IncobotulinumtoxinA
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Aparna Vuppala, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING
See full prescribing information for complete Boxed Warning.
DISTANT SPREAD OF TOXIN EFFECT
|
Overview
IncobotulinumtoxinA is a musculoskeletal agent that is FDA approved for the treatment of adults with cervical dystonia, blepharospasm, indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity. There is a Black Box Warning for this drug as shown here. Common adverse reactions include injection site pain, diarrhea , dysphagia , xerostomia, muscle weakness, musculoskeletal pain, neck pain, headache, dry eye, ptosis of eyelid, Visual impairment, dyspnea, nasopharyngitis, respiratory tract infection.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Cervical Dystonia
- IncobotulinumtoxinA is indicated for the treatment of adults with cervical dystonia to decrease the severity of abnormal head position and neck pain in both botulinum toxin-naïve and previously treated patients.
Blepharospasm
- IncobotulinumtoxinA is indicated for the treatment of adults with blepharospasm who were previously treated with onabotulinumtoxinA (Botox).
Glabellar Lines
- IncobotulinumtoxinA is indicated for the temporary improvement in the appearance of moderate to severe glabellar lines associated with corrugator and/or procerus muscle activity in adult patients.
Dosing Information
- The potency Units of incobotulinumtoxinA for injection are specific to the preparation and assay method utilized. They are not interchangeable with other preparations of botulinum toxin products and, therefore, Units of biological activity of incobotulinumtoxinA cannot be compared to or converted into Units of any other botulinum toxin products assessed with any other specific assay method
Cervical Dystonia
- The recommended initial total dose of incobotulinumtoxinA for cervical dystonia is 120 Units. In a placebo-controlled trial utilizing initial incobotulinumtoxinA doses of 120 Units and 240 Units, no meaningful difference in effectiveness was demonstrated between the doses . In previously treated patients, their past dose, response to treatment, duration of effect, and adverse event history should be taken into consideration when determining the incobotulinumtoxinA dose.
- In the treatment of cervical dystonia, incobotulinumtoxinA is usually injected into the sternocleidomastoid, levator scapulae, splenius capitis, scalenus, and/or the trapezius muscle(s). This list is not exhaustive, as any of the muscles responsible for controlling head position may require treatment . The dose and number of injection sites in each treated muscle should be individualized based on the number and location of the muscle(s) to be treated, the degree of spasticity/dystonia, muscle mass, body weight, and response to any previous botulinum toxin injections.
- The frequency of incobotulinumtoxinA repeat treatments should be determined by clinical response, but should generally be no more frequent than every 12 weeks
Blepharospasm
- The recommended initial total dose of incobotulinumtoxinA should be the same dose as the patient's previous treatment of onabotulinumtoxinA (Botox), although responses to incobotulinumtoxinA and onabotulinumtoxinA (Botox) may differ in individual patients. In a placebo-controlled trial in which patients were dosed with the same number of Units as they had received previously with onabotulinumtoxinA (Botox), the mean dose per eye was about 33 Units (range 10-50 Units), and the mean number of injections per eye was 6. The maximum dose per eye in the controlled trials was 50 Units, with a range of 10-50 Units. In the controlled trial, few patients received a total dose of greater than 75 Units.
- If the previous dose of Botox is not known, the initial dose of incobotulinumtoxinA should be between 1.25-2.5 Units/injection site.
- The total initial dose of incobotulinumtoxinA in both eyes should not exceed 70 Units (35 Units/eye).
- The number and location of injection sites should be based on the severity of blepharospasm, and previous dose and response to onabotulinumtoxinA (Botox) injections. Subsequent dosing should be tailored to the individual patient, based on response, up to a maximum dose of 35 Units per eye . IncobotulinumtoxinA dosing has not been established in patients with blepharospasm who have not been previously treated with onabotulinumtoxinA (Botox).
- The frequency of incobotulinumtoxinA repeat treatments should be determined by clinical response but should generally be no more frequent than every 12 weeks
Glabellar Lines
- The total recommended incobotulinumtoxinA dose is 20 Units per treatment session divided into five equal intramuscular injections of 4 Units each. The five injection sites are: two injections in each corrugator muscle and one injection in the procerus muscle.
- Retreatment with incobotulinumtoxinA should be administered no more frequently than every three months.
Special Populations
- The safety and effectiveness of incobotulinumtoxinA in the treatment of cervical dystonia, blepharospasm, and glabellar lines in patients below 18 years of age have not been assessed
Preparation and Reconstitution Technique
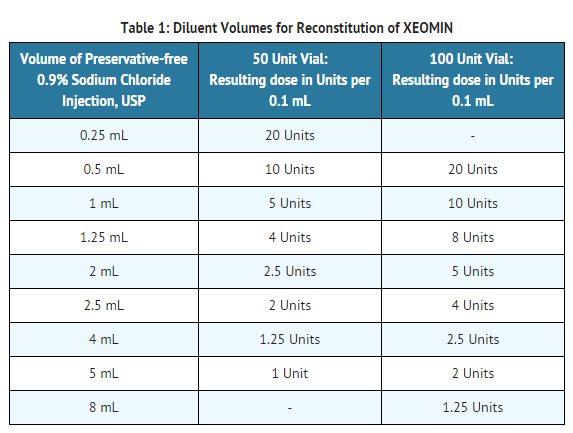
- Prior to injection, reconstitute each vial of incobotulinumtoxinA with sterile, preservative-free 0.9% Sodium Chloride Injection, USP. Draw up an appropriate amount of preservative-free 0.9% Sodium Chloride Injection, USP into a syringe (see TABLE 1). Clean the exposed portion of the rubber stopper of the vial with alcohol (70%) prior to insertion of the needle. Gently inject the saline solution into the vial. If the vacuum does not pull the solvent into the vial, then incobotulinumtoxinA must be discarded. Gently mix incobotulinumtoxinA with the saline by rotating the vial. Reconstituted incobotulinumtoxinA is a clear, colorless solution free of particulate matter. IncobotulinumtoxinA should not be used if the reconstituted solution has a cloudy appearance or contains floccular or particulate matter.
- Diluent volumes for reconstitution of incobotulinumtoxinA are indicated in Table 1.
- Reconstituted incobotulinumtoxinA solution should be administered within 24 hours after dilution. During this time period, reconstituted incobotulinumtoxinA should be stored in a refrigerator 2-8°C (36-46°F)
Administration
- Reconstituted incobotulinumtoxinA is intended for intramuscular injection only. After reconstitution, incobotulinumtoxinA should be used for only one injection session and for only one patient.
- If proposed injection sites are marked with a pen, the product must not be injected through the pen marks; otherwise a permanent tattooing effect may occur.
- The number of injection sites is dependent upon the size of the muscle to be treated and the volume of reconstituted incobotulinumtoxinA injected.
- IncobotulinumtoxinA should be injected carefully when injected at sites close to sensitive structures, such as the carotid artery, lung apices and esophagus. Before administering incobotulinumtoxinA , the physician should be familiar with the patient's anatomy and any anatomic alterations, e.g., due to prior surgical procedures.
Cervical Dystonia
- A suitable sterile needle (e.g., 26-gauge (0.45 mm diameter), 37 mm length for superficial muscles; or 22-gauge (0.70 mm diameter), 75 mm length for injections into deeper muscles) should be used in the administration in the treatment of cervical dystonia.
- Localization of the involved muscles with electromyographic guidance or nerve stimulation techniques may be useful.
Blepharospasm
- A suitable sterile needle (e.g., 26-gauge (0.45 mm diameter), 37 mm length for superficial muscles; or 22-gauge (0.70 mm diameter), 75 mm length for injections into deeper muscles) should be used in the administration in the treatment of blepharospasm.
Glabellar Lines
- A suitable sterile needle 30-33 gauge (0.3-0.2 mm diameter), 13 mm length should be used in the administration in the treatment of glabellar lines.
Monitoring to Assess Effectiveness
- The median first onset of incobotulinumtoxinA effect occurs within seven days after injection. The typical duration of effect of each treatment is up to 3 months; however, the effect may last significantly longer, or shorter, in individual patients.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of IncobotulinumtoxinA in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of IncobotulinumtoxinA in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of IncobotulinumtoxinA in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of IncobotulinumtoxinA in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of IncobotulinumtoxinA in pediatric patients.
Contraindications
Hypersensitivity
- Use in patients with a known hypersensitivity to the active substance botulinum neurotoxin type A, or to any of the excipients (human albumin, sucrose), could lead to a life-threatening allergic reaction. incobotulinumtoxinA is contraindicated in patients with known hypersensitivity to any botulinum toxin preparation or to any of the components in the formulation
Infection at Injection Site
- Use in patients with an infection at the injection site could lead to severe local or disseminated infection. IncobotulinumtoxinA is contraindicated in the presence of infection at the proposed injection site(s).
Warnings
|
WARNING
See full prescribing information for complete Boxed Warning.
DISTANT SPREAD OF TOXIN EFFECT
|
Spread of Toxin Effect
- Postmarketing safety data from incobotulinumtoxinA and other approved botulinum toxins suggest that botulinum toxin effects may, in some cases, be observed beyond the site of local injection. The symptoms are consistent with the mechanism of action of botulinum toxin and may include asthenia, generalized muscle weakness, diplopia, blurred vision, ptosis, dysphagia, dysphonia, dysarthria, urinary incontinence, and breathing difficulties. These symptoms have been reported hours to weeks after injection. Swallowing and breathing difficulties can be life threatening and there have been reports of death related to the spread of toxin effects. The risk of symptoms is probably greatest in children treated for spasticity but symptoms can occur in adults treated for spasticity and other conditions, and particularly in those patients who have underlying conditions that would predispose them to these symptoms. In unapproved uses, including spasticity in children and adults, and in approved indications, symptoms consistent with spread of toxin effect have been reported at doses comparable to or lower than doses used to treat cervical dystonia.
- Patients or caregivers should be advised to seek immediate medical care if swallowing, speech, or respiratory disorders occur.
Lack of Interchangeability between Botulinum Toxin Products
- The potency Units of incobotulinumtoxinA are specific to the preparation and assay method utilized. They are not interchangeable with the other preparations of botulinum toxin products and, therefore, Units of biological activity of incobotulinumtoxinA cannot be compared to or converted into Units of any other botulinum toxin products assessed with any other specific assay method .
Hypersensitivity Reactions
- Hypersensitivity reactions have been reported with botulinum toxin products (anaphylaxis, serum sickness, urticaria, soft tissue edema, and dyspnea). If serious and/or immediate hypersensitivity reactions occur further injection of incobotulinumtoxinA should be discontinued and appropriate medical therapy immediately instituted.
Dysphagia and Breathing Difficulties in Treatment of Cervical Dystonia
- Treatment with incobotulinumtoxinA and other botulinum toxin products can result in swallowing or breathing difficulties. Patients with pre-existing swallowing or breathing difficulties may be more susceptible to these complications. In most cases, this is a consequence of weakening of muscles in the area of injection that are involved in breathing or swallowing. When distant effects occur, additional respiratory muscles may be involved .
- Deaths as a complication of severe dysphagia have been reported after treatment with botulinum toxin. Dysphagia may persist for several months, and require use of a feeding tube to maintain adequate nutrition and hydration. Aspiration may result from severe dysphagia and is a particular risk when treating patients in whom swallowing or respiratory function is already compromised.
- Treatment of cervical dystonia with botulinum toxins may weaken neck muscles that serve as accessory muscles of ventilation. This may result in critical loss of breathing capacity in patients with respiratory disorders who may have become dependent upon these accessory muscles. There have been post-marketing reports of serious breathing difficulties, including respiratory failure, in patients with cervical dystonia treated with botulinum toxin products.
- Patients with smaller neck muscle mass and patients who require bilateral injections into the sternocleidomastoid muscles have been reported to be at greater risk of dysphagia. In general, limiting the dose injected into the sternocleidomastoid muscle may decrease the occurrence of dysphagia.
- Patients treated with botulinum toxin may require immediate medical attention should they develop problems with swallowing, speech or respiratory disorders. These reactions can occur within hours to weeks after injection with botulinum toxin.
Pre-existing Neuromuscular Disorders and other Special Populations
- Individuals with peripheral motor neuropathic diseases, amyotrophic lateral sclerosis, or neuromuscular junctional disorders (e.g., myasthenia gravis or Lambert-Eaton syndrome) should be monitored particularly closely when given botulinum toxin. Patients with neuromuscular disorders may be at increased risk of clinically significant effects including severe dysphagia and respiratory compromise from typical doses of incobotulinumtoxinA .
Corneal Exposure, Corneal Ulceration, and Ectropion in Patients Treated with incobotulinumtoxinA for Blepharospasm
- Reduced blinking from injection of botulinum toxin products in the orbicularis muscle can lead to corneal exposure, persistent epithelial defect and corneal ulceration, especially in patients with VII nerve disorders. Careful testing of corneal sensation in eyes previously operated upon, avoidance of injection into the lower lid area to avoid ectropion, and vigorous treatment of any epithelial defect should be employed. This may require protective drops, ointment, therapeutic soft contact lenses, or closure of the eye by patching or other means. Because of its anticholinergic effects, incobotulinumtoxinA should be used with caution in patients at risk of developing narrow angle glaucoma. To prevent ectropion, botulinum toxin products should not be injected into the medial lower eyelid area.
- Ecchymosis easily occurs in the soft tissues of the eyelid. Immediate gentle pressure at the injection site can limit that risk.
Risk of Ptosis in Patients Treated with incobotulinumtoxinA for Glabellar Lines
- Do not exceed the recommended dosage and frequency of administration of incobotulinumtoxinA .
- In order to reduce the complication of ptosis the following steps should be taken:
- Avoid injection near the levator palpebrae superioris, particularly in patients with larger brow depressor complexes.
- Corrugator injections should be placed at least 1 cm above the bony supraorbital ridge.
Human Albumin and Transmission of Viral Diseases
- This product contains albumin, a derivative of human blood. Based on effective donor screening and product manufacturing processes, it carries an extremely remote risk for transmission of viral diseases. A theoretical risk for transmission of Creutzfeldt-Jakob disease (CJD) is also considered extremely remote. No cases of transmission of viral diseases or CJD have ever been reported for albumin.
Adverse Reactions
Clinical Trials Experience
- The following adverse reactions to incobotulinumtoxinA are discussed in greater detail in other sections of the labeling:
- Hypersensitivity
- Dysphagia and Breathing Difficulties in Treatment of cervical dystonia
- Spread of Effects from Toxin
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug, and may not reflect the rates observed in practice.
Cervical Dystonia
- The data described below reflect exposure to a single intramuscular dose of incobotulinumtoxinA in a placebo-controlled, Phase 3 trial in patients with cervical dystonia . In this study, 159 patients received incobotulinumtoxinA (78 were randomized to receive a total dose of 120 Units, and 81 were randomized to receive a total dose of 240 Units). incobotulinumtoxinA -treated patients were 18 to 79 years old (mean 53 years), and were predominantly female (66%) and Caucasian (91%). At study baseline, approximately 25% had mild, 50% had moderate, and 25% had severe cervical dystonia. Approximately 61% of incobotulinumtoxinA -treated patients had previously received another botulinum toxin type A product. Common adverse events (≥5% in any incobotulinumtoxinA treatment group) observed in patients who received incobotulinumtoxinA (120 Units or 240 Units) included dysphagia, neck pain, muscle weakness, injection site pain, and musculoskeletal pain.
Blepharospasm
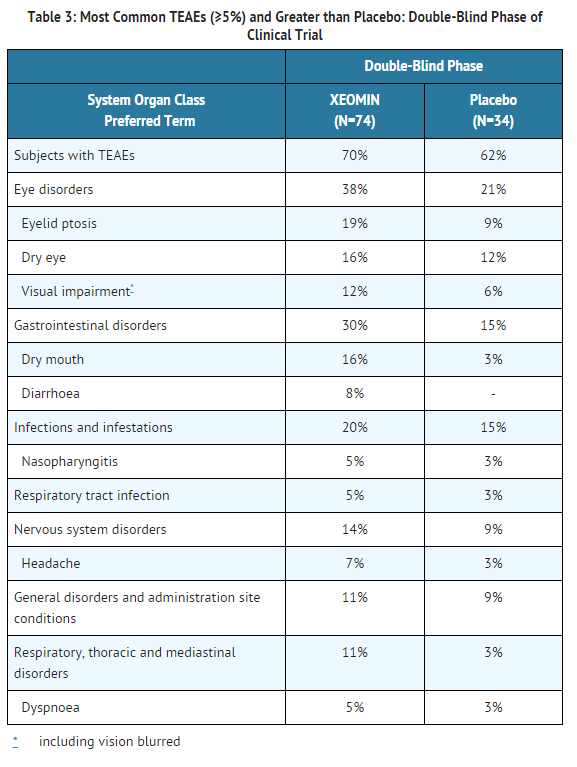
- In the placebo-controlled Phase 3 trial in patients with blepharospasm previously treated with onabotulinumtoxinA (Botox), 74 patients received incobotulinumtoxinA at a mean dose of approximately 33 Units per eye (minimum 10 Units, maximum 50 Units). incobotulinumtoxinA -treated patients were 22 to 79 years of age (mean 62 years), predominantly female (65%), Caucasian (79%), and had a mean time since diagnosis of approximately 5 years.
- The adverse events occurring in ≥5% of incobotulinumtoxinA -treated patients and greater than placebo in the Phase 3 study were eyelid ptosis, dry eye, dry mouth, diarrhea, headache, visual impairment, dyspnea, nasopharyngitis, and respiratory tract infection. No serious adverse events occurred in patients who received incobotulinumtoxinA ; one placebo-treated patient experienced a serious adverse event (dyspnea).
Glabellar Lines
- In three placebo-controlled trials in 803 subjects with glabellar lines, 535 subjects received a single dose of 20 Units incobotulinumtoxinA and 268 subjects received placebo. incobotulinumtoxinA treated subjects were 24 to 74 years old, and were predominantly female (88%). The most frequent adverse reactions in incobotulinumtoxinA treated subjects were: headache 29 (5.4%), facial paresis 4 (0.7%), injection site hematoma 3 (0.6%) and eyelid edema 2 (0.4%). Four serious adverse events occurred in two placebo-treated subjects. Six incobotulinumtoxinA treated subjects experienced six serious adverse events. All serious adverse events were assessed as unrelated to study drug.
- The adverse reactions below reflect exposure to incobotulinumtoxinA with glabellar lines in placebo-controlled studies. Adverse reactions are adverse events in which there is some basis to believe there is a causal relationship between the drug and the occurrence of the adverse event.
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- In open label, multiple dose trials, adverse reactions were reported for 105 of the 800 subjects (13.1%). Headache was the most common adverse reaction, reported for 57 subjects (7.1%), followed by injection site hematoma in 8 subjects (1.0%). Adverse reactions reported in less than 1% of subjects were: facial paresis (brow ptosis), muscle disorder (elevation of eyebrow), injection site pain, and eyelid edema.
Immunogenicity
- As with all therapeutic proteins, there is a potential for immunogenicity.
- The incidence of antibody formation is highly dependent on the sensitivity and specificity of the assay. In addition, the observed incidence of antibody positivity in an assay may be influenced by several factors including assay methodology, sample handling, timing of sample collection, concomitant medications, and underlying disease. For these reasons, comparison of the incidence of antibodies across products in this class may be misleading.
Postmarketing Experience
- The following adverse reactions have been reported during post-approval use with incobotulinumtoxinA . Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure: eye swelling, eyelid edema, dysphagia, nausea, flu-like symptoms, injection site pain, injection site reaction, allergic dermatitis, localized allergic reactions like swelling, edema, erythema, pruritus or rash, herpes zoster, muscular weakness, muscle spasm, dysarthria, myalgia and hypersensitivity.
Drug Interactions
- No formal drug interaction studies have been conducted with incobotulinumtoxinA .
- Coadministration of incobotulinumtoxinA and aminoglycoside antibiotics or other agents interfering with neuromuscular transmission, e.g., tubocurarine-type muscle relaxants, should only be performed with caution as these agents may potentiate the effect of the toxin.
- Use of anticholinergic drugs after administration of incobotulinumtoxinA may potentiate systemic anticholinergic effects.
- The effect of administering different botulinum toxin products at the same time or within several months of each other is unknown. Excessive neuromuscular weakness may be exacerbated by administration of another botulinum toxin prior to the resolution of the effects of a previously administered botulinum toxin. Excessive weakness may also be exaggerated by administration of a muscle relaxant before or after administration of incobotulinumtoxinA .
Use in Specific Populations
Pregnancy
- There are no adequate and well-controlled studies in pregnant women. incobotulinumtoxinA should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. incobotulinumtoxinA was embryotoxic in rats and increased abortions in rabbits when given at doses higher than the maximum recommended human dose (MRHD) for cervical dystonia (120 Units) on a body weight basis.
- When incobotulinumtoxinA was administered intramuscularly to pregnant rats during organogenesis (3, 10, or 30 Units/kg on gestational days [GDs] 6, 12, and 19; or 7 Units/kg on GDs 6 to 19; or 2, 6, or 18 Units/kg on GDs 6, 9, 12, 16, and 19), decreases in fetal body weight and skeletal ossification were observed at doses that were also maternally toxic. The no-effect level for embryotoxicity in rats was 6 Units/kg (3 times the MRHD for cervical dystonia on a body weight basis). Intramuscular administration to pregnant rabbits during organogenesis (1.25, 2.5, or 5.0 Units/kg on GDs 6, 18, and 28) resulted in an increased rate of abortion at the highest dose, which was also maternally toxic. In rabbits, the no-effect level for increased abortion was 2.5 Units/kg (similar to the MRHD for cervical dystonia on a body weight basis).
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of IncobotulinumtoxinA in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of IncobotulinumtoxinA during labor and delivery.
Nursing Mothers
- It is not known whether botulinum toxin type A is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when incobotulinumtoxinA is administered to a nursing woman.
Pediatric Use
- Safety and effectiveness of incobotulinumtoxinA in patients less than 18 years of age have not been established
Geriatic Use
- Cervical Dystonia
- In the Phase 3 study in cervical dystonia , 29 patients were older than 65 years of age, including 19 patients who received incobotulinumtoxinA and 10 patients who received placebo. Of these, ten (53%) incobotulinumtoxinA -treated patients and four (40%) placebo-treated patients experienced an adverse event. For patients over 65 years of age treated with incobotulinumtoxinA , the most common adverse events were dysphagia (4 patients, 21%) and asthenia (2 patients, 11%). One incobotulinumtoxinA -treated patient (5%) experienced severe dizziness.
- Blepharospasm
- In the Phase 3 study in blepharospasm , 41 patients were older than 65 years of age, including 29 of 75 patients (39%) who received incobotulinumtoxinA and 12 of 34 patients (35%) who received placebo. Of these patients, 22 of 29 (76%) incobotulinumtoxinA -treated patients, compared with 7 of 12 (58%) placebo-treated patients, experienced an adverse event. One incobotulinumtoxinA -treated patient experienced severe dysphagia.
- Glabellar Lines
- There are limited clinical data with incobotulinumtoxinA in subjects over 65 years of age and over in clinical studies with glabellar lines. Of the total number of subjects in the placebo-controlled clinical studies GL1 and GL2, 21 (4%) subjects were 65 and over. Efficacy was observed in 20% (3/15) of incobotulinumtoxinA subjects 65 years and over. For the entire safety database of geriatric subjects, there was no increase in the incidence of adverse events related to treatment with incobotulinumtoxinA .
Gender
There is no FDA guidance on the use of IncobotulinumtoxinA with respect to specific gender populations.
Race
There is no FDA guidance on the use of IncobotulinumtoxinA with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of IncobotulinumtoxinA in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of IncobotulinumtoxinA in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of IncobotulinumtoxinA in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of IncobotulinumtoxinA in patients who are immunocompromised.
Administration and Monitoring
Administration
- Intramuscular
Monitoring
There is limited information regarding Monitoring of IncobotulinumtoxinA in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of IncobotulinumtoxinA in the drug label.
Overdosage
- Excessive doses of incobotulinumtoxinA may be expected to produce neuromuscular weakness with a variety of symptoms. Respiratory support may be required where excessive doses cause paralysis of the respiratory muscles. In the event of overdose, the patient should be medically monitored for symptoms of excessive muscle weakness or muscle paralysis .Symptomatic treatment may be necessary.
- Symptoms of overdose are not likely to be present immediately following injection. Should accidental injection or oral ingestion occur, the person should be medically supervised for several weeks for signs and symptoms of excessive muscle weakness or paralysis.
- There is no significant information regarding overdose from clinical studies in cervical dystonia and blepharospasm.
- In the event of overdose, antitoxin raised against botulinum toxin is available from the Centers for Disease Control and Prevention (CDC) in Atlanta, GA. However, the antitoxin will not reverse any botulinum toxin-induced effects already apparent by the time of antitoxin administration. In the event of suspected or actual cases of botulinum toxin poisoning, please contact your local or state Health Department to process a request for antitoxin through the CDC.
Pharmacology
| |
Botulinum toxin A
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | |
| ATC code | M03 |
| PubChem | ? |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 149 kDa |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status |
[[Prescription drug|Template:Unicode-only]](US) |
| Routes | IM (approved), SC, intradermal, into glands |
| Bontoxilysin | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC number | 3.4.24.69 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
Mechanism of Action
- IncobotulinumtoxinA blocks cholinergic transmission at the neuromuscular junction by inhibiting the release of acetylcholine from peripheral cholinergic nerve endings. This inhibition occurs according to the following sequence: neurotoxin binding to cholinergic nerve terminals, internalization of the neurotoxin into the nerve terminal, translocation of the light-chain part of the molecule into the cytosol of the nerve terminal, and enzymatic cleavage of SNAP25, a presynaptic target protein essential for the release of acetylcholine. Impulse transmission is re-established by the formation of new nerve endings.
Structure
- The active ingredient of incobotulinumtoxinA is botulinum toxin type A produced from fermentation of Hall strainClostridium botulinum serotype A. The botulinum toxin complex is purified from the culture supernatant and then the active ingredient is separated from the proteins (hemagglutinins and non-hemagglutinins) through a series of steps yielding the active neurotoxin with molecular weight of 150 kDa, without accessory proteins. incobotulinumtoxinA is a sterile white to off-white lyophilized powder intended for intramuscular injection after reconstitution with preservative-free 0.9% Saline for Injection. One vial of incobotulinumtoxinA contains 50 or 100 Units of incobotulinumtoxinA, 1 mg of human albumin, and 4.7 mg sucrose.
- The primary release procedure for incobotulinumtoxinA uses a cell-based potency assay to determine the potency relative to a reference standard. One Unit corresponds to the median intraperitoneal lethal dose (LD50) in mice. As the method for conducting the assay is specific to incobotulinumtoxinA , Units of biological activity of incobotulinumtoxinA cannot be converted into Units of any other botulinum toxin assessed with other specific assays.
Pharmacodynamics
- In patients, recovery from paralysis after intramuscular injection normally occurs within 3-4 months as nerve terminals sprout and reconnect with the muscle endplate.
Pharmacokinetics
- General characteristics of the active substance:
- Using currently available analytical technology, it is not possible to detect incobotulinumtoxinA in the peripheral blood following intramuscular injection at the recommended doses.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Carcinogenesis
- Studies to evaluate the carcinogenic potential of incobotulinumtoxinA have not been conducted.
- Mutagenesis
- Genotoxicity studies have not been conducted for incobotulinumtoxinA .
- Impairment of Fertility
- In a fertility and early embryonic development study in rabbits, males and females were dosed with incobotulinumtoxinA (1.25, 2.5, or 3.5 Units/kg) intramuscularly every two weeks for 5 and 3 doses, respectively, beginning 2 weeks prior to mating. No effects on mating or fertility were observed . The highest dose tested is approximately twice the maximum recommended human dose for cervical dystonia (120 Units) on a body weight basis.
Clinical Studies
Cervical Dystonia
- incobotulinumtoxinA has been investigated in a Phase 3, randomized, double-blind, placebo-controlled, multi-center trial in a total of 233 patients with cervical dystonia. Patients had a clinical diagnosis of predominantly rotational cervical dystonia, with baseline Toronto Western Spasmodic Torticollis Rating Scale (TWSTRS) total score ≥20, TWSTRS severity score ≥10, TWSTRS disability score ≥3, and TWSTRS pain score ≥1. For patients who had previously received a botulinum toxin treatment for cervical dystonia, the trial required that ≥10 weeks had passed since the most recent botulinum toxin administration. Patients with swallowing disorders or any significant neuromuscular disease that might interfere with the study were excluded from enrollment. Patients were randomized (1:1:1) to receive a single administration of incobotulinumtoxinA 240 Units (n=81), incobotulinumtoxinA 120 Units (n=78), or placebo (n=74). Each patient received a single administration of 4.8 mL of reconstituted study agent (incobotulinumtoxinA 240 Units, incobotulinumtoxinA 120 Units, or placebo). The investigator at each site decided which muscles would receive injections of the study agent, the number of injection sites, and the volume at each site. The muscles most frequently injected were the splenius capitis/semispinalis, trapezius, sternocleidomastoid, scalene, and levator scapulae muscles. Table 5 indicates the average incobotulinumtoxinA dose, and percentage of total dose, injected into specific muscles in the pivotal clinical trial.
- Most patients received a total of 2-10 injections into the selected muscles. Patients were assessed by telephone at one week post-injection, during clinic visits at Weeks 4 and 8, and then by telephone assessments or clinic visits every two weeks up to Week 20.
- The mean age of the study patients was 53 years, and 66% of the patients were women. At study baseline, 61% of patients had previously received a botulinum toxin as treatment for cervical dystonia. The study was completed by 94% of study patients. Three patients discontinued the study prematurely due to adverse events: two patients in the 240 Unit group experienced musculoskeletal pain and muscle weakness, and one patient in the 120 Unit group experienced nausea and dizziness.
- The primary efficacy endpoint was the change in the TWSTRS total score from baseline to Week 4 post-injection, in the intent-to-treat (ITT) population, with missing values replaced by the patient's baseline value. In the ITT population, the difference between the incobotulinumtoxinA 240 Unit group and the placebo group in the change of the TWSTRS total score from baseline to Week 4 was -9.0 points, 95% confidence interval (CI) -12.0; -5.9 points; the difference between the incobotulinumtoxinA 120 Unit group and the placebo group in the change of the TWSTRS total score from baseline to Week 4 was -7.5 points, 95% CI -10.4; -4.6 points.
- Figure 2 illustrates the cumulative percentage of patients from each of the three treatment groups who had attained the specified change in TWSTRS Score from baseline versus 4 weeks post-injection. Three change scores have been identified for illustrative purposes, and the percent of patients in each group achieving that result is shown.
- The curves demonstrate that both patients assigned to placebo and incobotulinumtoxinA have a wide range of responses, but that the active treatment groups are more likely to show greater improvements. A curve for an effective treatment would be shifted to the left of the curve for placebo, while an ineffective or deleterious treatment would be superimposed upon or shifted to the right of the curve for placebo.
- Comparison of each incobotulinumtoxinA group to the placebo group was statistically significant at p<0.001. Initial incobotulinumtoxinA doses of 120 Units and 240 Units demonstrated no significant difference in effectiveness between the doses. The efficacy of incobotulinumtoxinA was similar in patients who were botulinum toxin naïve and those who had received botulinum toxin prior to this study.
- Examination of age and gender subgroups did not identify differences in response to incobotulinumtoxinA among these subgroups. There were too few African-American patients to adequately assess efficacy in that population.
Blepharospasm
- incobotulinumtoxinA has been investigated in a Phase 3, randomized, double-blind, placebo-controlled, multi-center trial in a total of 109 patients with blepharospasm. Patients had a clinical diagnosis of benign essential blepharospasm, with baseline Jankovic Rating Scale (JRS) Severity subscore ≥2, and a stable satisfactory therapeutic response to previous administrations of onabotulinumtoxinA (Botox). At least 10 weeks had to have elapsed since the most recent onabotulinumtoxinA administration. Patients with any significant neuromuscular disease that might interfere with the study were excluded from enrollment. Patients were randomized (2:1) to receive a single administration of incobotulinumtoxinA (n=75) or placebo (n=34). Each patient in the incobotulinumtoxinA group received a incobotulinumtoxinA treatment (dose, volume, dilution, and injection sites per muscle) that was similar to the most recent onabotulinumtoxinA injection sessions prior to study entry. The highest dose permitted in this study was 50 Units per eye; the mean incobotulinumtoxinA dose was 33 Units per eye.
- In Table 6 the most frequently injected sites, the median dose per injection site, and the median number (and range) of injection sites per eye are presented.
- Patients were assessed during clinic visits at Weeks 3 and 6, and then by telephone or at clinic visits every two weeks up to Week 20.
- The mean age of the study patients was 62 years, and 65% of the patients were women. The study was completed by 94% of study patients. Approximately one third of patients had other dystonic phenomena; in all but 1% this was limited to facial, cervical, perioral and mandibular muscles. No patients discontinued the study prematurely due to adverse events.
- The primary efficacy endpoint was the change in the JRS Severity subscore from baseline to Week 6 post-injection, in the intent-to-treat (ITT) population, with missing values replaced by the patient's most recent value (i.e., last observation carried forward). In the ITT population, the difference between the incobotulinumtoxinA group and the placebo group in the change of the JRS Severity subscore from baseline to Week 6 was -1.0 (95% CI -1.4; -0.5) points. Comparison of the incobotulinumtoxinA group to the placebo group was statistically significant at p<0.001.
- Examination of age and gender subgroups did not identify substantial differences in response to incobotulinumtoxinA among these subgroups. There were too few African-American patients to assess efficacy in that population.
Glabellar Lines
- Two identically designed randomized, double-blind, multi-center, placebo controlled clinical trials (Studies GL-1 and GL-2) were conducted to evaluate incobotulinumtoxinA for use in the temporary improvement of moderate to severe glabellar lines. The studies enrolled 547 healthy patients (≥18 years old) with glabellar lines of at least moderate severity at maximum frown. Three hundred sixty six subjects were treated with 20 U of incobotulinumtoxinA and 181 subjects were treated with placebo. Subjects were excluded if they had marked ptosis, deep dermal scarring, or an inability to lessen glabellar lines, even by physically spreading them apart. The mean age of study subjects was 46 years. The majority of patients were female (86% and 93% in Studies GL-1 and GL-2, respectively), and predominantly Caucasian (89% and 65% respectively). The study subjects received either 20 U of incobotulinumtoxinA or an equal amount of placebo. The total dose was delivered in 5 equally divided intramuscular injections of 4 Units each to specific sites (see FIGURE 1). Subjects were followed up for 120 days.
- Investigators and subjects assessed efficacy at maximum frown on Day 30 of treatment using a 4-point scale (0=none, 1=mild, 2=moderate, 3=severe). Composite treatment success was defined as a 2-grade improvement on this scale compared to baseline for both the investigator's and subject's assessments on Day 30. The percentage of subjects with treatment success was greater on the incobotulinumtoxinA arm than the placebo arm at Day 30 in both studies (see TABLE 7). The percentage of subjects with composite treatment success at each visit are presented in Figure 4.
How Supplied
- Type 1 borosilicate glass single-use vials with latex-free bromobutyl rubber closures and tamper-proof aluminum seals in the following pack sizes:
- Unopened vials of incobotulinumtoxinA can be stored at room temperature 20 to 25°C (68 to 77° F), in a refrigerator at 2 to 8°C (36 to 46°F), or a freezer at -20 to -10°C (-4 to 14°F) for up to 36 months. Do not use after the expiration date on the vial. Reconstituted incobotulinumtoxinA should be stored in a refrigerator at 2 to 8°C (36 to 46°F) and administered within 24 hours.
Handling
- incobotulinumtoxinA is reconstituted prior to use with sterile preservative-free 0.9% Sodium Chloride Injection, USP .
- incobotulinumtoxinA should not be used if the reconstituted solution has a cloudy appearance or contains floccular or particulate matter.
- Any reconstituted toxin solution for injection that has been stored for more than 24 hours, as well as any unused solution for injection, should be discarded.
Storage
There is limited information regarding IncobotulinumtoxinA Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::IncobotulinumtoxinA |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::IncobotulinumtoxinA |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patients or caregivers should be advised to seek immediate medical care if swallowing, speech or respiratory disorders arise.
- Previously immobile or sedentary patients should be reminded to gradually resume activities following the injection of incobotulinumtoxinA for the treatment of cervical dystonia and blepharospasm.
- Patients should be informed that injections of incobotulinumtoxinA may cause dyspnea, or mild to severe dysphagia, with the risk of aspiration
- Patients should be counseled that if loss of strength, muscle weakness, blurred vision, or drooping eyelids occur, they should avoid driving a car or engaging in other potentially hazardous activities.
- Patients should be informed that injections of incobotulinumtoxinA may cause reduced blinking or effectiveness of blinking, and that they should seek immediate medical attention if eye pain or irritation occur following treatment.
Precautions with Alcohol
- Alcohol-IncobotulinumtoxinA interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Xeomin
Look-Alike Drug Names
There is limited information regarding IncobotulinumtoxinA Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=IncobotulinumtoxinA |Label Name=IncobotulinumtoxinA12.png
}}
{{#subobject:
|Label Page=IncobotulinumtoxinA |Label Name=IncobotulinumtoxinA13.png
}}
{{#subobject:
|Label Page=IncobotulinumtoxinA |Label Name=IncobotulinumtoxinA14.png
}}
{{#subobject:
|Label Page=IncobotulinumtoxinA |Label Name=IncobotulinumtoxinA15.png
}}