Albumin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Albumin is a volume expander that is FDA approved for the treatment of hypovolemic shock, burn therapy, hypoproteinemia with or without edema, adult respiratory distress syndrome, acute liver failure, neonatal hemolytic disease, acute nephrosis. Common adverse reactions include hypersensitivity reaction.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
Emergency Treatment of Hypovolemic Shock
- Albumin 20 is hyperoncotic and on intravenous infusion will expand the plasma volume by an additional amount, three to four times the volume actually administered, by withdrawing fluid from the interstitial spaces, provided the patient is normally hydrated interstitially or there is interstitial edema. If the patient is dehydrated, additional crystalloids must be given, or alternatively, Albumin (Human) 5%, USP (albumin™ 5) should be used. The patient’s hemodynamic response should be monitored and the usual precautions against circulatory overload observed. The total dose should not exceed the level of albumin found in the normal individual; i.e., about 2 g per kg body weight in the absence of active bleeding. Although albumin is to be preferred for the usual volume deficits, albumin 20 with appropriate crystalloids may offer therapeutic advantages in oncotic deficits or in long-standing shock where treatment has been delayed.
- Removal of ascitic fluid from a patient with cirrhosis may cause changes in cardiovascular function and even result in hypovolemic shock. In such circumstances, the use of an albumin infusion may be required to support the blood volume.
Burn Therapy
- An optimal therapeutic regimen with respect to the administration of colloids, crystalloids, and water following extensive burns has not been established. During the first 24 hours after sustaining thermal injury, large volumes of crystalloids are infused to restore the depleted extracellular fluid volume. Beyond 24 hours albumin 20 can be used to maintain plasma colloid osmotic pressure.
Hypoproteinemia With or Without Edema
- During major surgery, patients can lose over half of their circulating albumin with the attendant complications of oncotic deficit.A similar situation can occur in sepsis or intensive care patients. Treatment with albumin 20 may be of value in such cases.
Adult Respiratory Distress Syndrome (ARDS)
- This is characterized by deficient oxygenation caused by pulmonary interstitial edema complicating shock and postsurgical conditions. When clinical signs are those of hypoproteinemia with a fluid volume overload, albumin 20 together with a diuretic may play a role in therapy.
Cardiopulmonary Bypass
- With the relatively small priming volume required with modern pumps, preoperative dilution of the blood using albumin and crystalloid has been shown to be safe and well-tolerated. Although the limit to which the hematocrit and plasma protein concentration can be safely lowered has not been defined, it is common practice to adjust the albumin and crystalloid pump prime to achieve a hematocrit of 20% and a plasma albumin concentration of 2.5 g per 100 mL in the patient.
Acute Liver Failure
- In the uncommon situation of rapid loss of liver function with or without coma, administration of albumin may serve the double purpose of supporting the colloid osmotic pressure of the plasma as well as binding excess plasma bilirubin.
Neonatal Hemolytic Disease
- The administration of albumin 20 may be indicated prior to exchange transfusion, in order to bind free bilirubin, thus lessening the risk of kernicterus. A dosage of 1 g/kg body weight is given about 1 hour prior to exchange transfusion. Caution must be observed in hypervolemic infants.
Sequestration of Protein Rich Fluids
- This occurs in such conditions as acute peritonitis, pancreatitis, mediastinitis, and extensive cellulitis. The magnitude of loss into the third space may require treatment of reduced volume or oncotic activity with an infusion of albumin.
Erythrocyte Resuspension
- Albumin may be required to avoid excessive hypoproteinemia during certain types of exchange transfusion, or with the use of very large volumes of previously frozen or washed red cells. About 25 g of albumin per liter of erythrocytes is commonly used, although the requirements in preexistent hypoproteinemia or hepatic impairment can be greater. Albumin 20 is added to the isotonic suspension of washed red cells immediately prior to transfusion.
Acute Nephrosis
- Certain patients may not respond to cyclophosphamide or steroid therapy. The steroids may even aggravate the underlying edema. In this situation a loop diuretic and 100 mL albumin 20 repeated daily for 7 to 10 days may be helpful in controlling the edema and the patient may then respond to steroid treatment.
Renal Dialysis
- Although not part of the regular regimen of renal dialysis, albumin 20 may be of value in the treatment of shock or hypotension in these patients. The usual volume administered is about 100 mL, taking particular care to avoid fluid overload as these patients are often fluid overloaded and cannot tolerate substantial volumes of salt solution.
Situations in Which Albumin Administration is Not Warranted
- In chronic nephrosis, infused albumin is promptly excreted by the kidneys with no relief of the chronic edema or effect on the underlying renal lesion. It is of occasional use in the rapid “priming” diuresis of nephrosis. Similarly, in hypoproteinemic states associated with chronic cirrhosis, malabsorption, protein-losing enteropathies, pancreatic insufficiency, and undernutrition, the infusion of albumin as a source of protein nutrition is not justified.
Dosage
Hypovolemic Shock — For treatment of hypovolemic shock, the volume administered and the speed of infusion should be adapted to the response of the individual patient.
Burns — After a burn injury (usually beyond 24 hours) there is a close correlation between the amount of albumin infused and the resultant increase in plasma colloid osmotic pressure. The aim should be to maintain the plasma albumin concentration in the region of 2.5 ± 0.5 g per 100 mL with a plasma oncotic pressure of 20 mm Hg (equivalent to a total plasma protein concentration of 5.2 g per 100 mL).(2) This is best achieved by the intravenous administration of albumin 20. The duration of therapy is decided by the loss of protein from the burned areas and in the urine. In addition, oral or parenteral feeding with amino acids should be initiated, as the long-term administration of albumin should not be considered as a source of nutrition.
Hypoproteinemia With or Without Edema — Unless the underlying pathology responsible for the hypoproteinemia can be corrected, the intravenous administration of albumin 20 must be considered purely symptomatic or supportive.The usual daily dose of albumin for adults is 50 to 75 g and for children 25 g. Patients with severe hypoproteinemia who continue to lose albumin may require larger quantities. Since hypoproteinemic patients usually have approximately normal blood volumes, the rate of administration of albumin 20 should not exceed 2 mL per minute, as more rapid injection may precipitate circulatory embarrassment and pulmonary edema.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Albumin in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Albumin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Albumin in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Albumin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Albumin in pediatric patients.
Contraindications
- Certain patients, e.g., those with a history of congestive cardiac failure, renal insufficiency or stabilized chronic anemia, are at special risk of developing circulatory overload. A history of an allergic reaction to albumin is a specific contraindication to usage.
Warnings
- Albumin (Human) 20%, USP (albumin™ 20) is made from human plasma. Products made from human plasma may contain infectious agents, such as viruses, and, theoretically, the Creutzfeldt-Jakob Disease (CJD) agent that can cause disease. The theoretical risk for transmission of CJD is considered extremely remote. No cases of transmission of viral diseases or CJD have ever been identified for albumin. The risk that such products will transmit an infectious agent has been reduced by screening plasma donors for prior exposure to certain viruses, by testing for the presence of certain current virus infections, and by inactivating and/or removing certain viruses. Despite these measures, such products can still potentially transmit disease. There is also the possibility that unknown infectious agents may be present in such products. Individuals who receive infusions of blood or plasma products may develop signs and/or symptoms of some viral infections, particularly hepatitis C. ALL infections thought by a physician possibly to have been transmitted by this product should be reported by the physician or other healthcare provider to Grifols Therapeutics Inc. [1-800-520-2807].
- The physician should discuss the risks and benefits of this product with the patient, before prescribing or administering it to the patient.
- As with any hyperoncotic protein solution likely to be administered in large volumes, severe hemolysis and acute renal failure may result from the inappropriate use of Sterile Water for Injection as a diluent for Albumin (Human), 20%. Acceptable diluents include 0.9% Sodium Chloride or 5% Dextrose in Water.
- Solutions which have been frozen should not be used. Do not use if turbid. Do not begin administration more than 4 hours after the container has been entered. Partially used vials must be discarded. Vials which are cracked or which have been previously entered or damaged should not be used, as this may have allowed the entry of microorganisms. Glbumin 20 contains no preservative.
Adverse Reactions
Clinical Trials Experience
- Adverse reactions to albumin are rare. Such reactions may be allergic in nature or due to high plasma protein levels from excessive albumin administration. Allergic manifestations include urticaria, chills, fever, and changes in respiration, pulse and blood pressure.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Albumin in the drug label.
Drug Interactions
- Albumin 20 is compatible with whole blood, packed red cells, as well as the standard carbohydrate and electrolyte solutions intended for intravenous use. It should, however, not be mixed with protein hydrolysates, amino acid solutions nor those containing alcohol.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Pregnancy Category C
- Animal reproduction studies have not been conducted with albumin 20. It is also not known whether albumin 20 can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Albumin 20 should be given to a pregnant woman only if clearly needed.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Albumin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Albumin during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Albumin with respect to nursing mothers.
Pediatric Use
- Safety and effectiveness in the pediatric population have not been established.
Geriatic Use
There is no FDA guidance on the use of Albumin with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Albumin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Albumin with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Albumin in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Albumin in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Albumin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Albumin in patients who are immunocompromised.
Administration and Monitoring
Administration
- Intravenous
- Albumin 20 should always be administered by intravenous infusion. Albuked 20 may be administered either undiluted or diluted in 0.9% Sodium Chloride or 5% Dextrose in Water. If sodium restriction is required, Albuked 20 should only be administered either undiluted or diluted in a sodium-free carbohydrate solution such as 5% Dextrose in Water.
- A number of factors beyond our control could reduce the efficacy of this product or even result in an ill effect following its use. These include improper storage and handling of the product after it leaves our hands, diagnosis, dosage, method of administration, and biological differences in individual patients. Because of these factors, it is important that this product be stored properly and that the directions be followed carefully during use.
Preparation for Administration
- Remove seal to expose stopper. Always swab stopper top immediately with a suitable antiseptic prior to entering vial.
- Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever solution and container permit.
- Only 16 gauge needles or dispensing pins should be used with 20 mL vial sizes and larger. Needles or dispensing pins should only be inserted within the stopper area delineated by the raised ring. The stopper should be penetrated perpendicular to the plane of the stopper within the ring.
Monitoring
There is limited information regarding Monitoring of Albumin in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Albumin in the drug label.
Overdosage
There is limited information regarding Chronic Overdose of Albumin in the drug label.
Pharmacology

Mechanism of Action
There is limited information regarding Albumin Mechanism of Action in the drug label.
Structure
There is limited information regarding Albumin Structure in the drug label.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Albumin in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Albumin in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Albumin in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Albumin in the drug label.
How Supplied
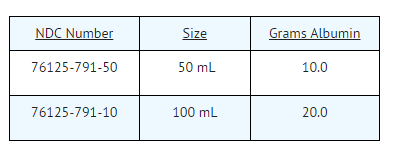
- Albuked 20 is available in 50 mL and 100 mL rubber-stoppered vials. Each single dose vial contains albumin in the following approximate amounts:
Storage
- Store at room temperature not exceeding 30°C (86°F). Do not freeze. Do not use after expiration date.
Images
Drug Images
{{#ask: Page Name::Albumin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Albumin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Albumin in the drug label.
Precautions with Alcohol
- Alcohol-Albumin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- ALBUKED®[1]
Look-Alike Drug Names
- A® — B®[2]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "ALBUKED - albumin (human) solution".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Label Page=Albumin |Label Name=Albumin image.jpg
}}
{{#subobject:
|Label Page=Albumin |Label Name=Albumin image 1.jpg
}}
{{#subobject:
|Label Page=Albumin |Label Name=Albumin ingredients and appearance.png
}}