Nebivolol
{{DrugProjectFormSinglePage |authorTag=Sophia Saad, Associate Editor - WikiDoc [1]; Turky Alkathery, M.D. [2] |genericName=Nebivolol |aOrAn=a |drugClass=Beta-Adrenergic Blocker |indicationType=treatment |indication=Hypertension |adverseReactions=Nausea, Dizziness, Headache, Somnolence |blackBoxWarningTitle=TITLE |blackBoxWarningBody=Condition Name: (Content)
|fdaLIADAdult=
- Dosing information
- The dose of Nebivolol must be individualized to the needs of the patient.
- Recommended starting dosage: 5 mg PO qd, with or without food, as monotherapy or in combination with other agents.
- For patients requiring further reduction in blood pressure, the dose can be increased at 2-week intervals up to 40 mg. A more frequent dosing regimen is unlikely to be beneficial.
|offLabelAdultGuideSupport=
- Class of Recommendation: Class I
- Level of Evidence: Level B
- Dosing Information
- Not applicable
|offLabelAdultNoGuideSupport=
- Dosing information
Prophylaxis of Migraine
- Dosing information
- 5 mg/day [3]
|fdaLIADPed=Safety and effectiveness in pediatric patients have not been established. Pediatric studies in ages newborn to 18 years old have not been conducted because of incomplete characterization of developmental toxicity and possible adverse effects on long-term fertility. |offLabelPedGuideSupport=There is limited information regarding Off-Label Guideline-Supported Use of Nebivolol in pediatric patients. |offLabelPedNoGuideSupport=There is limited information regarding Off-Label Non–Guideline-Supported Use of Nebivolol in pediatric patients. |contraindications=Nebivolol is contraindicated in the following conditions:
- Severe bradycardia.
- Heart block greater than first degree.
- Patients with cardiogenic shock.
- Decompensated cardiac failure
- Sick sinus syndrome (unless a permanent pacemaker is in place).
- Patients with severe hepatic impairment (Child-Pugh B).
- Patients who are hypersensitive to any component of this product.
|warnings====Abrupt Cessation of Therapy===
Do not abruptly discontinue Nebivolol therapy in patients with coronary artery disease. Severe exacerbation of angina, myocardial infarction and ventricular arrhythmias have been reported in patients with coronary artery disease following the abrupt discontinuation of therapy with β-blockers. myocardial infarction and ventricular arrhythmias may occur with or without preceding exacerbation of the angina pectoris. Caution patients without overt coronary artery disease against interruption or abrupt discontinuation of therapy. As with other β-blockers, when discontinuation of Nebivolol is planned, carefully observe and advise patients to minimize physical activity. Taper Nebivolol over 1 to 2 weeks when possible. If the angina worsens or acute coronary insufficiency develops, re-start Nebivolol promptly, at least temporarily.
Angina and Acute Myocardial Infarction
Nebivolol was not studied in patients with angina pectoris or who had a recent MI.
Bronchospastic Diseases
In general, patients with bronchospastic diseases should not receive β-blockers.
Anesthesia and Major Surgery
Because beta-blocker withdrawal has been associated with an increased risk of myocardial infarction and chest pain, patients already on beta-blockers should generally continue treatment throughout the perioperative period. If Nebivolol is to be continued perioperatively, monitor patients closely when anesthetic agents which depress myocardial function, such as ether, cyclopropane, and trichloroethylene, are used. If β-blocking therapy is withdrawn prior to major surgery, the impaired ability of the heart to respond to reflex adrenergic stimuli may augment the risks of general anesthesia and surgical procedures.
The β-blocking effects of Nebivolol can be reversed by β-agonists, e.g., dobutamine or isoproterenol. However, such patients may be subject to protracted severe hypotension. Additionally, difficulty in restarting and maintaining the heartbeat has been reported with β-blockers.
Diabetes and Hypoglycemia
β-blockers may mask some of the manifestations of hypoglycemia, particularly tachycardia. Nonselective β-blockers may potentiate insulin-induced hypoglycemia and delay recovery of serum glucose levels. It is not known whether nebivolol has these effects. Advise patients subject to spontaneous hypoglycemia and diabetic patients receiving insulin or oral hypoglycemic agents about these possibilities.
Thyrotoxicosis
β-blockers may mask clinical signs of hyperthyroidism, such as tachycardia. Abrupt withdrawal of β-blockers may be followed by an exacerbation of the symptoms of hyperthyroidism or may precipitate a thyroid storm.
Peripheral Vascular Disease
β-blockers can precipitate or aggravate symptoms of arterial insufficiency in patients with peripheral vascular disease.
Non-dihydropyridine Calcium Channel Blockers
Because of significant negative inotropic and chronotropic effects in patients treated with β-blockers and calcium channel blockers of the verapamil and diltiazem type, monitor the ECG and blood pressure in patients treated concomitantly with these agents.
Use with CYP2D6 Inhibitors
Nebivolol exposure increases with inhibition of CYP2D6 . The dose of Nebivolol may need to be reduced.
Impaired Renal Function
Renal clearance of nebivolol is decreased in patients with severe renal impairment. Nebivolol has not been studied in patients receiving dialysis.
Impaired Hepatic Function
Metabolism of nebivolol is decreased in patients with moderate hepatic impairment. Nebivolol has not been studied in patients with severe hepatic impairment.
Risk of Anaphylactic Reactions
While taking β-blockers, patients with a history of severe anaphylactic reactions to a variety of allergens may be more reactive to repeated accidental, diagnostic, or therapeutic challenge. Such patients may be unresponsive to the usual doses of epinephrine used to treat allergic reactions.
Pheochromocytoma
In patients with known or suspected pheochromocytoma, initiate an α-blocker prior to the use of any β-blocker. |clinicalTrials=Nebivolol has been evaluated for safety in patients with hypertensionand in patients with heart failure. The observed adverse reaction profile was consistent with the pharmacology of the drug and the health status of the patients in the clinical trials. Adverse reactions reported for each of these patient populations are provided below. Excluded are adverse reactions considered too general to be informative and those not reasonably associated with the use of the drug because they were associated with the condition being treated or are very common in the treated population.
The data described below reflect worldwide clinical trial exposure to Nebivolol in 6545 patients, including 5038 patients treated for hypertensionand the remaining 1507 subjects treated for other cardiovascular diseases. Doses ranged from 0.5 mg to 40 mg. Patients received Nebivolol for up to 24 months, with over 1900 patients treated for at least 6 months, and approximately 1300 patients for more than one year.
HYPERTENSION: In placebo-controlled clinical trials comparing Nebivolol with placebo, discontinuation of therapy due to adverse reactions was reported in 2.8% of patients treated with nebivolol and 2.2% of patients given placebo. The most common adverse reactions that led to discontinuation of Nebivolol were headache (0.4%), nausea (0.2%) and bradycardia (0.2%).
Table 1 lists treatment-emergent adverse reactions that were reported in three 12-week, placebo-controlled monotherapy trials involving 1597 hypertensive patients treated with either 5 mg, 10 mg, or 20-40 mg of Nebivolol and 205 patients given placebo and for which the rate of occurrence was at least 1% of patients treated with nebivolol and greater than the rate for those treated with placebo in at least one dose group.
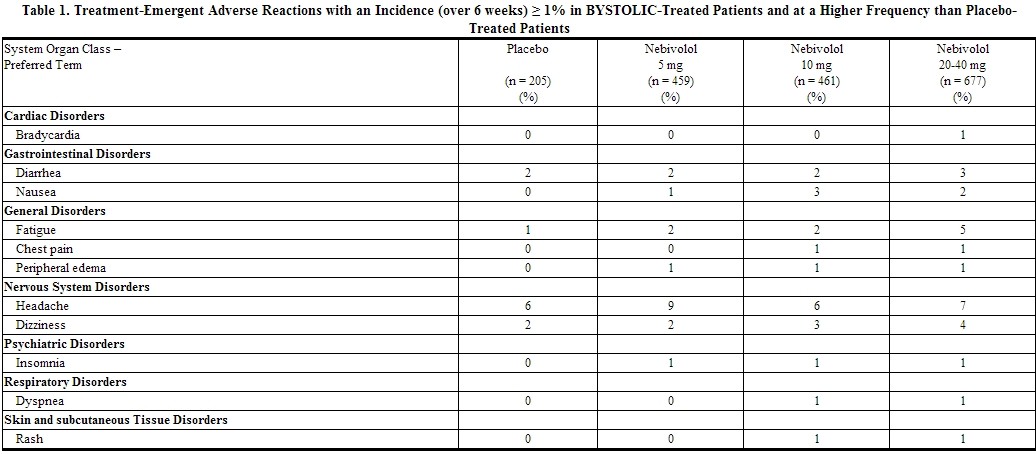
Listed below are other reported adverse reactions with an incidence of at least 1% in the more than 4300 patients treated with Nebivolol in controlled or open-label trials except for those already appearing in Table 1, terms too general to be informative, minor symptoms, or adverse reactions unlikely to be attributable to drug because they are common in the population. These adverse reactions were in most cases observed at a similar frequency in placebo-treated patients in the controlled studies.
Body as a Whole: asthenia.
Gastrointestinal System Disorders: abdominal pain.
Metabolic and Nutritional Disorders: hypercholesterolemia.
Nervous System Disorders: paraesthesia.
Laboratory Abnormalities
In controlled monotherapy trials of hypertensive patients, Nebivolol was associated with an increase in BUN, uric acid, triglycerides and a decrease in HDL cholesterol and platelet count. |postmarketing=The following adverse reactions have been identified from spontaneous reports of Nebivolol received worldwide and have not been listed elsewhere. These adverse reactions have been chosen for inclusion due to a combination of seriousness, frequency of reporting or potential causal connection to Nebivolol. Adverse reactions common in the population have generally been omitted. Because these adverse reactions were reported voluntarily from a population of uncertain size, it is not possible to estimate their frequency or establish a causal relationship to Nebivolol exposure: abnormal hepatic function (including increased AST, ALT and bilirubin), acute pulmonary edema, acute renal failure, atrioventricular block (both second and third degree), bronchospasm, erectile dysfunction, hypersensitivity (including urticaria, allergic vasculitis and rare reports of angioedema), myocardial infarction, pruritus, psoriasis, Raynaud's phenomenon, peripheral ischemia/claudication, somnolence, syncope, thrombocytopenia, various rashes and skin disorders, vertigo, and vomiting. |drugInteractions==== CYP2D6 Inhibitors===
Use caution when Nebivolol is co-administered with CYP2D6 inhibitors (quinidine, propafenone, fluoxetine, paroxetine, etc.).
Hypotensive Agents
Do not use Nebivolol with other β-blockers. Closely monitor patients receiving catecholamine-depleting drugs, such as reserpine or guanethidine, because the added β-blocking action of Nebivolol may produce excessive reduction of sympathetic activity. In patients who are receiving Nebivolol and clonidine, discontinue Nebivolol for several days before the gradual tapering of clonidine.
Digitalis Glycosides
Both digitalis glycosides and β-blockers slow atrioventricular conduction and decrease heart rate. Concomitant use can increase the risk of bradycardia.
Calcium Channel Blockers
Nebivolol can exacerbate the effects of myocardial depressants or inhibitors of AV conduction, such as certain calcium antagonists (particularly of the phenylalkylamine [verapamil] and benzothiazepine [diltiazem] classes), or antiarrhythmic agents, such as disopyramide.
Drugs that inhibit CYP2D6 can be expected to increase plasma levels of nebivolol. When Nebivolol is co-administered with an inhibitor or an inducer of this enzyme, monitor patients closely and adjust the nebivolol dose according to blood pressure response. In vitro studies have demonstrated that at therapeutically relevant concentrations, d- and l-nebivolol do not inhibit any cytochrome P450 pathways.
Digoxin: Concomitant administration of Nebivolol (10 mg once daily) and digoxin (0.25 mg once daily) for 10 days in 14 healthy adult individuals resulted in no significant changes in the pharmacokinetics of digoxin or nebivolol.
Warfarin: Administration of Nebivolol (10 mg once daily for 10 days) led to no significant changes in the pharmacokinetics of nebivolol or R- or S-warfarin following a single 10 mg dose of warfarin. Similarly, nebivolol has no significant effects on the anticoagulant activity of warfarin, as assessed by Prothrombin time and INR profiles from 0 to 144 hours after a single 10 mg warfarin dose in 12 healthy adult volunteers.
Diuretics: No pharmacokinetic interactions were observed in healthy adults between nebivolol (10 mg daily for 10 days) and furosemide (40 mg single dose),hydrochlorothiazide (25 mg once daily for 10 days), or spironolactone (25 mg once daily for 10 days).
Ramipril: Concomitant administration of Nebivolol (10 mg once daily) and ramipril (5 mg once daily) for 10 days in 15 healthy adult volunteers produced no pharmacokinetic interactions.
Losartan: Concomitant administration of Nebivolol (10 mg single dose) and losartan (50 mg single dose) in 20 healthy adult volunteers did not result in pharmacokinetic interactions.
Fluoxetine: Fluoxetine, a CYP2D6 inhibitor, administered at 20 mg per day for 21 days prior to a single 10 mg dose of nebivolol to 10 healthy adults, led to an 8-fold increase in the AUC and 3-fold increase in Cmax for d-nebivolol.
Histamine-2 Receptor Antagonists: The pharmacokinetics of nebivolol (5 mg single dose) were not affected by the co-administration of ranitidine (150 mg twice daily). Cimetidine (400 mg twice daily) causes a 23% increase in the plasma levels of d-nebivolol.
Charcoal: The pharmacokinetics of nebivolol (10 mg single dose) were not affected by repeated co-administration (4, 8, 12, 16, 22, 28, 36, and 48 hours after nebivolol administration) of activated charcoal (Actidose-Aqua®).
Sildenafil: The co-administration of nebivolol and sildenafil decreased AUC and Cmax of sildenafil by 21 and 23% respectively. The effect on the Cmax and AUC for d-nebivolol was also small (< 20%). The effect on vital signs (e.g., pulse and blood pressure) was approximately the sum of the effects of sildenafil and nebivolol.
Other Concomitant Medications: Utilizing population pharmacokinetic analyses, derived from hypertensive patients, the following drugs were observed not to have an effect on the pharmacokinetics of nebivolol: acetaminophen, acetylsalicylic acid, atorvastatin, esomeprazole, ibuprofen, levothyroxine sodium, metformin, sildenafil, simvastatin, or tocopherol.
|FDAPregCat=C
|useInPregnancyFDA=Decreased pup body weights occurred at 1.25 and 2.5 mg/kg in rats, when exposed during the perinatal period (late gestation, parturition and lactation). At 5 mg/kg and higher doses (1.2 times the MRHD), prolonged gestation, dystocia and reduced maternal care were produced with corresponding increases in late fetal deaths and stillbirths and decreased birth weight, live litter size and pup survival. Insufficient numbers of pups survived at 5 mg/kg to evaluate the offspring for reproductive performance.
In studies in which pregnant rats were given nebivolol during organogenesis, reduced fetal body weights were observed at maternally toxic doses of 20 and 40 mg/kg/day (5 and 10 times the MRHD), and small reversible delays in sternal and thoracic ossification associated with the reduced fetal body weights and a small increase in resorption occurred at 40 mg/kg/day (10 times the MRHD). No adverse effects on embryo-fetal viability, sex, weight or morphology were observed in studies in which nebivolol was given to pregnant rabbits at doses as high as 20 mg/kg/day (10 times the MRHD). |useInLaborDelivery=Nebivolol caused prolonged gestation and dystocia at doses ≥ 5 mg/kg in rats (1.2 times the MRHD). These effects were associated with increased fetal deaths and stillborn pups, and decreased birth weight, live litter size and pup survival rate, events that occurred only when nebivolol was given during the perinatal period (late gestation, parturition and lactation).
No studies of nebivolol were conducted in pregnant women. Use Nebivolol during pregnancy only if the potential benefit justifies the potential risk to the fetus. |useInNursing=Studies in rats have shown that nebivolol or its metabolites cross the placental barrier and are excreted in breast milk. It is not known whether this drug is excreted in human milk.
Because of the potential for β-blockers to produce serious adverse reactions in nursing infants, especially bradycardia, Nebivolol is not recommended during nursing. |useInPed=Safety and effectiveness in pediatric patients have not been established. Pediatric studies in ages newborn to 18 years old have not been conducted because of incomplete characterization of developmental toxicity and possible adverse effects on long-term fertility. |useInGeri=Of the 2800 patients in the U.S. sponsored placebo-controlled clinical hypertension studies, 478 patients were 65 years of age or older. No overall differences in efficacy or in the incidence of adverse events were observed between older and younger patients. |othersTitle=Heart Failure |useInOthers=In a placebo-controlled trial of 2128 patients (1067 Nebivolol, 1061 placebo) over 70 years of age with chronic heart failure receiving a maximum dose of 10 mg per day for a median of 20 months, no worsening of heart failure was reported with nebivolol compared to placebo. However, if heart failure worsens consider discontinuation of Nebivolol. |administration=Oral |monitoring=FDA Package Insert for Nebivolol contains no information regarding drug monitoring . |IVCompat=There is limited information about the IV Compatibility. |overdose=In clinical trials and worldwide postmarketing experience there were reports of Nebivolol overdose. The most common signs and symptoms associated with Nebivolol overdosage are bradycardiaand hypotension. Other important adverse reactions reported with Nebivolol overdose include cardiac failure, dizziness, hypoglycemia, fatigue and vomiting. Other adverse reactions associated with β-blocker overdose include bronchospasm and heart block.
The largest known ingestion of Nebivolol worldwide involved a patient who ingested up to 500 mg of Nebivolol along with several 100 mg tablets of acetylsalicylic acid in a suicide attempt. The patient experienced hyperhydrosis, pallor, depressed level of consciousness, hypokinesia, hypotension, sinus bradycardia, hypoglycemia, hypokalemia, respiratory failure and vomiting. The patient recovered.
Because of extensive drug binding to plasma proteins, hemodialysis is not expected to enhance nebivolol clearance.
If overdose occurs, provide general supportive and specific symptomatic treatment. Based on expected pharmacologic actions and recommendations for other β-blockers, consider the following general measures, including stopping Nebivolol, when clinically warranted:
Bradycardia: Administer IV atropine. If the response is inadequate, isoproterenol or another agent with positive chronotropic properties may be given cautiously. Under some circumstances, transthoracic or transvenous pacemaker placement may be necessary.
Hypotension: Administer IV fluids and vasopressors. Intravenous glucagon may be useful.
Heart Block (second or third degree): Monitor and treat with isoproterenol infusion. Under some circumstances, transthoracic or transvenous pacemaker placement may be necessary.
Congestive Heart Failure: Initiate therapy with digitalis glycoside and diuretics. In certain cases, consider the use of inotropic and vasodilating agents.
Bronchospasm: Administer bronchodilator therapy such as a short acting inhaled β2-agonist and/or aminophylline.
Hypoglycemia: Administer IV glucose. Repeated doses of IV glucose or possibly glucagon may be required.
Supportive measures should continue until clinical stability is achieved. The half-life of low doses of nebivolol is 12-19 hours.
Call the National Poison Control Center (800-222-1222) for the most current information on β-blocker overdose treatment.
|drugBox={{Drugbox2
| Verifiedfields = changed
| Watchedfields = changed
| verifiedrevid = 462258880
| IUPAC_name = 1-(6-Fluorochroman-2-yl)-{[2-(6-fluorochroman-2-yl)-2-hydroxy-ethyl]amino}ethanol
OR
2,2'-Azanediylbis(1-(6-fluorochroman-2-yl)ethanol)
OR
1-(6-Fluoro-3,4-dihydro-2H-1-benzopyran-2-yl)-2-{[2-(6-fluoro-3,4-dihydro-2H-1-benzopyran-2-yl)-2-hydroxyethyl]amino}ethan-1-ol
| image = Nebivolol.png
| image2 = Nebivolol ball-and-stick.png
| tradename = Nebilet, Nebivolol
| Drugs.com = Monograph
| MedlinePlus = a608029
| licence_US = Nebivolol
| pregnancy_US = C
| legal_UK = POM
| legal_US = Rx-only
| routes_of_administration = Oral
| protein_bound = 98% | metabolism = Hepatic (CYP2D6-mediated) | elimination_half-life = 10 hours | excretion = Renal and fecal
| CAS_number_Ref =
| CAS_number = 99200-09-6
| ATC_prefix = C07
| ATC_suffix = AB12
| PubChem = 71301
| DrugBank_Ref =
| DrugBank = DB04861
| ChemSpiderID_Ref =
| ChemSpiderID = 64421
| UNII_Ref =
| UNII = 030Y90569U
| KEGG_Ref =
| KEGG = D05127
| ChEMBL_Ref =
| ChEMBL = 434394
| C=22 | H=25 | F=2 | N=1 | O=4
| molecular_weight = 405.435 g/mol
| smiles = Fc4cc1c(OC(CC1)C(O)CNCC(O)C3Oc2ccc(F)cc2CC3)cc4
| InChI = 1/C22H25F2NO4/c23-15-3-7-19-13(9-15)1-5-21(28-19)17(26)11-25-12-18(27)22-6-2-14-10-16(24)4-8-20(14)29-22/h3-4,7-10,17-18,21-22,25-27H,1-2,5-6,11-12H2
| InChIKey = KOHIRBRYDXPAMZ-UHFFFAOYAH
| StdInChI_Ref =
| StdInChI = 1S/C22H25F2NO4/c23-15-3-7-19-13(9-15)1-5-21(28-19)17(26)11-25-12-18(27)22-6-2-14-10-16(24)4-8-20(14)29-22/h3-4,7-10,17-18,21-22,25-27H,1-2,5-6,11-12H2
| StdInChIKey_Ref =
| StdInChIKey = KOHIRBRYDXPAMZ-UHFFFAOYSA-N
}}
|mechAction=The mechanism of action of the antihypertensive response of Nebivolol has not been definitively established. Possible factors that may be involved include: (1) decreased heart rate, (2) decreased myocardial contractility, (3) diminution of tonic sympathetic outflow to the periphery from cerebral vasomotor centers, (4) suppression of renin activity and (5) vasodilation and decreased peripheral vascular resistance.
|structure=The chemical name for the active ingredient in Nebivolol (nebivolol) tablets is (1RS,1'RS)-1,1'-[(2RS,2'SR)-bis(6-fluoro-3,4-dihydro-2H-1-benzopyran-2-yl)]- 2,2'-iminodiethanol hydrochloride. Nebivolol is a racemate composed of d-Nebivolol and l-Nebivolol with the stereochemical designations of [SRRR]-nebivolol and [RSSS]-nebivolol, respectively. Nebivolol's molecular formula is (C22H25F2NO4•HCl) with the following structural formula:
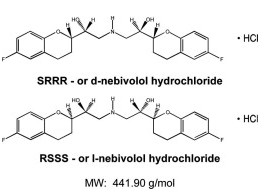
Nebivolol hydrochloride is a white to almost white powder that is soluble in methanol, dimethylsulfoxide, and N,N-dimethylformamide, sparingly soluble in ethanol, propylene glycol, and polyethylene glycol, and very slightly soluble in hexane, dichloromethane, and methylbenzene. |PD=Nebivolol is metabolized by a number of routes, including glucuronidation and hydroxylation by CYP2D6. The active isomer (d-nebivolol) has an effective half-life of about 12 hours in CYP2D6 extensive metabolizers (most people), and 19 hours in poor metabolizers and exposure to d-nebivolol is substantially increased in poor metabolizers. This has less importance than usual, however, because the metabolites, including the hydroxyl metabolite and glucuronides (the predominant circulating metabolites), contribute to β-blocking activity.
Plasma levels of d–nebivolol increase in proportion to dose in extensive metabolizers (EMs) and poor metabolizers (PMs) for doses up to 20mg. Exposure to l-nebivolol is higher than to d-nebivolol but l-nebivolol contributes little to the drug's activity as d-nebivolol's beta receptor affinity is > 1000-fold higher than l-nebivolol. For the same dose, PMs attain a 5-fold higher Cmax and 10-fold higher AUC of d-nebivolol than do EMs. d-Nebivolol accumulates about 1.5-fold with repeated once-daily dosing in EMs. |PK=====Absorption====
Absorption of Nebivolol is similar to an oral solution. The absolute bioavailability has not been determined.
Mean peak plasma nebivolol concentrations occur approximately 1.5 to 4 hours post-dosing in EMs and PMs.
Food does not alter the pharmacokinetics of nebivolol. Under fed conditions, nebivolol glucuronides are slightly reduced. Nebivolol may be administered without regard to meals.
Distribution
The in vitro human plasma protein binding of nebivolol is approximately 98%, mostly to albumin, and is independent of nebivolol concentrations.
Metabolism
Nebivolol is predominantly metabolized via direct glucuronidation of parent and to a lesser extent via N-dealkylation and oxidation via cytochrome P450 2D6. Its stereospecific metabolites contribute to the pharmacologic activity.
Elimination
After a single oral administration of 14C-nebivolol, 38% of the dose was recovered in urine and 44% in feces for EMs and 67% in urine and 13% in feces for PMs. Essentially all nebivolol was excreted as multiple oxidative metabolites or their corresponding glucuronide conjugates.
Pharmacokinetics in Special Populations
Hepatic Disease
d-Nebivolol peak plasma concentration increased 3-fold, exposure (AUC) increased 10-fold, and the apparent clearance decreased by 86% in patients with moderate hepatic impairment (Child-Pugh Class B). No formal studies have been performed in patients with severe hepatic impairment and nebivolol should be contraindicated for these patients.
Renal Disease
The apparent clearance of nebivolol was unchanged following a single 5 mg dose of Nebivolol in patients with mild renal impairment (ClCr 50 to 80 mL/min, n=7), and it was reduced negligibly in patients with moderate (ClCr 30 to 50 mL/min, n=9), but clearance was reduced by 53% in patients with severe renal impairment (ClCr <30 mL/min, n=5). No studies have been conducted in patients on dialysis. |nonClinToxic====Carcinogenesis, Mutagenesis, Impairment of Fertility===
In a two-year study of nebivolol in mice, a statistically significant increase in the incidence of testicular Leydig cell hyperplasia and adenomas was observed at 40 mg/kg/day (5 times the maximally recommended human dose of 40 mg on a mg/m2 basis). Similar findings were not reported in mice administered doses equal to approximately 0.3 or 1.2 times the maximum recommended human dose. No evidence of a tumorigenic effect was observed in a 24-month study in Wistar rats receiving doses of nebivolol 2.5, 10 and 40 mg/kg/day (equivalent to 0.6, 2.4, and 10 times the maximally recommended human dose). Co-administration of dihydrotestosterone reduced blood LH levels and prevented the Leydig cell hyperplasia, consistent with an indirect LH-mediated effect of nebivolol in mice and not thought to be clinically relevant in man.
A randomized, double-blind, placebo- and active-controlled, parallel-group study in healthy male volunteers was conducted to determine the effects of nebivolol on adrenal function, luteinizing hormone, and testosterone levels. This study demonstrated that 6 weeks of daily dosing with 10 mg of nebivolol had no significant effect on ACTH-stimulated mean serum cortisol AUC0-120 min, serum LH, or serum total testosterone.
Effects on spermatogenesis were seen in male rats and mice at ≥ 40 mg/kg/day (10 and 5 times the MRHD, respectively). For rats the effects on spermatogenesis were not reversed and may have worsened during a four week recovery period. The effects of nebivolol on sperm in mice, however, were partially reversible.
Mutagenesis: Nebivolol was not genotoxic when tested in a battery of assays (Ames, in vitro mouse lymphoma TK+/-, in vitro human peripheral lymphocyte chromosome aberration, in vivo Drosophila melanogaster sex-linked recessive lethal, and in vivo mouse bone marrow micronucleus tests). |clinicalStudies====Hypertension===
The antihypertensive effectiveness of Nebivolol as monotherapy has been demonstrated in three randomized, double-blind, multi-center, placebo-controlled trials at doses ranging from 1.25 to 40 mg for 12 weeks (Studies 1, 2, and 3). A fourth placebo-controlled trial demonstrated additional antihypertensive effects of Nebivolol at doses ranging from 5 to 20 mg when administered concomitantly with up to two other antihypertensive agents (ACE inhibitors, angiotensin II receptor antagonists, and thiazide diuretics) in patients with inadequate blood pressure control.
The three monotherapy trials included a total of 2016 patients (1811 Nebivolol, 205 placebo) with mild to moderate hypertension who had baseline diastolic blood pressures (DBP) of 95 to 109 mmHg. Patients received either Nebivolol or placebo once daily for twelve weeks. Two of these monotherapy trials (Studies 1 and 2) studied 1716 patients in the general hypertensive population with a mean age of 54 years, 55% males, 26% non-Caucasians, 7% diabetics and 6% genotyped as PMs. The third monotherapy trial (Study 3) studied 300 Black patients with a mean age of 51 years, 45% males, 14% diabetics, and 3% as PMs.
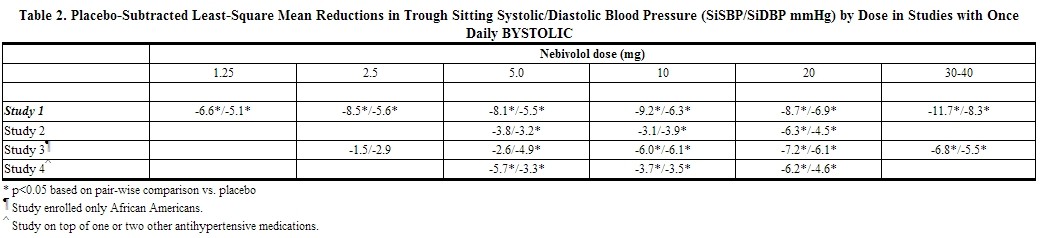
Placebo-subtracted blood pressure reductions by dose for each study are presented in Table 2. Most studies showed increasing response to doses above 5 mg. |{ |-
|
|- |}
Study 4 enrolled 669 patients with a mean age of 54 years, 55% males, 54% Caucasians, 29% Blacks, 15% Hispanics, 1% Asians, 14% diabetics, and 5% PMs. Nebivolol, 5 mg to 20 mg, administered once daily concomitantly with stable doses of up to two other antihypertensive agents (ACE inhibitors, angiotensin II receptor antagonists, and thiazide diuretics) resulted in significant additional antihypertensive effects over placebo compared to baseline blood pressure.
Effectiveness was similar in subgroups analyzed by age and sex. Effectiveness was established in Blacks, but as monotherapy the magnitude of effect was somewhat less than in Caucasians.
The blood pressure lowering effect of Nebivolol was seen within two weeks of treatment and was maintained over the 24-hour dosing interval.
There are no trials of Nebivolol demonstrating reductions in cardiovascular risk in patients with hypertension, but at least one pharmacologically similar drug has demonstrated such benefits. |howSupplied=Nebivolol is available as tablets for oral administration containing nebivolol hydrochloride equivalent to 2.5, 5, 10, and 20 mg of nebivolol.
Nebivolol tablets are triangular-shaped, biconvex, unscored, differentiated by color and are engraved with “FL” on one side and the number of mg (2 ½, 5, 10, or 20) on the other side. Nebivolol tablets are supplied in the following strengths and package configurations: |storage=Store at 20° to 25°C (68° to 77°F) [see USP for Controlled Room Temperature].
Dispense in a tight, light-resistant container as defined in the USP using a child-resistant closure. |fdaPatientInfo====Patient Advice===
Advise patients to take Nebivolol regularly and continuously, as directed. Nebivolol can be taken with or without food. If a dose is missed, take the next scheduled dose only (without doubling it). Do not interrupt or discontinue Nebivolol without consulting the physician.
Patients should know how they react to this medicine before they operate automobiles, use machinery, or engage in other tasks requiring alertness.
Advise patients to consult a physician if any difficulty in breathing occurs, or if they develop signs or symptoms of worsening congestive heart failure such as weight gain or increasing shortness of breath, or excessive bradycardia.
Caution patients subject to spontaneous hypoglycemia, or diabetic patients receiving insulin or oral hypoglycemic agents, that β-blockers may mask some of the manifestations of hypoglycemia, particularly tachycardia. |alcohol=FDA Package Insert for Abcixmab contains no information regarding Adverse Reactions. |brandNames=BYSTOLIC }} {{#subobject:
|Page Name=Nebivolol |Pill Name=Nebivolol_2.5 mg_NDC 0456-1402.JPG |Drug Name=Nebivolol 2.5 MG Oral Tablet |Pill Ingred=silicon dioxide, croscarmellose sodium, d&c red no. 27, fd&c blue no. 2, fd&c yellow no. 6, hypromelloses, lactose monohydrate, magnesium stearate, cellulose, microcrystalline, starch, corn, polysorbate 80, sodium lauryl sulfate|+sep=; |Pill Imprint=2;1;2;FL |Pill Dosage=2.5 mg |Pill Color=Blue|+sep=; |Pill Shape=Triangular |Pill Size (mm)=8.00 |Pill Scoring=1 |Pill Image= |Drug Author=Forest laboratories, Inc. |NDC=0456-1402
}}
{{#subobject:
|Page Name=Nebivolol |Pill Name=Nebivolol_5 mg_NDC 0456-1405.JPG |Drug Name=Nebivolol 5 MG Oral Tablet |Pill Ingred=silicon dioxide, croscarmellose sodium, d&c red no. 27, fd&c blue no. 2, fd&c yellow no. 6, hypromelloses, lactose monohydrate, magnesium stearate, cellulose, microcrystalline, starch, corn, polysorbate 80, sodium lauryl sulfate|+sep=; |Pill Imprint=5;FL |Pill Dosage=5 mg |Pill Color=Brown|+sep=; |Pill Shape=Triangular |Pill Size (mm)=10.00 |Pill Scoring=1 |Pill Image= |Drug Author=Forest laboratories, Inc. |NDC=0456-1405
}}
{{#subobject:
|Page Name=Nebivolol |Pill Name=Nebivolol_10 mg_NDC 0456-1410.JPG |Drug Name=Nebivolol 10 MG Oral Tablet |Pill Ingred=silicon dioxide, croscarmellose sodium, d&c red no. 27, fd&c blue no. 2, fd&c yellow no. 6, hypromelloses, lactose monohydrate, magnesium stearate, cellulose, microcrystalline, starch, corn, polysorbate 80, sodium lauryl sulfate|+sep=; |Pill Imprint=10;FL |Pill Dosage=10 mg |Pill Color=Purple|+sep=; |Pill Shape=Triangular |Pill Size (mm)=10.00 |Pill Scoring=1 |Pill Image= |Drug Author=Forest laboratories, Inc. |NDC=0456-1410
}}
{{#subobject:
|Page Name=Nebivolol |Pill Name=Nebivolol_20 mg_NDC 0456-1420.JPG |Drug Name= |Pill Ingred=silicon dioxide, croscarmellose sodium, d&c red no. 27, fd&c blue no. 2, fd&c yellow no. 6, hypromelloses, lactose monohydrate, magnesium stearate, cellulose, microcrystalline, starch, corn, polysorbate 80, sodium lauryl sulfate|+sep=; |Pill Imprint=20;FL |Pill Dosage=20 mg |Pill Color=Blue|+sep=; |Pill Shape=Triangular |Pill Size (mm)=10.00 |Pill Scoring=1 |Pill Image= |Drug Author=Forest laboratories, Inc. |NDC=0456-1420
}}
{{#subobject:
|Label Page=Nebivolol |Label Name=Nebivolol06.jpg
}}
{{#subobject:
|Label Page=Nebivolol |Label Name=Nebivolol07.jpg
}}
{{#subobject:
|Label Page=Nebivolol |Label Name=Nebivolol08.jpg
}}
{{#subobject:
|Label Page=Nebivolol |Label Name=Nebivolol09.jpg
}}
{{#subobject:
|Label Page=Nebivolol |Label Name=Nebivolol_panel-01.png
}}
{{#subobject:
|Label Page=Nebivolol |Label Name=Nebivolol_panel-02.png
}}
{{#subobject:
|Label Page=Nebivolol |Label Name=Nebivolol_panel-03.png
}}
{{#subobject:
|Label Page=Nebivolol |Label Name=Nebivolol_panel-04.png
}}
- ↑ Flather MD, Shibata MC, Coats AJ, Van Veldhuisen DJ, Parkhomenko A, Borbola J; et al. (2005). "Randomized trial to determine the effect of nebivolol on mortality and cardiovascular hospital admission in elderly patients with heart failure (SENIORS)". Eur Heart J. 26 (3): 215–25. doi:10.1093/eurheartj/ehi115. PMID 15642700.
- ↑ Dobre D, van Veldhuisen DJ, Mordenti G, Vintila M, Haaijer-Ruskamp FM, Coats AJ; et al. (2007). "Tolerability and dose-related effects of nebivolol in elderly patients with heart failure: data from the Study of the Effects of Nebivolol Intervention on Outcomes and Rehospitalisation in Seniors with Heart Failure (SENIORS) trial". Am Heart J. 154 (1): 109–15. doi:10.1016/j.ahj.2007.03.025. PMID 17584562.
- ↑ Schellenberg R, Lichtenthal A, Wöhling H, Graf C, Brixius K (2008). "Nebivolol and metoprolol for treating migraine: an advance on beta-blocker treatment?". Headache. 48 (1): 118–25. doi:10.1111/j.1526-4610.2007.00785.x. PMID 18184294.