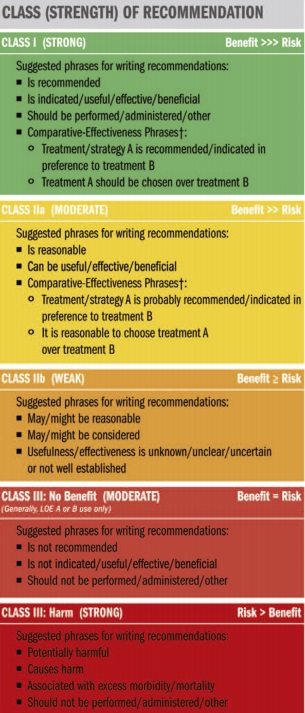
ACC AHA guidelines classification scheme
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
The Class designation is used to indicate whether a therapy is recommended or not and the certainty surrounding that recommendation.
Classification of Recommendations
Class I: Benefit >>> Risk
Conditions for which there is evidence and/or general agreement that a given procedure or treatment is beneficial, useful, and effective.
Class II:
Conditions for which there is conflicting evidence and/or a divergence of opinion about the usefulness/efficacy of a procedure or treatment.
Class IIa: Benefit >> Risk
Weight of evidence/opinion is in favor of usefulness/efficacy.
Class IIb: Benefit ≥ Risk
Usefulness/efficacy is less well established by evidence/opinion.
Class III
Conditions for which there is evidence and/or general agreement that a procedure/treatment is not useful/effective and in some cases may be harmful.
Class III: No Benefit
Is not recommended, not indicated, should not be performed/ administered/ other. Is not useful/beneficial/effective.
Class III: Harm
Potentially harmful, causes harm, associated with excess morbidity/mortality. Should not be performed/administered/other.
Use the Level of Evidence designation to indicate the strength of the data associated with that recommendation.
Level of Evidence
Level of Evidence A:
Data derived from multiple randomized clinical trials or meta-analyses of such studies.
Level of Evidence B:
- Level of Evidence B-R: Data derived from one or more randomized trials or meta-analysis of such studies.
- Level of Evidence B-NR: Data derived from one or more non-randomized trials or meta-analysis of such studies.
Level of Evidence C:
- Level of Evidence C-LD:: Non randomized observational studies with limitations in design or execution or Metanalysis of such studies.
- Level of Evidence C-EO: Consensus opinion of experts based on clinical experience.
Applying Classification of Recommendations and Level of Evidence
2016 Classification of Recommendations and Level of Evidence
| “ | ” |
2014 Classification of Recommendations and Level of Evidence
| “ |
 |
” |