Appendix cancer pathophysiology
|
Appendix cancer Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Appendix cancer pathophysiology On the Web |
|
American Roentgen Ray Society Images of Appendix cancer pathophysiology |
|
Risk calculators and risk factors for Appendix cancer pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Soroush Seifirad, M.D.[2]
Overview
The pathophysiology of appendix cancer depends on the histological subtype. There are two major subtypes of appendix cancer, adenocarcinomas and carcinoid tumors. While carcinoid tumors arises from enterochromaffin cells (Kulchitsky cells), which are secretory cells that are normally involved in neuroendocrine hormonal secretions, adenocarcinomas are the result of mutations in mucus producing epithelial cells. Their physiology, pathophysiology, genetic pathways, prognosis as well as epidemiology are different and hence, discussed separately. The progression to adenocarcinoma usually involves the KRAS, APC, TP53, and RAF pathways, while β-catenin, NF1, and MEN1 genes are major contributors of carcinoid tumors progression.
Pathophysiology
Physiology
- The normal physiology of enterochromaffin cells is secretion of serotonin (5-HT), histamine, kallikrein, prostaglandins, and tachykinins.[1]
- Glandular epithelial cells are responsible for mucus production.
Pathogenesis
- The pathophysiology of appendix cancer depends on the histological subtype.[2]
- Adenocarcinoma arises from epithelial glandular cells, which are normally involved in mucous production.
- Carcinoid tumors arise from enterochromaffin cell, which are neuroendocrine cells that are normally involved in secretion of serotonin (5-HT), histamine, kallikrein, prostaglandins, and tachykinins.[1]
- The pathogenesis of appendix cancer is characterized by an initial epithelial dysplasia, followed by the formation of cystic structures and angiolymphatic invasion. Subsequently, in the advanced stages of appendix cancer, tumor cells detach from the primary tumor mass and gain access to the peritoneal cavity.[3]
Genetics
Genes involved in the pathogenesis of carcinoid tumors of appendix include:[4][5]
The development of appendiceal adenocarcinoma is the result of multiple genetic mutations such as:[5]
Associated Conditions
- Conditions associated with appendiceal cancers include:
- Chronic inflammatory disease such as ulcerative colitis
- Familial cancer syndromes:[5]
Gross Pathology
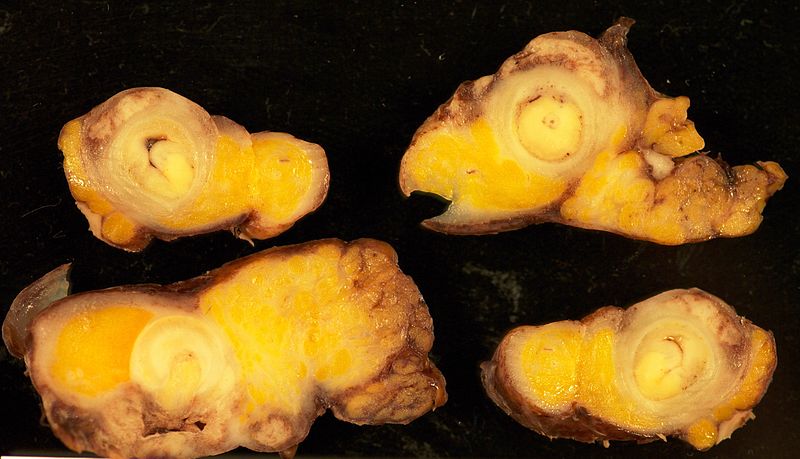 |
- On gross pathology, findings of appendix cancer, include:[3]
- Well-demarcated mass
- Average size between 1 and 5 cm
- Gray or yellowish color
- Deformed appendix
- Adenocarcinoma
- Gray/yellow color
- Cystic structures with angiolymphatic invasion
- Appendix might be buried within the mass
- Carcinoid tumors
- Prevalent at the tip of appendix
- Generally less than 1 cm
- Gray or yellow
- Well-demarcated firm
- Intramural nodules that may narrow or obliterate appendiceal lumen
- Proximal tumors may cause obstruction and appendicitis
- Goblet cell carcinoids
- No gross tumor might be present
- Thickened appendiceal wall
Microscopic Pathology
|
The images below demonstrate different |
 |
 |
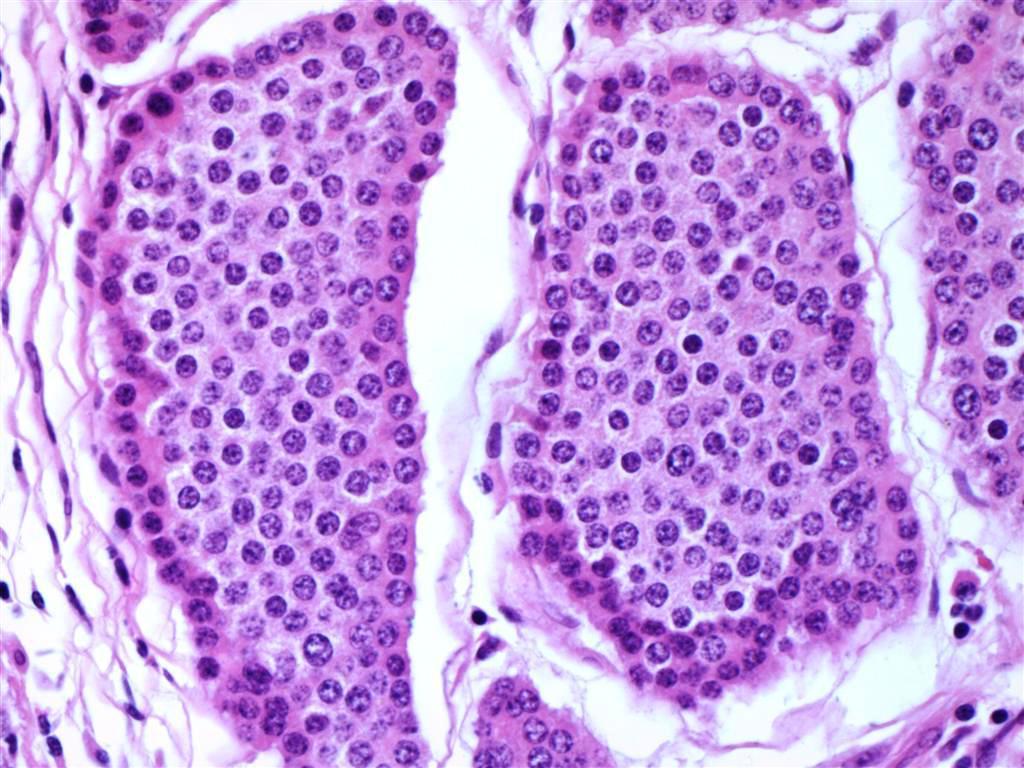 |
- On microscopic histopathological analysis findings will depend on the subtype of appendicular cancer.
- Common histopathological findings, may include:[3]
- Cystic structures
- Angiolymphatic invasion
- Adenocarcinoma
- Intestinal, mucinous or signet ring cell types
- Coexisting acute appendicitis is common
- immunohistochemistry (IHC) might be positive for the following stains:[6]
- MUC 2
- MUC5AC
- CK 8/18
- CK 13
- CK 19
- CK 20
- Carcinoid tumor
- Insular growth pattern of solid islands of uniform polygonal cells with minimal pleomorphism
- Retraction of peripheral tumor cells from stroma
- Angiolymphatic invasion is common
- Granular eosinophilic cytoplasm with either diffusely scattered or peripherally clumped granules
- Two types of well differentiated tumors: EC cell (serotonin producing) and rarely L-cell (enteroglucagon or peptide YY producing)[7]
- IHC might be positive for S100
- Goblet cell
- GCC generally spares mucosa and infiltrates muscularis propria and periappendiceal fat
- Tumor cell clusters
- Crypt-like structures
- Tubules of mucus-secreting cells distended with mucin resembling goblet cells
- Eosinophilic cytoplasm resembling carcinoid tumors
- Pools of extracellular mucin
- Scattered Paneth cells in tumors with crypt like structures
- Extensive perineural invasion
- Carcinomatous growth pattern:
- Cribriform growth pattern, solid sheets of infiltrating signet ring cells
- Nuclear pleomorphism
- Increased mitotic activity
- IHC might be positive for the followings:
- Mucin (mucicarmine, PAS, PAS diastase, Alcian blue)
- CEA
- Cytokeratin (especially CK20)
- lysozyme
- Chromogranin A
- Serotonin
- Synaptophysin
References
- ↑ 1.0 1.1 Gunawardene AR, Corfe BM, Staton CA (2011) Classification and functions of enteroendocrine cells of the lower gastrointestinal tract. Int J Exp Pathol 92 (4):219-31. DOI:10.1111/j.1365-2613.2011.00767.x PMID: 21518048
- ↑ Modlin IM, Lye KD, Kidd M (2003) A 5-decade analysis of 13,715 carcinoid tumors. Cancer 97 (4):934-59. DOI:10.1002/cncr.11105 PMID: 12569593
- ↑ 3.0 3.1 3.2 Ruoff C, Hanna L, Zhi W, Shahzad G, Gotlieb V, Saif MW (2011). "Cancers of the appendix: review of the literatures". ISRN Oncol. 2011: 728579. doi:10.5402/2011/728579. PMC 3200132. PMID 22084738.
- ↑ Modlin IM, Kidd M, Latich I, Zikusoka MN, Eick GN, Mane SM et al. (2006) Genetic differentiation of appendiceal tumor malignancy: a guide for the perplexed. Ann Surg 244 (1):52-60. DOI:10.1097/01.sla.0000217617.06782.d5 PMID: 16794389
- ↑ 5.0 5.1 5.2 Hassan MM, Phan A, Li D, Dagohoy CG, Leary C, Yao JC (2008) Family history of cancer and associated risk of developing neuroendocrine tumors: a case-control study. Cancer Epidemiol Biomarkers Prev 17 (4):959-65. DOI:10.1158/1055-9965.EPI-07-0750 PMID: 18398037
- ↑ Lee MJ, Lee HS, Kim WH, Choi Y, Yang M (2003) Expression of mucins and cytokeratins in primary carcinomas of the digestive system. Mod Pathol 16 (5):403-10. DOI:10.1097/01.MP.0000067683.84284.66 PMID: 12748245
- ↑ Iwafuchi M, Watanabe H, Ajioka Y, Shimoda T, Iwashita A, Ito S (1990) Immunohistochemical and ultrastructural studies of twelve argentaffin and six argyrophil carcinoids of the appendix vermiformis. Hum Pathol 21 (7):773-80. PMID: 2193876