Carbohydrate
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Carbohydrates (from 'hydrates of carbon') or saccharides (Greek σάκχαρον meaning "sugar") are simple organic compounds that are aldehydes or ketones with many hydroxyl groups added, usually one on each carbon atom that is not part of the aldehyde or ketone functional group. Carbohydrates are the most abundant of the four major classes of biomolecules, which also include proteins, lipids and nucleic acids. They fill numerous roles in living things, such as the storage and transport of energy (starch, glycogen) and structural components (cellulose in plants, chitin in animals). Additionally, carbohydrates and their derivatives play major roles in the working process of the immune system, fertilization, pathogenesis, blood clotting, and development.
The basic carbohydrate units are called monosaccharides, such as glucose, galactose, and fructose. The general stoichiometric formula of an unmodified monosaccharide is (C·H2O)n, where n is any number of three or greater; however, many molecules with formulae that differ slightly from this are still called carbohydrates and other compounds that possess formulae that agree with this general rule may not be in fact carbohydrates (eg formaldehyde).[1] Despite the inexactness of the term, "carbohydrate" remains a useful descriptive name and with a little experience even a novice will soon become aware of what is, and is not, a carbohydrate. Monosaccharides can be linked together in almost limitless ways. Many carbohydrates contain one or more modified monosaccharide units that have had one or more groups replaced or removed. For example, deoxyribose, a component of DNA, is a modified version of ribose; chitin is composed of repeating units of N-acetylglucosamine, a nitrogen-containing form of glucose. The names of carbohydrates often end in the suffix -ose.
Monosaccharides
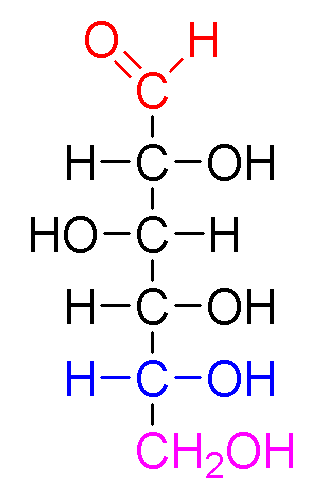
Monosaccharides are the simplest carbohydrates in that they cannot be hydrolyzed to smaller carbohydrates. The general chemical formula of an unmodified monosaccharide is (C•H2O)n, where n is any number of three or greater.
Classification of monosaccharides
Monosaccharides are classified according to three different characteristics: the placement of its carbonyl group, the number of carbon atoms it contains, and its chiral handedness. If the carbonyl group is an aldehyde, the monosaccharide is an aldose; if the carbonyl group is a ketone, the monosaccharide is a ketose. Monosaccharides with three carbon atoms are called trioses, those with four are called tetroses, five are called pentoses, six are hexoses, and so on. These two systems of classification are often combined. For example, glucose is an aldohexose (a six-carbon aldehyde), ribose is an aldopentose (a five-carbon aldehyde), and fructose is a ketohexose (a six-carbon ketone).
Each carbon atom bearing a hydroxyl group (-OH), with the exception of the first and last carbons, are asymmetric, making them stereocenters with two possible configurations each (R or S). Because of this asymmetry, a number of isomers may exist for any given monosaccharide formula. The aldohexose D-glucose, for example, has the formula (C·H2O)6, of which all but two of its six carbons atoms are stereogenic, making D-glucose one of 24 = 16 possible stereoisomers. In the case of glyceraldehyde, an aldotriose, there is one pair of possible stereoisomers, which are enantiomers and epimers. 1,3-dihydroxyacetone, the ketose corresponding to the aldose glyceraldehye, is a symmetric molecule with no stereocenters). The assignment of D or L is made according to the orientation of the asymmetric carbon furthest from the carbonyl group: in a standard Fischer projection if the hydroxyl group is on the right the molecule is a D sugar, otherwise it is an L sugar. Because D sugars are biologically far more common, the D is often omitted.
Conformation
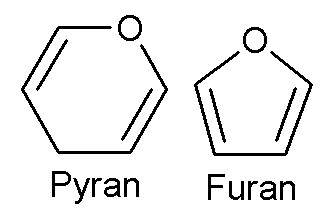
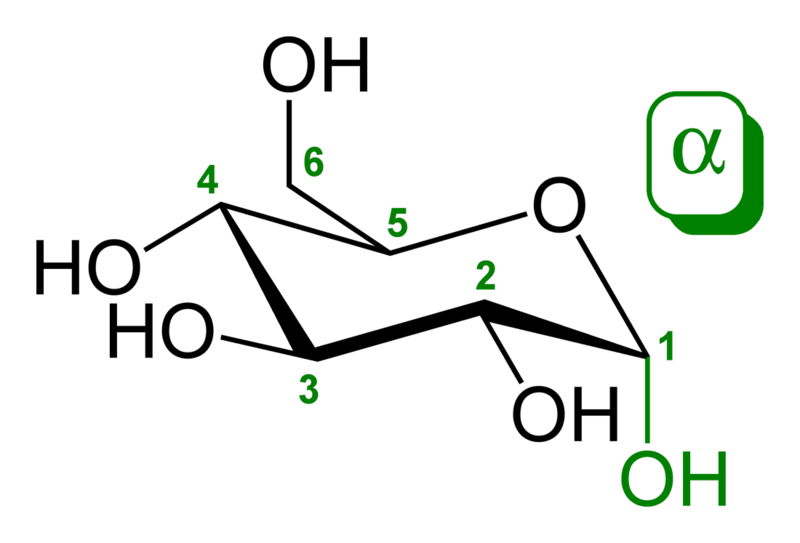
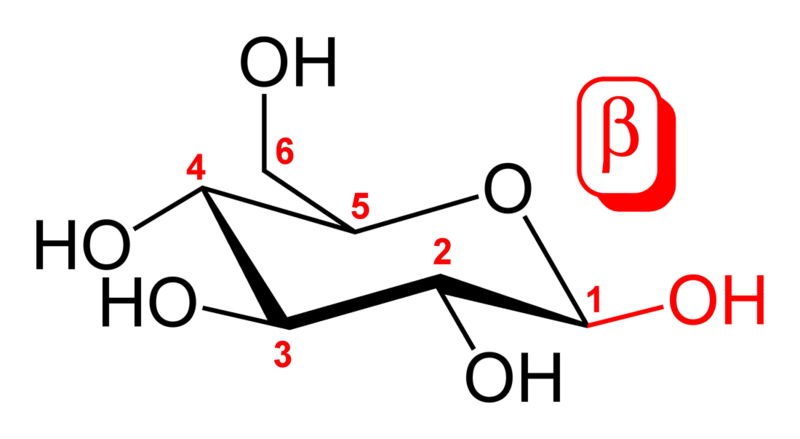
The aldehyde or ketone group of a straight-chain monosaccharide will react reversibly with a hydroxyl group on a different carbon atom to form a hemiacetal or hemiketal, forming a heterocyclic ring with an oxygen bridge between two carbon atoms. Rings with five and six atoms are called furanose and pyranose forms, respectively, and exist in equilibrium with the straight-chain form.
During the conversion from straight-chain form to cyclic form, the carbon atom containing the carbonyl oxygen, called the anomeric carbon, becomes a chiral center with two possible configurations: the oxygen atom may take a position either above or below the plane of the ring. The resulting possible pair of stereoisomers are called anomers. In the α anomer, the -OH substituent on the anomeric carbon rests on the opposite side (trans) of the ring from the CH2OH side branch. The alternative form, in which the CH2OH substituent and the anomeric hydroxyl are on the same side (cis) of the plane of the ring, is called the β anomer. Because the ring and straight-chain forms readily interconvert, both anomers exist in equilibrium.
Use in living organisms
Monosaccharides are the major source of fuel for metabolism, being used both as an energy source (glucose being the most important in nature) and in biosynthesis. When monosaccharides are not needed by cells they are quickly converted into another form, such as polysaccharides.
Disaccharides
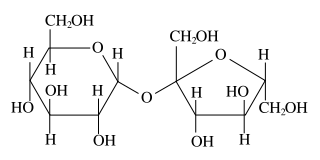
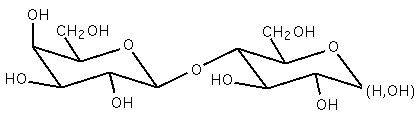
Two joined monosaccharides are called disaccharides and represent the simplest polysaccharides. Examples include sucrose and lactose. They are composed of two monosaccharide units bound together by a covalent bond known as a glycosidic linkage formed via a dehydration reaction, resulting in the loss of a hydrogen atom from one monosaccharide and a hydroxyl group from the other. The formula of unmodified disaccharides is C12H22O11. Although there are numerous kinds of disaccharides, a handful of disaccharides are particularly notable.
Sucrose, pictured to the right, is the most abundant disaccharide and the main form in which carbohydrates are transported in plants. It is composed of one D-glucose molecule and one D-fructose molecule. The systematic name for sucrose, O-α-D-glucopyranosyl-(1→2)-D-fructofuranoside, indicates four things:
- Its monosaccharides: glucose and fructose
- Their ring types: glucose is a pyranose, and fructose is a furanose
- How they are linked together: the oxygen on carbon number 1 (C1) of α-D-glucose is linked to the C2 of D-fructose.
- The -oside suffix indicates that the anomeric carbon of both monosaccharides participates in the glycosidic bond.
Lactose, a disaccharide composed of one D-galactose molecule and one D-glucose molecule, occurs naturally in milk. The systematic name for lactose is O-β-D-galactopyranosyl-(1→4)-D-glucopyranose. Other notable disaccharides include maltose (two D-glucoses linked α-1,4) and cellobiose (two D-glucoses linked β-1,4).
Oligosaccharides and polysaccharides
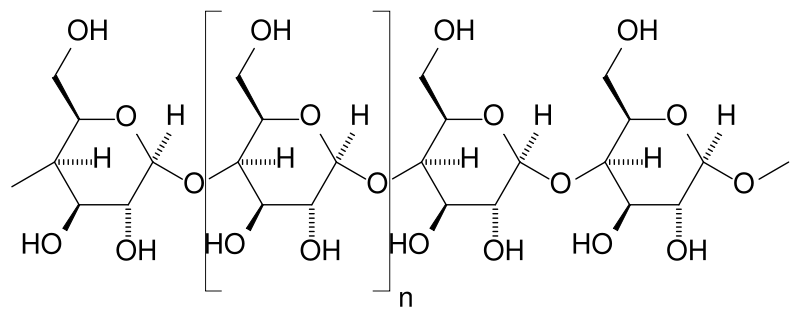
Oligosaccharides and polysaccharides are composed of longer chains of monosaccharide units bound together by glycosidic bonds. The distinction between the two is based upon the number of monosaccharide units present in the chain. Oligosaccharides typically contain between two and nine monosaccharide units, and polysaccharides contain greater than ten monosaccharide units. Definitions of how large a carbohydrate must be to fall into each category vary according to personal opinion. Examples of oligosaccharides include the disaccharides mentioned above, the trisaccharide raffinose and the tetrasaccharide stachyose.
Oligosaccharides are found as a common form of protein posttranslational modification. Such posttranslational modifications include the Lewis and ABO oligosaccharides responsible for blood group incompatibilities, the alpha-Gal epitope responsible for hyperacute rejection in xenotransplanation, and O-GlcNAc modifications.
Polysaccharides represent an important class of biological polymer. Their function in living organisms is usually either structure or storage related. Starch is used as a storage polysaccharide in plants, being found in the form of both amylose and the branched amylopectin. In animals, the structurally similar but more densely branched glycogen is used instead. Glycogen's properties allow it to be metabolized more quickly, which suits the active lives of locomotive animals.
Cellulose and chitin are examples of structural polysaccharides. Cellulose is used in the cell walls of plants and other organisms, and is claimed to be the most abundant organic molecule on earth.[2] It has a variety of uses including in the paper and textile industry and as a feedstock for the production of rayon (in the viscose process), cellulose acetate, celluloid and nitrocellulose. Chitin has a similar structure to cellulose but has nitrogen containing side branches, increasing its strength. It is found in arthropod exoskeletons and in the cell walls of some fungi. It has a variety of uses, for example in surgical threads.
Other polysaccharides include callose or laminarin, xylan, mannan, fucoidan, and galactomannan.
Nutrition
Carbohydrates require less water to digest than proteins or fats and are the most common source of energy. Proteins and fat are vital building components for body tissue and cells, and thus it could be considered advisable not to deplete such resources by necessitating their use in energy production.
Carbohydrates are not essential nutrients: the body can obtain all its energy from protein and fats [3] [4]. The brain cannot burn fat and needs glucose for energy, but the body can make this glucose from protein. Carbohydrates and proteins contain 4 kilocalories per gram while fats contain 9 kilocalories and alcohol contains 7 kilocalories per gram.[citation needed]
Foods that are high in carbohydrates include breads, pastas, beans, potatoes, bran, rice and cereals.
Based on evidence for risk of heart disease and obesity, the Institute of Medicine recommends that American and Canadian adults get between 40-65% of dietary energy from carbohydrates.[5] The Food and Agriculture Organization and World Health Organization jointly recommend that national dietary guidelines set a goal of 55-75% of total energy from carbohydrates, but only 10% should be from Free sugars (their definition of simple carbohydrates).[6]
The distinction between "good carbs" and "bad carbs" is an important attribute of low-carbohydrate diets, which promote a reduction in the consumption of grains and starches in favor of protein. The result is a reduction in insulin levels used to metabolize sugars, and an increase in the use of fat for energy through ketosis, a process also known as Rabbit starvation.
Classification
Dieticians and nutritionists commonly classify carbohydrates as simple (monosaccharides and disaccharides) or complex (oligosaccharides and polysaccharides). The term complex carbohydrate was first used in the Senate Select Committee publication Dietary Goals for the United States (1977), where it denoted "fruit, vegetables and whole-grains".[7] Dietary guidelines generally recommend that complex carbohydrates and nutrient-rich simple carbohydrates such as fruit and dairy products should make up the bulk of carbohydrate consumption. The USDA's Dietary Guidelines for Americans 2005 dispenses with the simple/complex distinction, instead recommending fiber-rich foods and whole grains.[8]
The glycemic index and glycemic load systems are popular alternative classification methods which rank carbohydrate-rich foods based on their effect on blood glucose levels. The insulin index is a similar, more recent classification method which ranks foods based on their effects on blood insulin levels. This system assumes that high glycemic index foods and low glycemic index foods can be mixed to make the intake of high glycemic foods more acceptable.[citation needed]
The World Health Organisation and Food and Agriculture Organization's joint expert report on Diet, Nutrition and the Prevention of Chronic Diseases (WHO Technical Report Series 916) advises carbohydrate consumption of 55-75% carbohydrate, but restricts "Free sugar" intake to 10%. Its definition is "The term "free sugars" refers to all monosaccharides and disaccharides added to foods by the manufacturer, cook or consumer, plus sugars naturally present in honey, syrups and fruit juices." (page 56 of the report; note to Table 6: Ranges of population nutrient intake goals). This is their effective split between simple and complex carbohydrates.
Metabolism
Catabolism
Catabolism is the metabolic reaction cells undergo in order to extract energy. There are two major metabolic pathways of monosaccharide catabolism:
Oligo/polysaccharides are cleaved first to smaller monosaccharides by enzymes called Glycoside hydrolases. The monosaccharide units can then enter into monosaccharide catabolism.
Carbohydrate chemistry
Carbohydrates are reactants in many organic reactions. For example:
- Carbohydrate acetalisation
- Cyanohydrin reaction
- Lobry-de Bruyn-van Ekenstein transformation
- Amadori rearrangement
- Nef reaction
- Wohl degradation
- Koenigs-Knorr reaction
An artificial carbohydrate is sorbitol.
See also
| Wikimedia Commons has media related to Carbohydrates. |
- Biochemistry
- Gluconeogenesis
- Glycolipid
- Glycoprotein
- Macromolecules
- Nutrition
- Pentose phosphate pathway
- Photosynthesis
- Sugar
Template:BranchesofFoodChemistry Template:Carbohydrates
References
- ↑ Matthews, C. E.; K. E. Van Holde; K. G. Ahern (1999) Biochemistry. 3rd edition. Benjamin Cummings. ISBN 0-8053-3066-6
- ↑ N.A.Campbell (1996) Biology (4th edition). Benjamin Cummings NY. p.23 ISBN 0-8053-1957-3
- ↑ *http://www.ajcn.org/cgi/content/full/75/5/951-a
- ↑ *http://jn.nutrition.org/cgi/reprint/136/5/1256?ijkey=ebf0450b5cf21e8d83dd43f62b5559254694f65f
- ↑ Food and Nutrition Board (2002/2005). Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids. Washington, DC: The National Academies Press. Page 769. ISBN 0-309-08537-3
- ↑ Joint WHO/FAO expert consultation (2003). Diet, Nutrition and the Prevention of Chronic Diseases (PDF). Geneva: World Health Organization. Pages 55-56. ISBN 92-4-120916-X
- ↑ Joint WHO/FAO expert consultation (1998), Carbohydrates in human nutrition, chapter 1. ISBN 92-5-104114-8.
- ↑ DHHS and USDA, Dietary Guidelines for Americans 2005, Chapter 7 Carbohydrates
External links
- Carbohydrates, including interactive models and animations (Requires MDL Chime)
- IUPAC-IUBMB Joint Commission on Biochemical Nomenclature (JCBN): Carbohydrate Nomenclature
- Carbohydrates detailed
- Carbohydrates and Glycosylation - The Virtual Library of Biochemistry and Cell Biology
- Consortium for Functional Glycomics
af:Koolhidraat ar:سكريات bn:শর্করা zh-min-nan:Carbohydrate bs:Ugljikohidrati bg:Въглехидрат ca:Hidrat de carboni cs:Sacharidy da:Kulhydrat de:Kohlenhydrate eo:Karbonhidrato eu:Karbono hidrato fa:کربوهیدرات gl:Carbohidrato ko:탄수화물 hr:Ugljikohidrati id:Karbohidrat is:Sykra it:Glucidi he:פחמימה la:Saccharidum lv:Ogļhidrāti lb:Glucid lt:Angliavandenis hu:Szénhidrát mk:Јаглехидрат ms:Karbohidrat nl:Koolhydraat no:Karbohydrat nn:Karbohydrat om:Carbohydrate simple:Carbohydrate sl:Ogljikovi hidrati sr:Угљени хидрати sh:Ugljeni hidrati su:Karbohidrat fi:Hiilihydraatti sv:Kolhydrat th:คาร์โบไฮเดรต uk:Вуглеводи bat-smg:Onglėondėnē
- Pages with script errors
- All articles with unsourced statements
- Articles with unsourced statements from August 2007
- Articles with invalid date parameter in template
- Articles with unsourced statements from February 2007
- Commons category link is locally defined
- Commons category link is on Wikidata using P373
- Carbohydrates
- Nutrition