Appendix cancer diagnostic study of choice
|
Appendix cancer Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Appendix cancer diagnostic study of choice On the Web |
|
American Roentgen Ray Society Images of Appendix cancer diagnostic study of choice |
|
Risk calculators and risk factors for Appendix cancer diagnostic study of choice |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Soroush Seifirad, M.D.[2]
Overview
Either CT-scan or MRI are diagnostic study of choice for appendix cancer. Both MRI (particularly diffusion weighted MRI) and CT scan has been recommended as method of choice for disease staging. Histopathology is the gold standard test for the diagnosis as well as classification of appendix cancers.
Diagnostic Study of Choice
Study of choice
- Both MRI (particularly diffusion weighted MRI) and CT scan has been recommended as method of choice for disease staging.
- Peritoneal carcinomatosis index (PCI) using either CT-scan or preferably diffusion weighted MRI has been globally accepted to determine disease burden in appendix cancer.
- Histopathology is the gold standard test for the diagnosis as well as classification of appendix cancer.
- Among the patients who present with clinical signs of carcinoid syndrome, somatostatin scintigraphy with 111-indium-octreotide and serum 5-HIAA levels are the most specific tests for the diagnosis.
- Among the patients who present with clinical signs of appendicitis, the CT-scan is the most sensitive test for diagnosis.
Peritoneal carcinomatosis index
- Peritoneal carcinomatosis index (PCI): A widely accepted metric for assessment of disease border in appendix cancer:[2]
- Estimated by contrast enhanced cross sectional imaging.
- Both MRI and CT scan has been used and are globally accepted imaging modalities.
- Small peritoneal seeding might be difficult to appreciate on CT.
- Sometimes it is challenging to distinguish between tumor and mucin.
- There are reports in favor of diffusion weighted MRI superiority compared to CT in evaluating extent of peritoneal involvement.[1]
- Table and figure below demonstrate abdominal regions as well as scoring system for PCI.
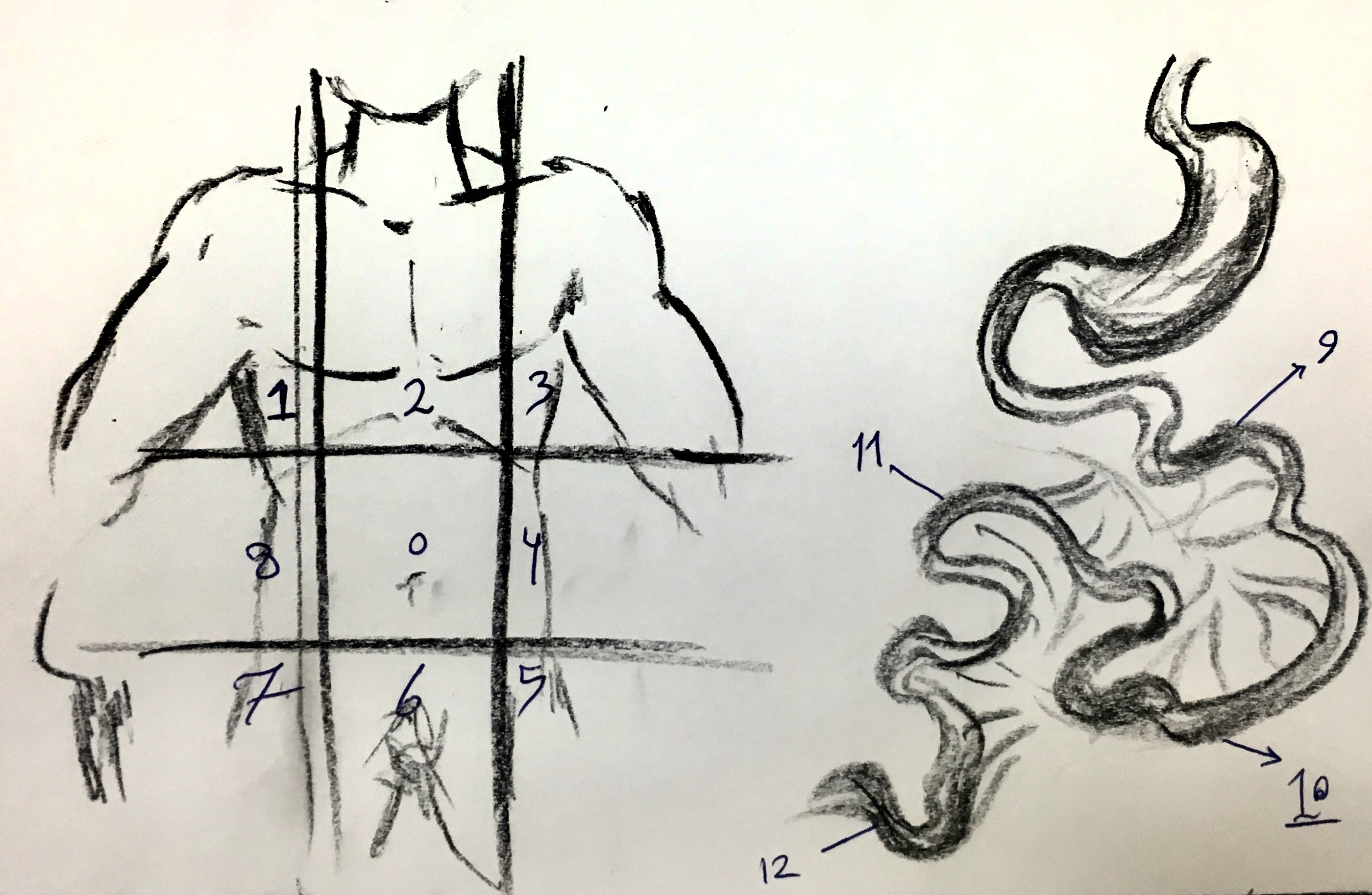
| Lesion Size Score | |
|---|---|
| LS0 | No tumor seen |
| LS1 | Tumor up to 0.5 cm |
| LS2 | Tumor up to 5 cm cm |
| LS3 | Tumor > 5 cm or confluence |
| Maximum Score = 3 | |
| Regions (0-3) | |
| 0 | Central |
| 1 | Right Upper |
| 2 | Epigasterium |
| 3 | Left Upper |
| 4 | Left Flank |
| 5 | Left Lower |
| 6 | Pelvis |
| 7 | Right Upper |
| 8 | Right Flank |
| 9 | Upper Jejunum |
| 10 | Lower Jejunum |
| 11 | Upper Illeum |
| 12 | lower Illeum |
| Maximum Score = 36 | |
| Total Maximum Score = 39 | |
TNM classification of tumors of the appendix
- Major prognostic factors are tumor stage, tumor size, histologic as well genetic characteristics of appendiceal tumors.
- TNM classification of tumors of the appendix has been shown in the table below.[3]
| TNM classification of tumors of the appendix 1 | |
|---|---|
| Primary tumor (T) | |
| Tx | Primary tumor cannot be assessed |
| T0 | No evidence of primary tumor |
| Tis | Carcinoma in situ: intraepithelial or invasion of lamina propria 2 |
| T1 | Tumor invades submucosa |
| T2 | Tumour invades muscularis propria |
| T3 | Tumor invades through muscularis propria into subserosa,
or into non-peritonealized periappendiceal tissue |
| T4 | Tumor directly invades other organs or structures
and/or perforates visceral peritoneum |
| Regional Lymph Nodes (N) | |
| Nx | Regional lymph nodes cannot be assessed |
| N0 | No regional lymph node metastasis |
| N1 | Metastasis in 1 to 3 regional lymph nodes |
| N2 | Metastasis in 4 or more regional lymph nodes |
| Distant Metastasis (M) | |
| Mx | Distant metastasis cannot be assessed |
| M0 | No distant metastasis |
| M1 | Distant metastasis |
| Stage Grouping | |
| Stage 0 | Tis N0 M0 |
| Stage I | T1 N0 M0
T2 N0 M0 |
| Stage II | T3 N0 M0
T4 N0 M0 |
| Stage III | Any T N1 M0
Any T N2 M0 |
| Stage IV | Any T Any N M1 |
| 1 The classification applies only to carcinomas.
2 This includes cancer cells confined within the glandular basement membrane (intraepithelial) or lamina propria (intramucosal) with no extension through muscularis mucosae into submucosa. | |
References
- ↑ 1.0 1.1 Low RN, Barone RM (2012) Combined diffusion-weighted and gadolinium-enhanced MRI can accurately predict the peritoneal cancer index preoperatively in patients being considered for cytoreductive surgical procedures. Ann Surg Oncol 19 (5):1394-1401. DOI:10.1245/s10434-012-2236-3 PMID: 22302265
- ↑ Jacquet P, Sugarbaker PH (1996) Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat Res 82 ():359-74. PMID: 8849962
- ↑ Chapter 5: Tumours of the Appendix - IARC. https://www.iarc.fr/en/publications/pdfs-online/pat-gen/bb2/bb2-chap5.pdf Accessed on January 15, 2019