Abdominal aortic aneurysm surgery
|
Abdominal Aortic Aneurysm Microchapters |
|
Differentiating Abdominal Aortic Aneurysm from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Abdominal aortic aneurysm surgery On the Web |
|
Directions to Hospitals Treating Abdominal aortic aneurysm surgery |
|
Risk calculators and risk factors for Abdominal aortic aneurysm surgery |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Cafer Zorkun, M.D., Ph.D. [2]; Vishnu Vardhan Serla M.B.B.S. [3] Ramyar Ghandriz MD[4]
Overview
Indications for operative repair of an abdominal aortic aneurysm include the presence of symptoms such as back pain, the presence of a rupture or contained rupture, an abdominal aortic aneurysm diameter greater than 5.5 cm, a rate of expansion of greater than 1 cm in one year, and an inflammatory or infectious etiology. Open surgical repair is favored in young patients as an elective procedure, the presence of an expanding aneurysm, symptomatic aneurysm, or ruptured aneurysm. An endovascular repair (EVAR) is favored in older patients and high risk patients or those unfit for open repair. Risk factors for postoperative morbidity and mortality include extensive atheromatous disease, mural calcification, thrombosis, juxtarenal extension of the aneurysm and the presence of inflammatory changes.
Indications
- Symptoms
- Rupture/contained rupture
- Distal emboli
- Pain or bowel obstruction
- AAA diameter > 5.5 cm
- AAA expansion > 1.0 cm in 1 year
- Inflammatory or infectious etiology
Surgery
The treatment options for asymptomatic AAA are immediate repair, surveillance with a view to eventual repair, and a conservative approach. Appropriate patient selection and estimation of the risk of rupture is important, as once rupture occurs, emergency surgery is indicated and mortality from exsanguination is high. There are currently two modes of repair available for an AAA: open aneurysm repair (OR), and endovascular aneurysm repair (EVAR).
Conservative Treatment
- Smoking cessation
- Blood pressure control
- Indicated when repair carries a high risk of mortality and is unlikely to improve life expectancy.
Surveillance
- Surveillance is indicated when the risk of repair exceeds the risk of rupture.
- Although some controversies exist around the world, most vascular surgeons would not consider repair until the aneurysm reached a diameter of 5cm.
- The threshold for repair varies slightly from individual to individual, depending on the balance of risks and benefits when considering repair versus ongoing surveillance.
- The size of an individual's native aorta may influence this, along with the presence of comorbidities that increase operative risk or decrease life expectancy.
Open Repair
An open repair is indicated in the following scenarios:
- Young patients as an elective procedure
- Expanding aneurysms
- Symptomatic aneurysms
- Ruptured aneurysms
Endovascular Repair (EVAR)
An endovascular repair (EVAR) is indicated in:
- Older patients
- High risk patients or those unfit for open repair
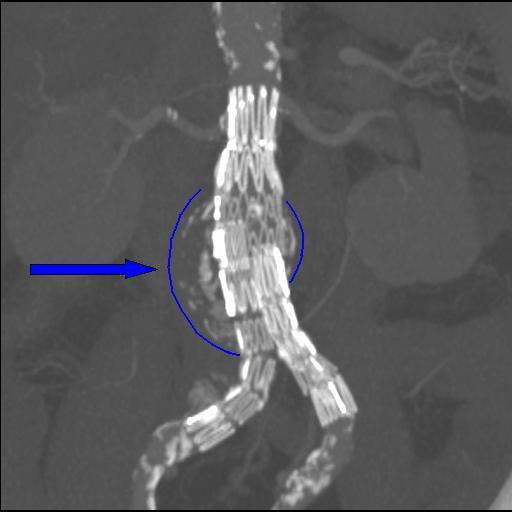
Copyleft image obtained curtesy of http://en.wikipedia.org/wiki/File:Endovasc.jpg; Dr Haudebourg.
Advantages
Advantages of an endovascular repair over an open repair include the following:
- Lower mortality compared to open repair
- Reduced length of stay including a reduced time in the intensive care unit
- Earlier return to normal activity
Disadvantages
Disadvantages of an endovascular repair compared to an open repair include the following:
- Endovascular repair requires more frequent ongoing surveillance and a higher chance of requiring an additional procedure.
Complications
The most common complications related to repair of abdominal aortic aneurysm is related to cardiovascular and pulmonary status. Smokers, patients with co-existent chronic obstructive pulmonary disease and coronary artery disease are more at risk of developing complications such as:
Other complications include:
- Incomplete seal at proximal or distal stent attachment
- Leaking into the sac from branch vessels
- Failure of anastomosis between stent components
- Leakage through graft material
- Graft infection
- Acute renal failure
- Distal embolization
- Wound infection
- Colonic ischemia
- False aneurysm formation
- Aorto-duodenal fistula
- Peri-operative bleeding
- Perianeurysmal aortitis
Pre-Operative Assessment
- The ADAM trial suggested that the "biological age," as reflected by the condition of the vital organs such as the lungs, kidneys, and heart, was more important than the chronological age as a determinant of operative outcome.
- Patients with poor renal and pulmonary function have worse operative outcomes:
- Chronic obstructive pulmonary disease
- Elevated creatinine concentrations
- Electrocardiographic evidence of ischemia
- Aortic factors implicated in postoperative morbidity and mortality include:
- Extensive atheromatous disease
- Mural calcification
- Thrombosis
- Juxtarenal extension of aneurysm
- Inflammatory aortic aneurysms
- The increased risk resulted from:
- Longer suprapenal clamping time
- Need for complex dissection
- Increased hemodynamic stresses
Management of Patients With Peripheral Artery Disease
- 2011 and 2005 ACCF/AHA Guidelines for the Management of Patients With Peripheral Artery Disease (Lower Extremity, Renal, Mesenteric, and Abdominal Aortic)
| Class I |
| "1. Open or endovascular repair of infrarenal AAAs and/or common iliac aneurysms is indicated in patients who are good surgical candidates.[3][4] (Level of Evidence: A)" |
| "2. Periodic long-term surveillance imaging should be performed to monitor for endoleak, confirm graft position, document shrinkage or stability of the excluded aneurysm sac, and determine the need for further intervention in patients who have undergone endovascular repair of infrarenal aortic and/or iliac aneurysms.[3][5] (Level of Evidence: A)" |
| Class IIa |
| "1. Open aneurysm repair is reasonable to perform in patients who are good surgical candidates but who cannot comply with the periodic long-term surveillance required after endovascular repair. (Level of Evidence:C)" |
| Class IIb |
| "1. Endovascular repair of infrarenal aortic aneurysms in patients who are at high surgical or anesthetic risk as determined by the presence of coexisting severe cardiac, pulmonary, and/or renal disease is of uncertain effectiveness.[6] (Level of Evidence:B)" |
Visceral Artery Aneurysms [1]
| Class I |
| "1. Open repair or catheter-based intervention is indicated for visceral aneurysms measuring 2.0 cm in diameter or larger in women of childbearing age who are not pregnant and in patients of either gender undergoing liver transplantation. (Level of Evidence: B)" |
| Class IIa |
| "1. Open repair or catheter-based intervention is probably indicated for visceral aneurysms 2.0 cm in diameter or larger in women beyond childbearing age and in men. (Level of Evidence: B)" |
Symptomatic Aortic or Iliac Aneurysms [1]
| Class I |
| "1. In patients with the clinical triad of abdominal and/or back pain, a pulsatile abdominal mass, and hypotension, immediate surgical evaluation is indicated. (Level of Evidence: B)" |
| "2. In patients with symptomatic aortic aneurysms, repair is indicated regardless of diameter. (Level of Evidence: C)" |
Aortic Aneurysm Rupture [1]
| Class I |
| "1. Patients with infrarenal or juxtarenal AAAs measuring 5.5 cm or larger should undergo repair to eliminate the risk of rupture. (Level of Evidence: B)" |
| "2. Patients with infrarenal or juxtarenal AAAs measuring 4.0 to 5.4 cm in diameter should be monitored by ultrasound or computed tomographic scans every 6 to 12 months to detect expansion. (Level of Evidence: A)" |
| Class III (No Benefit) |
| "1. Intervention is not recommended for asymptomatic infrarenal or juxtarenal AAAs if they measure less than 5.0 cm in diameter in men or less than 4.5 cm in diameter in women. (Level of Evidence: A)" |
| Class IIa |
| "1. Repair can be beneficial in patients with infrarenal or juxtarenal AAAs 5.0 to 5.4 cm in diameter. (Level of Evidence: B)" |
| "2. Repair is probably indicated in patients with suprarenal or type IV thoracoabdominal aortic aneurysms larger than 5.5 to 6.0 cm. (Level of Evidence: B)" |
| "3. In patients with AAAs smaller than 4.0 cm in diameter, monitoring by ultrasound examination every 2 to 3 years is reasonable. (Level of Evidence: B)" |
References
- ↑ 1.0 1.1 1.2 1.3 Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL, Hiratzka LF, Murphy WR, Olin JW, Puschett JB, Rosenfield KA, Sacks D, Stanley JC, Taylor LM, White CJ, White J, White RA, Antman EM, Smith SC, Adams CD, Anderson JL, Faxon DP, Fuster V, Gibbons RJ, Hunt SA, Jacobs AK, Nishimura R, Ornato JP, Page RL, Riegel B (2006). "ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation". Circulation. 113 (11): e463–654. doi:10.1161/CIRCULATIONAHA.106.174526. PMID 16549646. Retrieved 2012-10-09. Unknown parameter
|month=ignored (help) - ↑ "2011 ACCF/AHA Focused Update of the Guideline for the Management of patients with peripheral artery disease (Updating the 2005 Guideline): a report of the American College of Cardiology Foundation/American Heart Association Task Force on practice guidelines". Circulation. 124 (18): 2020–45. 2011. doi:10.1161/CIR.0b013e31822e80c3. PMID 21959305. Retrieved 2012-10-10. Unknown parameter
|month=ignored (help) - ↑ 3.0 3.1 Greenhalgh RM, Brown LC, Powell JT, Thompson SG, Epstein D, Sculpher MJ (2010). "Endovascular versus open repair of abdominal aortic aneurysm". The New England Journal of Medicine. 362 (20): 1863–71. doi:10.1056/NEJMoa0909305. PMID 20382983. Retrieved 2012-11-06. Unknown parameter
|month=ignored (help) - ↑ Blankensteijn JD, de Jong SE, Prinssen M, van der Ham AC, Buth J, van Sterkenburg SM, Verhagen HJ, Buskens E, Grobbee DE (2005). "Two-year outcomes after conventional or endovascular repair of abdominal aortic aneurysms". The New England Journal of Medicine. 352 (23): 2398–405. doi:10.1056/NEJMoa051255. PMID 15944424. Retrieved 2012-11-06. Unknown parameter
|month=ignored (help) - ↑ De Bruin JL, Baas AF, Buth J, Prinssen M, Verhoeven EL, Cuypers PW, van Sambeek MR, Balm R, Grobbee DE, Blankensteijn JD (2010). "Long-term outcome of open or endovascular repair of abdominal aortic aneurysm". The New England Journal of Medicine. 362 (20): 1881–9. doi:10.1056/NEJMoa0909499. PMID 20484396. Retrieved 2012-11-06. Unknown parameter
|month=ignored (help) - ↑ Greenhalgh RM, Brown LC, Powell JT, Thompson SG, Epstein D (2010). "Endovascular repair of aortic aneurysm in patients physically ineligible for open repair". The New England Journal of Medicine. 362 (20): 1872–80. doi:10.1056/NEJMoa0911056. PMID 20382982. Retrieved 2012-11-06. Unknown parameter
|month=ignored (help)