meeting of the Biological Product Advisory Committee (BPAC) of the US FDA examined fatalities among plasmapheresis donors, who donate much more frequently, and in available information did not find a difference between the death rate of blood donors and that of the US population as a whole. Reported donor deaths were mostly in donors who had a previously undiagnosed cardiovascular disease.
Benefits
In patients prone to iron overload (e.g. due to hemochromatosis), blood donation prevents the accumulation of iron. Blood banks in the US must label the blood if it is from a therapeutic donor, so most do not accept donations from donors with any blood disease[17] . Others, such as the Australian Red Cross Blood Service, accept blood from donors with hemochromatosis. It is a genetic disorder that does not affect the safety of the blood.
Donating blood may reduce the risk of heart disease for men[18] and stimulate the generation of red blood cells. Differences between men and women in heart attack risk may be due to circulating iron levels which are reduced by menstruation. Donating blood may reduce men's risk to that of women's, but the link has not been firmly established. [19]
Other incentives are sometimes added by employers, usually time off for the purposes of donating. Blood centers will also sometimes add incentives such as assurances that donors would have priority during shortages or other programs. Other incentives may include prize drawings for donors and rewards for organizers of successful drives, especially during periods when blood is often in short supply. Some blood donors, such as for plasmapheresis in the US, are paid for donating. This again leads to concerns about donors being less than truthful about their health in order to receive money.
A donor effectively burns about 650 calories by donating one pint of blood. [20] Donors who are on aggressive diets may be at a higher risk for some complications.
The majority of blood donors, however, donate as an act of charity and do not expect to receive any direct benefit from the donation.
Autologous donation
A person who anticipates the need for a blood transfusion at a later date (usually because of scheduled surgery) may make an autologous donation, in which their blood is stored and later transfused back into its original donor. Besides ensuring the availability of compatible blood (especially important for patients with rare blood types), this procedure also eliminates the risk of disease transmission from infected donors. Autologous donation is sometimes done by the hospital instead of a community blood bank. Eligibility requirements are relaxed for autologous donors, as the blood is not used for anyone else. Generally, any patient who is eligible for elective surgery is eligible for autologous donations, though there are some exceptions, particularly history of heart disease.
This process is accepted by Jehovah's Witnesses because it does not introduce foreign blood into the body.
See also
| Wikimedia Commons has media related to Blood donation. |
References
- ↑ 1.0 1.1 1.2 "Giving Blood -> What to Expect". Australian Red Cross Blood Service. Retrieved 2007-10-06.
- ↑ 2.0 2.1 2.2 "The Donation Experience". Canadian Blood Services. Retrieved 2006-12-17.
- ↑ 3.0 3.1 3.2 "Tips for a Good Donation Experience". American Red Cross. Retrieved 2006-12-17.
- ↑ "What is low hematocrit?". American Red Cross. Retrieved 2006-12-17.
- ↑ "American Red Cross Dogged by Allegations of Discrimination (published: January 28, 2002)". The Harvard business school newspaper. Retrieved 2006-12-17.
- ↑ "Blood Donation Eligibility Guidelines (vCJD)". American Red Cross. Retrieved 2006-12-17.
- ↑ "List of countries that may disqualify a potential donor for the vCJD screening purposes". American Red Cross. Retrieved 2006-12-17.
- ↑ "Donor Qualification criteria". Héma-Québec, Canada. Retrieved 2006-12-17.
- ↑ "Precautionary West Nile virus blood sample testing". Héma-Québec, Canada. Retrieved 2006-12-17.
- ↑ ""Double Up to Save Lives"". United Blood Services. Retrieved 2007-02-23.
- ↑ 11.0 11.1 ""Double the Difference"". American Red Cross (Greater Chesapeake and Potomac). Retrieved 2007-02-23.
- ↑ 12.0 12.1 ""Double Red Cell"". American Red Cross (New England). Retrieved 2007-02-23.
- ↑ American Red Cross. Tips for a Good Donation Experience. Retrieved on March 22, 2007.
- ↑ "Red blood cell transfusions in newborn infants: Revised guidelines". Canadian Paediatric Society (CPS). Retrieved 2007-02-02.
- ↑ "Neonatal Transfusion Recommendations at RCH". The Royal Children's Hospital (Melbourne, Australia). Retrieved 2007-02-02.
- ↑ Ranasinghe E, Harrison JF. "Bruising following blood donation (abstract)". NCBI. Unknown parameter
|accessyear=ignored (|access-date=suggested) (help); Unknown parameter|accessmonthday=ignored (help) - ↑ "Variances for Blood Collection from Individuals with Hereditary Hemochromatosis". US Food and Drug Administration. Retrieved 2007-07-18.
- ↑ "Cohort study of relation between donating blood and risk of myocardial infarction in 2682 men in eastern Finland". BMJ. Retrieved 2006-12-17.
- ↑ "Evidence suggests that giving blood has health benefits". WebMD. Retrieved 2007-12-2. Check date values in:
|accessdate=(help) - ↑ "Donate Blood - Frequently Asked Questions". Mayo Clinic. Retrieved 2007-11-27.
External links
- US Blood Donation Campaign - home of the Red Defender
- LifeSource - Blood Donation Centers in Illinois
- Blood Donation and Processing
- FAQs and donor information from the American Red Cross
- USA Directory of Blood Donation Centers
- National Blood Service (England and North Wales)
- Give and Let Live, NHS Blood and Transplant service, education site for schools (England and North Wales)
- The Welsh Blood Service Gwasanaeth Gwaed Cymru (South Wales)
- Scottish National Blood Transfusion Service
- Australian Red Cross Blood Service
- Breakthrough makes all blood types universal - Joyce Howard Price, The Washington Times - April 4, 2007
- Indian Blood donors information and Blood Donation Facts
- Slovenian donors information and Blood Donation
- JMH Blood Services, Abingdon, Va. Blood bank serving Southwestern Virginia
- Mumbai Blood Donor Database and Information Resource
bg:Кръводаряване cs:Darování krve da:Bloddonor de:Blutspende ko:헌혈 id:Donor darah it:Donazione del sangue he:תרומת דם nl:Bloeddonatie no:Blodgivning pa:ਖੂਨ ਦਾਨ sk:Darovanie krvi fi:Verenluovutus sv:Blodgivning th:การบริจาคโลหิต bat-smg:Krauja donorīstė