Cefazolin sodium
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Rabin Bista, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Cefazolin sodium is a semi-synthetic cephalosporin for parenteral administration that is FDA approved for the treatment of respiratory tract infections,urinary tract infections, skin and skin structure infections, biliary tract infections, bone and joint infections, genital infections, septicemia, endocarditis and perioperative Prophylaxis. Common adverse reactions include diarrhea, oral candidiasis, vomiting and nausea.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Respiratory Tract Infections due to S. pneumoniae, S. aureus (including beta-lactamase-producing strains) and S. pyogenes.
- Urinary Tract Infections due to E. coli, P. mirabilis.
- Skin and Skin Structure Infections due to S. aureus (including beta-lactamase-producing strains), S. pyogenes, and other strains of streptococci.
- Biliary Tract Infections due to E. coli, various strains of streptococci, P. mirabilis, and S. aureus.
- Bone and Joint Infections Due to S. aureus.
- Genital Infections (i.e., prostatitis, epididymitis) due to E. coli, P. mirabilis.
- Septicemia due to S. pneumoniae, S. aureus (including beta-lactamase-producing strains), P. mirabilis, E. coli.
- Endocarditis due to S. aureus (including beta-lactamase-producing strains) and S. pyogenes.
- Perioperative Prophylaxis
- The prophylactic administration of Cefazolin for injection preoperatively, intraoperatively, and postoperatively may reduce the incidence of certain postoperative infections in patients undergoing surgical procedures which are classified as contaminated or potentially contaminated (e.g., vaginal hysterectomy, and cholecystectomy in high-risk patients such as those older than 70 years, with acute cholecystitis, obstructive jaundice, or common duct bile stones).
- The perioperative use of Cefazolin for injection may also be effective in surgical patients in whom infection at the operative site would present a serious risk (e.g., during open-heart surgery and prosthetic arthroplasty).
- The prophylactic administration of Cefazolin for injection should usually be discontinued within a 24-hour period after the surgical procedure. In surgery where the occurrence of infection may be particularly devastating (e.g., open-heart surgery and prosthetic arthroplasty), the prophylactic administration of CEFAZOLIN FOR INJECTION may be continued for 3 to 5 days following the completion of surgery.
Dosage
Adult Dosage

Perioperative Prophylactic Use
- To prevent postoperative infection in contaminated or potentially contaminated surgery, recommended doses are:
- 1 gram IV administered ½ hour to 1 hour prior to the start of surgery.
- For lengthy operative procedures (e.g., 2 hours or more), 500 mg to 1 gram IV during surgery (administration modified depending on the duration of the operative procedure).
- 500 mg to 1 gram IV every 6 to 8 hours for 24 hours postoperatively.
- It is important that (1) the preoperative dose be given just (½ to 1 hour) prior to the start of surgery so that adequate antibiotic levels are present in the serum and tissues at the time of initial surgical incision; and (2) Cefazolin for injection be administered, if necessary, at appropriate intervals during surgery to provide sufficient levels of the antibiotic at the anticipated moments of greatest exposure to infective organisms.
Dosage Adjustment for Patients With Reduced Renal Function
- Cefazolin for injection may be used in patients with reduced renal function with the following dosage adjustments: Patients with a creatinine clearance of 55 mL/min. or greater or a serum creatinine of 1.5 mg % or less can be given full doses. Patients with creatinine clearance rates of 35 to 54 mL/min. or serum creatinine of 1.6 to 3 mg % can also be given full doses but dosage should be restricted to at least 8 hour intervals. Patients with creatinine clearance rates of 11 to 34 mL/min. or serum creatinine of 3.1 to 4.5 mg % should be given ½ the usual dose every 12 hours. Patients with creatinine clearance rates of 10 mL/min. or less or serum creatinine of 4.6 mg % or greater should be given ½ the usual dose every 18 to 24 hours. All reduced dosage recommendations apply after an initial loading dose appropriate to the severity of the infection.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- Bacterial endocarditis; Prophylaxis[1]
- Streptococcus group B infection of the infant, Intrapartum; Prophylaxis[2]
Non–Guideline-Supported Use
- Bacteremia associated with intravascular line[3]
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Pediatric Dosage
- In pediatric patients, a total daily dosage of 25 to 50 mg per kg (approximately 10 to 20 mg per pound) of body weight, divided into 3 or 4 equal doses, is effective for most mild to moderately severe infections. Total daily dosage may be increased to 100 mg per kg (45 mg per pound) of body weight for severe infections. Since safety for use in premature infants and in neonates has not been established, the use of Cefazolin for injection in these patients is not recommended.
- In pediatric patients with mild to moderate renal impairment (creatinine clearance of 70 to 40 mL/min.), 60 percent of the normal daily dose given in equally divided doses every 12 hours should be sufficient. In patients with moderate impairment (creatinine clearance of 40 to 20 mL/min.), 25 percent of the normal daily dose given in equally divided doses every 12 hours should be adequate. Pediatric patients with severe renal impairment (creatinine clearance of 20 to 5 mL/min.) may be given 10 percent of the normal daily dose every 24 hours. All dosage recommendations apply after an initial loading dose.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of cefazolin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of cefazolin in pediatric patients.
Contraindications
- Cefazolin for injection is contraindicated in patients with known allergy to the cephalosporin group of antibiotics.
Warnings
- BEFORE THERAPY WITH CEFAZOLIN FOR INJECTION IS INSTITUTED, CAREFUL INQUIRY SHOULD BE MADE TO DETERMINE WHETHER THE PATIENT HAS HAD PREVIOUS HYPERSENSITIVITY REACTIONS TO CEFAZOLIN, CEPHALOSPORINS, PENICILLINS, OR OTHER DRUGS. IF THIS PRODUCT IS GIVEN TO PENICILLIN-SENSITIVE PATIENTS, CAUTION SHOULD BE EXERCISED BECAUSE CROSS-HYPERSENSITIVITY AMONG BETA-LACTAM ANTIBIOTICS HAS BEEN CLEARLY DOCUMENTED AND MAY OCCUR IN UP TO 10% OF PATIENTS WITH A HISTORY OF PENICILLIN ALLERGY. IF AN ALLERGIC REACTION TO CEFAZOLIN FOR INJECTION OCCURS, DISCONTINUE TREATMENT WITH THE DRUG. SERIOUS ACUTE HYPERSENSITIVITY REACTIONS MAY REQUIRE TREATMENT WITH EPINEPHRINE AND OTHER EMERGENCY MEASURES, INCLUDING OXYGEN, IV FLUIDS, IV ANTIHISTAMINES, CORTICOSTEROIDS, PRESSOR AMINES, AND AIRWAY MANAGEMENT, AS CLINICALLY INDICATED.
- Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including CEFAZOLIN FOR INJECTION, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
- C. difficile produces toxins A and B which contribute to the development of CDAD.
- Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
- If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
Precautions
General
- Prolonged use of Cefazolin for injection may result in the overgrowth of nonsusceptible organisms. Careful clinical observation of the patient is essential.
- When Cefazolin for injection is administered to patients with low urinary output because of impaired renal function, lower daily dosage is required.
- As with other beta-lactam antibiotics, seizures may occur if inappropriately high doses are administered to patients with impaired renal function.
- Cefazolin for injection, as with all cephalosporins, should be prescribed with caution in individuals with a history of gastrointestinal disease, particularly colitis.
- Cephalosporins may be associated with a fall in prothrombin activity. Those at risk include patients with renal or hepatic impairment or poor nutritional state, as well as patients receiving a protracted course of antimicrobial therapy, and patients previously stabilized on anticoagulant therapy. Prothrombin time should be monitored in patients at risk and exogenous vitamin K administered as indicated.
- Prescribing Cefazolin for injection in the absence of a proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Adverse Reactions
Clinical Trials Experience
- The following adverse reactions were reported from clinical trials:
Digestive
- Diarrhea
- Oral candidiasis (oral thrush)
- Mouth ulcers
- Vomiting
- Nausea
- Stomach cramps
- Epigastric pain
- Heartburn
- Flatus
- Anorexia
- Pseudomembranous colitis
- Onset of pseudomembranous colitis symptoms may occur during or after antibacterial treatment
Hematologic and Lymphatic
Allergic
Renal
- Increased BUN and creatinine levels
- Renal failure
Local Reactions
Other reactions
- Pruritus (including genital, vulvar and anal pruritus, genital moniliasis, and vaginitis)
- Dizziness
- Fainting
- Lightheadedness
- Confusion
- Weakness
- Tiredness
- Hypotension
- Somnolence
- Headache
Cephalosporin-class Adverse Reactions
Adverse Reactions
- Allergic reactions
- Urticaria
- Serum sickness-like reaction
- Erythema multiforme
- Toxic epidermal necrolysis
- Colitis
- Renal dysfunction
- Toxic nephropathy
- Abdominal pain
- Reversible hyperactivity
- Hypertonia
- Hepatic dysfunction including cholestasis
- Aplastic anemia
- Hemolytic anemia
- Hemorrhage
- Superinfection
- Seizures
Altered Laboratory Tests
- Prolonged prothrombin time
- Positive direct Coombs' test
- False-positive test for urinary glucose
- Elevated bilirubin
- Elevated LDH
- Increased creatinine
- pancytopenia
- agranulocytosis
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Cefazolin in the drug label.
Drug Interactions
- Probenecid may decrease renal tubular secretion of cephalosporins when used concurrently, resulting in increased and more prolonged cephalosporin blood levels
Drug/Laboratory Test Interactions
- A false positive reaction for glucose in the urine may occur with Benedict's solution, Fehling's solution or with CLINITEST® tablets, but not with enzyme-based tests such as CLINISTIX®.
- Positive direct and indirect antiglobulin (Coombs) tests have occurred; these may also occur in neonates whose mothers received cephalosporins before delivery.
Use in Specific Populations
Pregnancy
Pregnancy
Teratogenic Effects
- Reproduction studies have been performed in rats, mice and rabbits at doses up to 25 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to Cefazolin for injection. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
- There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Cefazolin in women who are pregnant.
Labor and Delivery
- When cefazolin has been administered prior to caesarean section, drug levels in cord blood have been approximately one quarter to one third of maternal drug levels. The drug appears to have no adverse effect on the fetus.
Nursing Mothers
- Cefazolin is present in very low concentrations in the milk of nursing mothers. Caution should be exercised when CEFAZOLIN FOR INJECTION is administered to a nursing woman.
Pediatric Use
- Safety and effectiveness for use in premature infants and neonates have not been established.
Geriatic Use
- Of the 920 subjects who received Cefazolin for injection in clinical studies, 313 (34%) were 65 years and over, while 138 (15%) were 75 years and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
- This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function
Gender
There is no FDA guidance on the use of Cefazolin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Cefazolin with respect to specific racial populations.
Renal Impairment
- When CEFAZOLIN FOR INJECTION is administered to patients with low urinary output because of impaired renal function, lower daily dosage is required
Hepatic Impairment
There is no FDA guidance on the use of Cefazolin in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Cefazolin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Cefazolin in patients who are immunocompromised.
Administration and Monitoring
Administration
- intravenous
Reconstitution
Preparation of Parenteral Solution
- Parenteral drug products should be shaken well when reconstituted, and inspected visually for particulate matter prior to administration. If particulate matter is evident in reconstituted fluids, the drug solutions should be discarded.
- Reconstituted solutions may range in color from pale yellow to yellow without a change in potency.
Directions for Proper Use of a Pharmacy Bulk Package
- Not for direct infusion. The Pharmacy Bulk Package is for use in the hospital pharmacy admixture service only in a suitable work area, such as a laminar flow hood. Using aseptic technique, the closure may be penetrated only one time using a suitable sterile dispensing set that allows measured dispensing of the contents. Use of a syringe and needle is not recommended as it may cause leakage. After entry, use entire contents of Pharmacy Bulk Package promptly. The entire contents of the Pharmacy Bulk Package should be dispensed within 4 hours of initial entry. This time limit should begin with the introduction of the solvent or diluent into the Pharmacy Bulk Package. Discard Pharmacy Bulk Package within 4 hours after initial entry.
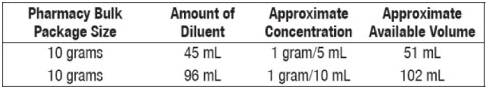
Pharmacy Bulk Packages
- Add Sterile Water for Injection, Bacteriostatic Water for Injection or Sodium Chloride Injection according to the table below. Shake well.
Intermittent or continuous infusion
- Dilute reconstituted CEFAZOLIN FOR INJECTION in 50 to 100 mL of 1 of the following solutions:
- Sodium Chloride Injection, USP
- 5% or 10% Dextrose Injection, USP
- 5% Dextrose in Lactated Ringer's Injection, USP
- 5% Dextrose and 0.9% Sodium Chloride Injection, USP
- 5% Dextrose and 0.45% Sodium Chloride Injection, USP
- 5% Dextrose and 0.2% Sodium Chloride Injection, USP
- Lactated Ringer's Injection, USP
- Invert Sugar 5% or 10% in Sterile Water for Injection
- Ringer's Injection, USP
- 5% Sodium Bicarbonate Injection, USP
- When diluted according to the instructions above, Cefazolin for injection is stable for 24 hours at room temperature or for 10 days if stored under refrigeration (5°C or 41°F).
- Prior to administration parenteral drug products should be inspected visually for particulate matter and discoloration whenever solution and container permit.
Monitoring
There is limited information regarding Monitoring of Cefazolin in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Cefazolin sodium in the drug label.
Overdosage
There is limited information regarding Overdose of Cefazolin in the drug label.
Pharmacology
Mechanism of Action
- Cefazolin is a bactericidal agent that acts by inhibition of bacterial cell wall synthesis.
Structure
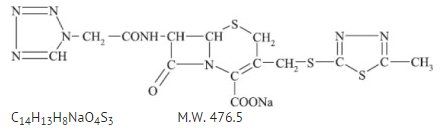
- Cefazolin is the sodium salt of 3-{[(5-methyl-1,3,4-thiadiazol-2-yl)thio]-methyl}-8-oxo-7-[2-(1H-tetrazol-1-yl)acetamido]-5-thia-1-azabicyclo [4.2.0]oct-2-ene-2-carboxylic acid.
- Cefazolin sodium USP has the following structural formula:
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Cefazolin in the drug label.
Pharmacokinetics
- Studies have shown that following intravenous administration of cefazolin to normal volunteers, mean serum concentrations peaked at approximately 185 mcg/mL and were approximately 4 mcg/mL at 8 hours for a 1 gram dose.
- The serum half-life for cefazolin is approximately 1.8 hours following intravenous administration.
- In a study, using normal volunteers, of constant intravenous infusion with dosages of 3.5 mg/kg for 1 hour (approximately 250 mg) and 1.5 mg/kg the next 2 hours (approximately 100 mg), cefazolin produced a steady serum concentration at the third hour of approximately 28 mcg/mL.
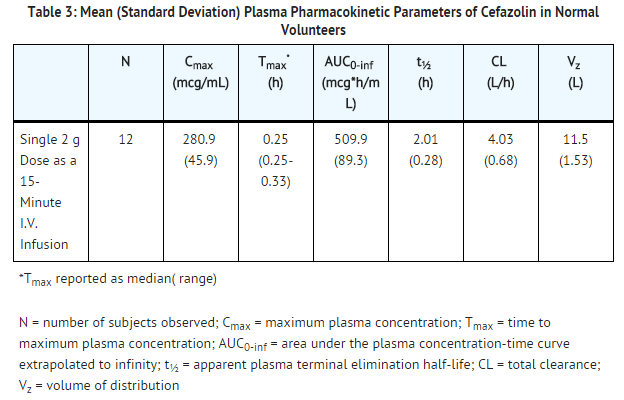
- Plasma pharmacokinetic parameters of cefazolin in normal volunteers (N=12) following a single 15-minute intravenous infusion of 2 grams of Cefazolin for Injection, USP are summarized in Table 3.
- Studies in patients hospitalized with infections indicate that cefazolin produces mean peak serum concentrations approximately equivalent to those seen in normal volunteers.
- Bile concentrations in patients without obstructive biliary disease can reach or exceed serum concentrations by up to five times; however, in patients with obstructive biliary disease, bile concentrations of cefazolin are considerably lower than serum concentrations (< 1 mcg/mL).
- In synovial fluid, the cefazolin concentration becomes comparable to that reached in serum at about 4 hours after drug administration.
- Studies of cord blood show prompt transfer of cefazolin across the placenta. Cefazolin is present in very low concentrations in the milk of nursing mothers.
- Cefazolin is excreted unchanged in the urine. In the first 6 hours approximately 60% of the drug is excreted in the urine and this increases to 70% to 80% within 24 hours.
Microbiology
Mechanism of Resistance
- Predominant mechanisms of bacterial resistance to cephalosporins include the presence of extended-spectrum beta-lactamases and enzymatic hydrolysis.
List of Microorganisms
- Cefazolin has been shown to be active against most isolates of the following microorganisms both in vitro and in clinical infections
Gram-Positive Bacteria
- Methicillin-resistant staphylococci are uniformly resistant to cefazolin
Gram-Negative Bacteria
- Most strains of indole positive Proteus (Proteus vulgaris), Enterobacter spp., Morganella morganii, Providencia rettgeri, Serratia spp., and Pseudomonas spp. are resistant to cefazolin.
Susceptibility Test Methods
- When available, the clinical microbiology laboratory should provide the results of in vitro susceptibility test results for antimicrobial drug products used in resident hospitals to the physician as periodic reports that describe the susceptibility profile of nosocomial and community-acquired pathogens. These reports should aid the physician in selecting an antibacterial drug product for treatment.
Dilution Techniques
- Quantitative methods are used to determine minimum inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standard test1,2 (broth and/or agar). The MIC values obtained should be interpreted according to criteria as provided in table 4.
Diffusion Techniques
- Quantitative methods that require measurement of zone diameters provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. The zone size provides an estimate of the susceptibility of bacteria to antimicrobial compounds. The zone size should be interpreted using a standard test method 2,3. This procedure uses paper disks impregnated with 30 mcg cefazolin to test the susceptibility of microorganisms to cefazolin. The disk diffusion interpretive criteria are provided in table 4.
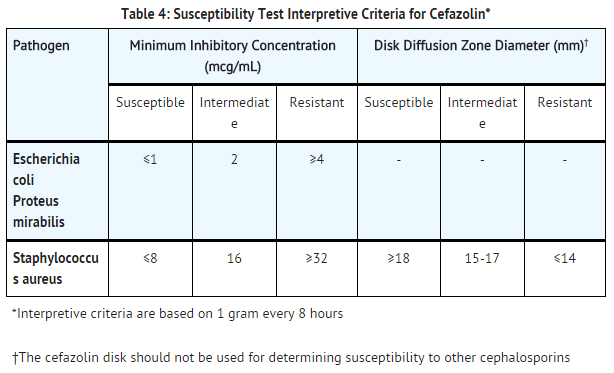
- S. pyogenes and S. agalactiae that have a penicillin MIC of ≤0.12 mcg/mL or disk diffusion zone diameters of ≥24 mm with a 10 mcg penicillin disk may be interpreted as susceptible to cefazolin. Non-meningitis isolates of S. pneumoniae that have a penicillin MIC of ≤0.06 mcg/mL may be interpreted as susceptible to cefazolin.
- A report of Susceptible indicates that the antimicrobial is likely to inhibit growth of the pathogen if the antimicrobial compound reaches the concentrations at the infection site necessary to inhibit growth of the pathogen. A report of Intermediate indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug product is physiologically concentrated or in situations where a high dosage of the drug product can be used. This category also provides a buffer zone that prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of Resistant indicates that the antimicrobial is not likely to inhibit growth of the pathogen if the antimicrobial compound reaches the concentrations usually achievable at the infection site; other therapy should be selected.
Quality Control
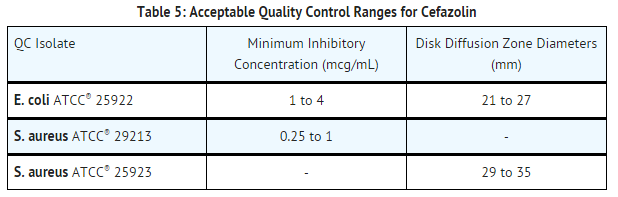
- Standardized susceptibility test procedures require the use of laboratory controls to monitor and ensure the accuracy and precision of supplies and reagents used in the assay and the techniques of the individual performing the test. Standard cefazolin powder should provide the following MIC values noted in TABLE 5. For the diffusion technique using the 30 mcg disk, the criteria in TABLE 5 should be achieved.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Cefazolin in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Cefazolin in the drug label.
How Supplied
- Each Pharmacy Bulk Package contains cefazolin sodium equivalent to 10 grams of cefazolin.
- As with other cephalosporins, reconstituted Cefazolin for injection tends to darken depending on storage conditions, within the stated recommendations. However, product potency is not adversely affected.
Storage
- Before reconstitution, store at 20° to 25°C (68° to 77°F). [See USP Controlled Room Temperature.] Protect from light.
- Sterile, Nonpyrogenic, Preservative-free.
- The container closure is not made with natural rubber latex.
Images
Drug Images
{{#ask: Page Name::Cefazolin sodium |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
Principal display Panel
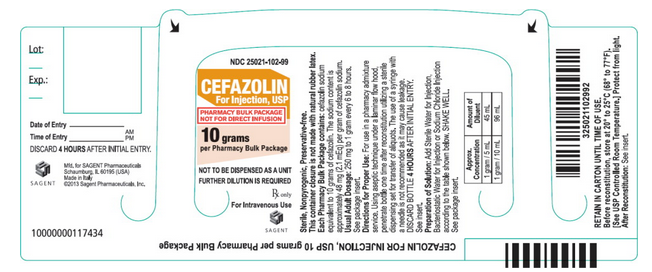
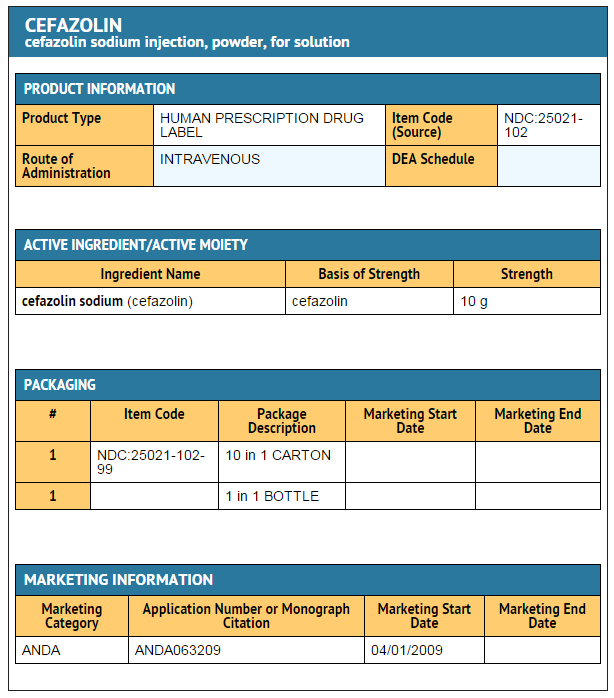
- PACKAGE LABEL – PRINCIPAL DISPLAY PANEL – Bottle Label
- NDC 25021-102-99
- Cefazolin For Injection, USP
- PHARMACY BULK PACKAGE
- NOT FOR DIRECT INFUSION
- 10 grams per Pharmacy Bulk Package
- NOT TO BE DISPENSED AS A UNIT
- FURTHER DILUTION IS REQUIRED
- Rx only
- For Intravenous Use
Ingredients and Appearance
{{#ask: Label Page::Cefazolin sodium |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patients should be advised that allergic reactions, including serious allergic reactions could occur and that serious reactions require immediate treatment and discontinuation of cefazolin. Patients should report to their health care provider any previous allergic reactions to cefazolin, cephalosporins, penicillins or other similar antibacterials.
- Patients should be advised that diarrhea is a common problem caused by antibiotics, which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibacterials, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibacterials. If this occurs, patients should contact a physician as soon as possible.
- Patients should be counseled that antibacterial drugs including cefazolin, should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When cefazolin is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may: (1) decrease the effectiveness of the immediate treatment, and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by cefazolin or other antibacterial drugs in the future.
Precautions with Alcohol
- Alcohol-Cefazolin interaction has not been established.
Brand Names
Look-Alike Drug Names
There is limited information regarding Cefazolin sodium Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Wilson W, Taubert KA, Gewitz M, Lockhart PB, Baddour LM, Levison M; et al. (2007). "Prevention of infective endocarditis: guidelines from the American Heart Association: a guideline from the American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee, Council on Cardiovascular Disease in the Young, and the Council on Clinical Cardiology, Council on Cardiovascular Surgery and Anesthesia, and the Quality of Care and Outcomes Research Interdisciplinary Working Group". Circulation. 116 (15): 1736–54. doi:10.1161/CIRCULATIONAHA.106.183095. PMID 17446442.
- ↑ Verani JR, McGee L, Schrag SJ, Division of Bacterial Diseases, National Center for Immunization and Respiratory Diseases, Centers for Disease Control and Prevention (CDC) (2010). "Prevention of perinatal group B streptococcal disease--revised guidelines from CDC, 2010". MMWR Recomm Rep. 59 (RR-10): 1–36. PMID 21088663.
- ↑ Mermel LA, Allon M, Bouza E, Craven DE, Flynn P, O'Grady NP; et al. (2009). "Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 Update by the Infectious Diseases Society of America". Clin Infect Dis. 49 (1): 1–45. doi:10.1086/599376. PMC 4039170. PMID 19489710.
- ↑ "Cefazolin sodium".
- ↑ "Cefazolin sodium".