Fluvastatin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Editor-In-Chief: C. Michael Gibson, M.S., M.D. [2], Sheng Shi, M.D. [3]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Fluvastatin is a HMG-COA Reductase Inhibitor that is FDA approved for the {{{indicationType}}} of Hypercholesterolemia (Heterozygous Familial and Nonfamilial) and Mixed Dyslipidemia, Secondary Prevention of Cardiovascular Disease. Common adverse reactions include indigestion, nausea, headache.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Fluvastatin Tablet
- Dose range: 20 mg to 80 mg/ day.
- LESCOL/LESCOL XL can be administered orally as a single dose, with or without food.
- Do not break, crush or chew LESCOL XL tablets or open LESCOL capsules prior to administration.
- Do not take two LESCOL 40 mg capsules at one time.
- Since the maximal effect of a given dose is seen within 4 weeks, periodic lipid determinations should be performed at this time and dosage adjusted according to the patient’s response to therapy and established treatment guidelines.
- For patients requiring LDL-C reduction to a goal of ≥25%, the recommended starting dose is 40 mg as one capsule in the evening, 80 mg as one LESCOL XL tablet administered as a single dose at any time of the day or 80 mg in divided doses of the 40 mg capsule given twice daily. For patients requiring LDL-C reduction to a goal of <25% a starting dose of 20 mg may be used.
Adult Patients with Hypercholesterolemia (Heterozygous Familial and Nonfamilial) and Mixed Dyslipidemia
- Indication
- as an adjunct to diet to reduce elevated total cholesterol (Total-C), low-density lipoprotein cholesterol (LDL-C), triglyceride (TG) and apolipoprotein B (Apo B) levels, and to increase high-density lipoprotein cholesterol (HDL-C) in patients with primary hypercholesterolemia and mixed dyslipidemia (Fredrickson Type IIa and IIb).
- as an adjunct to diet to reduce Total-C, LDL-C, and Apo B levels in adolescent boys and adolescent girls who are at least one year post-menarche, 10 to 16 years of age, with heterozygous familial hypercholesterolemia and the following findings are present:
- LDL-C remains ≥ 190 mg/dL or
- LDL-C remains ≥ 160 mg/dL and:
there is a positive family history of premature cardiovascular disease or two or more other cardiovascular disease risk factors are present The NCEP classification of cholesterol levels in pediatric patients with a familial history of hypercholesterolemia or premature CVD is summarized below.

Children treated with fluvastatin in adolescence should be re-evaluated in adulthood and appropriate changes made to their cholesterol-lowering regimen to achieve adult treatment goals.
- Dosing information (LESCOL XL tablet)
- 80 mg PO qd at any time of the day.
- Dosing information (LESCOL capsule)
- Recommended starting dosage for LESCOL:
- 40 mg PO qd in the evening
- or 40 mg PO bid
- Do not take two LESCOL 40 mg capsules at one time.
Use with Cyclosporine
- Do not exceed a dose of 20 mg b.i.d. LESCOL in patients taking cyclosporine [see Drug Interactions 7.1].
Use with Fluconazole
- Do not exceed a dose of 20 mg b.i.d. LESCOL in patients taking fluconazole [see Drug Interactions 7.2].
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information about the Non-Guideline-Supported use.
Non–Guideline-Supported Use
Acute coronary syndrome
- Dosing information
- Higher doses 17000936
Prophylaxis treatment of Atrial fibrillation
- Dosing information
- Not applicable 18242276
Prophylaxis treatment of Primary Cardiovascular event risk
- Dosing information
- Not applicable 17130382
Reduction of Cardiovascular event risk in patients with renal impairment
- Dosing information
- 20 mg- 80 mg/day16889855
Coronary artery bypass graft
- Dosing information
- 10-20 mg/day 1074212
chronic heart failure
- Dosing information
- Not appllicable 11074212
Nephrotic syndrome
- Dosing information
- 40 mg every evening 10460940
Percutaneous coronary intervention
- Dosing information
- 40 mg bid 14697476
Prophylaxis of Postoperative cardiac complication
- Dosing information
- 80 mg/day 19726772
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Pediatric Patients (10-16 years of age) with Heterozygous Familial Hypercholesterolemia
- Dosing information (capsule)
- Recommended starting dosage: 20 mg PO qd , up to a maximum daily dose administered either as LESCOL capsules 40 mg PO bid
- Doses should be individualized according to the goal of therapy [see NCEP Pediatric Panel Guidelines and CLINICAL STUDIES (14)]1.
- Dosing information (tablet)
- 80 mg PO qd
- Doses should be individualized according to the goal of therapy [see NCEP Pediatric Panel Guidelines and CLINICAL STUDIES (14)].
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information about Off-Label Guideline-Supported Use of Fluvastatin sandbox in pediatric patients.
Non–Guideline-Supported Use
There is limited information about Off-Label Non–Guideline-Supported Use of Fluvastatin sandbox in pediatric patients.
Contraindications
Hypersensitivity to any Component of this Medication
LESCOL and LESCOL XL are contraindicated in patients with hypersensitivity to any component of this medication.
Active Liver Disease
LESCOL and LESCOL XL are contraindicated in patients with active liver disease or unexplained, persistent elevations in serum transaminases [see Warnings and Precautions (5.2)].
Pregnancy
LESCOL and LESCOL XL are contraindicated in women who are pregnant or may become pregnant. Serum cholesterol and triglycerides increase during normal pregnancy, and cholesterol or cholesterol derivatives are essential for fetal development. LESCOL and LESCOL XL may cause fetal harm when administered to pregnant women. Atherosclerosis is a chronic process and the discontinuation of lipid-lowering drugs during pregnancy should have little impact on the outcome of long-term therapy of primary hypercholesterolemia.
LESCOL and LESCOL XL should be administered to women of childbearing age only when such patients are highly unlikely to conceive and have been informed of the potential hazards. If the patient becomes pregnant while taking this drug, LESCOL and LESCOL XL should be discontinued and the patient should be apprised of the potential hazard to the fetus [see Use In Specific Populations (8.1)].
Nursing Mothers
Fluvastatin is secreted into the breast milk of animals and because HMG-CoA reductase inhibitors have the potential to cause serious adverse reactions in nursing infants, women who require treatment with LESCOL or LESCOL XL should be advised not to breastfeed their infants [see Use In Specific Populations (8.3)].
Warnings
Skeletal Muscle
Rhabdomyolysis with acute renal failure secondary to myoglobinuria have been reported with LESCOL/LESCOL XL and other drugs in this class.
LESCOL/LESCOL XL should be prescribed with caution in patients with predisposing factors for myopathy. These factors include advanced age (>65 years), renal impairment, and inadequately treated hypothyroidism.
The risk of myopathy and/or rhabdomyolysis with statins is increased with concurrent therapy with cyclosporine, erythromycin, fibrates or niacin. Myopathy was not observed in a clinical trial in 74 patients involving patients who were treated with LESCOL/LESCOL XL together with niacin. Isolated cases of myopathy have been reported during post-marketing experience with concomitant administration of LESCOL/LESCOL XL and colchicine. No information is available on the pharmacokinetic interaction between LESCOL/LESCOL XL and colchicine. Uncomplicated myalgia has also been reported in LESCOL-treated patients [see Adverse Reactions (6)]. In clinical trials, uncomplicated myalgia has been observed infrequently in patients treated with LESCOL at rates indistinguishable from placebo. myopathy, defined as muscle aching or muscle weakness in conjunction with increases in CPK values to greater than 10 times the upper limit of normal, was less than 0.1% in fluvastatin clinical trials. myopathy should be considered in any patient with diffuse myalgias, muscle tenderness or weakness, and/or marked elevation of CPK.
There have been rare reports of immune-mediated necrotizing myopathy (IMNM), an autoimmune myopathy, associated with statin use. IMNM is characterized by: proximal muscle weakness and elevated serum creatine kinase, which persist despite discontinuation of statin treatment; muscle biopsy showing necrotizing myopathy without significant inflammation; improvement with immunosuppressive agents.
All patients should be advised to promptly report to their physician unexplained muscle pain, tenderness, or weakness, particularly if accompanied by malaise or fever or if muscle signs and symptoms persist after discontinuing LESCOL/LESCOL XL.
LESCOL/LESCOL XL therapy should be discontinued if markedly elevated CPK levels occur or myopathy is diagnosed or suspected. LESCOL/LESCOL XL therapy should also be temporarily withheld in any patient experiencing an acute or serious condition predisposing to the development of renal failure secondary to rhabdomyolysis, e.g., sepsis; hypotension; major surgery; trauma; severe metabolic, endocrine, or electrolyte disorders; or uncontrolled epilepsy.
Liver Enzymes
Increases in serum transaminases (aspartate aminotransferase [AST]/serum glutamic-oxaloacetic transaminase, or alanine aminotransferase [ALT]/serum glutamic-pyruvic transaminase) have been reported with HMG-CoA reductase inhibitors, including LESCOL/LESCOL XL. In most cases, the elevations were transient and resolved or improved on continued therapy or after a brief interruption in therapy.
Approximately 1.1% of patients treated with LESCOL capsules in worldwide trials developed dose-related, persistent elevations of serum transaminase levels to more than 3 times the upper limit of normal. Fourteen of these patients (0.6%) were discontinued from therapy. In all clinical trials, a total of 33/2969 patients (1.1%) had persistent transaminase elevations with an average LESCOL exposure of approximately 71.2 weeks; 19 of these patients (0.6%) were discontinued. The majority of patients with these abnormal biochemical findings were asymptomatic.
In a pooled analysis of all placebo-controlled studies in which LESCOL capsules were used, persistent transaminase elevations (greater than 3 times the upper limit of normal [ULN] on two consecutive weekly measurements) occurred in 0.2%, 1.5%, and 2.7% of patients treated with daily doses of 20, 40, and 80 mg (titrated to 40 mg twice daily) LESCOL capsules, respectively. Ninety-one percent of the cases of persistent liver function test abnormalities (20 of 22 patients) occurred within 12 weeks of therapy and in all patients with persistent liver function test abnormalities there was an abnormal liver function test present at baseline or by Week 8.
In the pooled analysis of the 24-week controlled trials, persistent transaminase elevation occurred in 1.9%, 1.8% and 4.9% of patients treated with LESCOL XL 80 mg, LESCOL 40 mg and LESCOL 40 mg twice daily, respectively. In 13 of 16 patients treated with LESCOL XL the abnormality occurred within 12 weeks of initiation of treatment with LESCOL XL 80 mg.
It is recommended that liver enzyme tests be performed prior to the initiation of LESCOL/LESCOL XL, and if signs or symptoms of liver injury occur.
There have been rare postmarketing reports of fatal and non-fatal hepatic failure in patients taking [statins]], including fluvastatin. If serious liver injury with clinical symptoms and/or hyperbilirubinemia or jaundice occurs during treatment with LESCOL/LESCOL XL, promptly interrupt therapy. If an alternate etiology is not found do not restart LESCOL/LESCOL XL.
In very rare cases, possibly drug-related hepatitis was observed that resolved upon discontinuation of treatment.1 Active liver disease or unexplained serum transaminase elevations are contraindications to the use of LESCOL and LESCOL XL [see Contraindications (4) and Warnings and Precautions (5.2)]. Caution should be exercised when LESCOL is administered to patients with a history of liver disease or heavy alcohol ingestion [see Clinical Pharmacology (12.3)]. Such patients should be closely monitored.
Endocrine Effects
Increases in HbA1c and fasting serum glucose levels have been reported with HMG-CoA reductase inhibitors, including LESCOL/LESCOL XL.
Statins interfere with cholesterol synthesis and lower circulating cholesterol levels and, as such, might theoretically blunt adrenal or gonadal steroid hormone production.
LESCOL/LESCOL XL exhibited no effect upon non-stimulated cortisol levels and demonstrated no effect upon thyroid metabolism as assessed by measurement of thyroid stimulating hormone (TSH). Small declines in total serum testosterone have been noted in treated groups, but no commensurate elevation in LH occurred, suggesting that the observation was not due to a direct effect upon testosterone production. No effect upon FSH in males was noted. Due to the limited number of premenopausal females studied to date, no conclusions regarding the effect of LESCOL/LESCOL XL upon female sex hormones may be made.
Two clinical studies in patients receiving fluvastatin at doses up to 80 mg daily for periods of 24 to 28 weeks demonstrated no effect of treatment upon the adrenal response to ACTH stimulation. A clinical study evaluated the effect of LESCOL at doses up to 80 mg daily for 28 weeks upon the gonadal response to HCG stimulation. Although the mean total testosterone response was significantly reduced (p less than 0.05) relative to baseline in the 80 mg group, it was not significant in comparison to the changes noted in groups receiving either 40 mg of LESCOL or placebo.
Patients treated with LESCOL/LESCOL XL who develop clinical evidence of endocrine dysfunction should be evaluated appropriately. Caution should be exercised if a statin or other agent used to lower cholesterol levels is administered to patients receiving other drugs (e.g. ketoconazole, spironolactone, cimetidine) that may decrease the levels of endogenous steroid hormones.
CNS Toxicity
CNS effects, as evidenced by decreased activity, ataxia, loss of righting reflex, and ptosis were seen in the following animal studies: the 18-month mouse carcinogenicity study at 50 mg/kg/day, the 6-month dog study at 36 mg/kg/day, the 6-month hamster study at 40 mg/kg/day, and in acute, high-dose studies in rats and hamsters (50 mg/kg), rabbits (300 mg/kg) and mice (1500 mg/kg). CNS toxicity in the acute high-dose studies was characterized (in mice) by conspicuous vacuolation in the ventral white columns of the spinal cord at a dose of 5000 mg/kg and (in rats) by edema with separation of myelinated fibers of the ventral spinal tracts and sciatic nerve at a dose of 1500 mg/kg. CNS toxicity, characterized by periaxonal vacuolation, was observed in the medulla of dogs that died after treatment for 5 weeks with 48 mg/kg/day; this finding was not observed in the remaining dogs when the dose level was lowered to 36 mg/kg/day. CNS vascular lesions, characterized by perivascular hemorrhages, edema, and mononuclear cell infiltration of perivascular spaces, have been observed in dogs treated with other members of this drug class. No CNS lesions have been observed after chronic treatment for up to 2 years with fluvastatin in the mouse (at doses up to 350 mg/kg/day), rat (up to 24 mg/kg/day), or dog (up to 16 mg/kg/day).
Prominent bilateral posterior Y suture lines in the ocular lens were seen in dogs after treatment with 1, 8, and 16 mg/kg/day for 2 years.
Adverse Reactions
Clinical Trials Experience
The following serious adverse reactions are discussed in greater detail in other sections of the label:
Rhabdomyolysis with myoglobinuria and acute renal failure and myopathy (including myositis) [see Warnings and Precautions (5.1)]. Liver Enzyme Abnormalities [see Warnings and Precautions (5.2)].
Clinical Studies Experience in Adult Patients
Because clinical studies on LESCOL/LESCOL XL are conducted in varying study populations and study designs, the frequency of adverse reactions observed in the clinical studies of LESCOL/LESCOL XL cannot be directly compared with that in the clinical studies of other statins and may not reflect the frequency of adverse reactions observed in clinical practice.
In the LESCOL placebo-controlled clinical trials databasese of 2326 patients treated with LESCOL1 (age range 18-75 years, 44% women, 94% Caucasians, 4% Blacks, 2% other ethnicities) with a median treatment duration of 24 weeks, 3.4% of patients on LESCOL and 2.3% patients on placebo discontinued due to adverse reactions regardless of causality. The most common adverreactions that led to treatment discontinuation and occurred at an incidence greater than placebo were: transaminase increased (0.8%), upper abdominal pain (0.3%), dyspepsia (0.3%), fatigue (0.2%) and diarrhea (0.2%).
In the LESCOL XL database of controlled clinical trials of 912 patients treated with LESCOL XL (age range 21-87 years, 52% women, 91% Caucasians, 4% Blacks, 5% other ethnicities) with a median treatment duration of 24 weeks, 3.9% of patients on LESCOL XL discontinued due to adverse reactions regardless of causality. The most common adverse reactions that led to treatment discontinuation were abdominal pain (0.7%), diarrhea (0.5%), nausea (0.4%), dyspepsia (0.4%) and chest pain (0.3%).
Clinically relevant adverse experiences occurring in the LESCOL and LESCOL XL controlled studies with a frequency greater than 2%, regardless of causality, included the following:
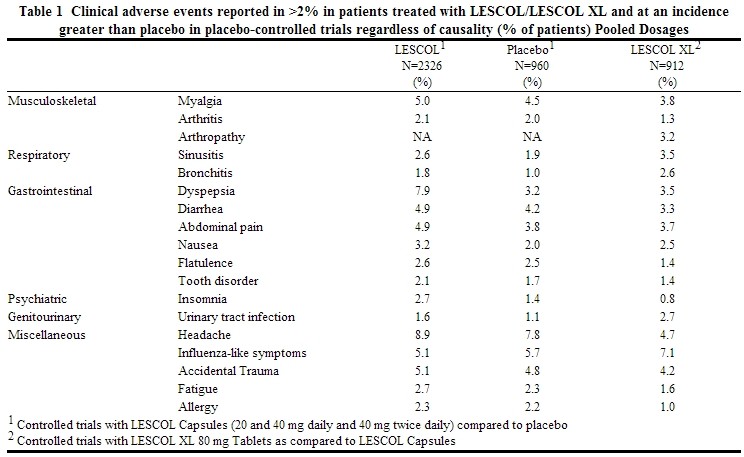
LESCOL Intervention Prevention Study
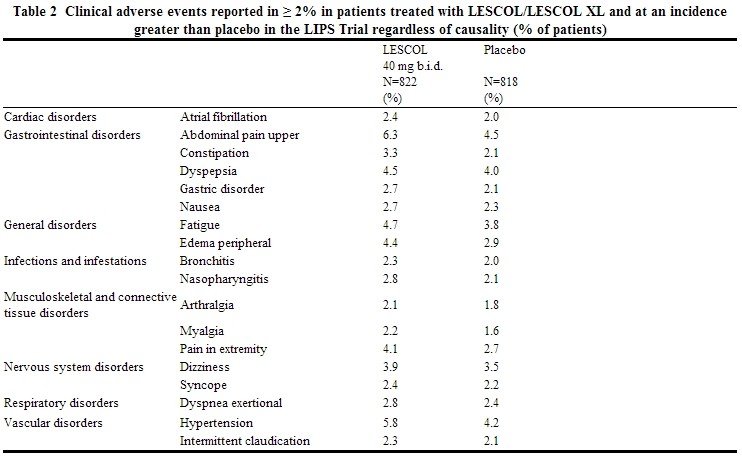
In the LESCOL Intervention Prevention Study (LIPS), the effect of LESCOL 40 mg, administered twice daily on the risk of recurrent cardiac events was assessed in 1677 patients with CHD who had undergone a percutaneous coronary intervention (PCI) procedure. This was a multicenter, randomized, double-blind, placebo-controlled study, patients were treated with dietary/lifestyle counseling and either LESCOL 40 mg (n=844) or placebo (n=833) given twice daily for a median of 3.9 years [see Clinical Studies (14.3)].
Clinical Studies Experience in Pediatric Patients
In patients aged less than 18 years, efficacy and safety have not been studied for treatment periods longer than two years.
In two open-label, uncontrolled studies, 66 boys and 48 girls with heterozygous familial hypercholesterolemia ( 9-16 years of age, 80% Caucasian, 19% Other [ mixed ethnicity], 1% Asians) were treated with fluvastatin sodium administered as LESCOL capsules 20 mg -40 mg twice daily, or LESCOL XL 80 mg extended-release tablet [see Clinical Studies (14.2) and Use In Specific Populations (8.4)].
Postmarketing Experience
Because adverse reactions from spontaneous reports are reported voluntarily from a population of uncertain size, it is generally not possible to reliably estimate their frequency or establish a causal relationship to drug exposure. The following effects have been reported with drugs in this class. Not all the effects listed below have necessarily been associated with fluvastatin sodium therapy.
Musculoskeletal: muscle cramps, myalgia, myopathy, Rhabdomyolysis, arthralgias, muscle spasms, muscle weakness, myositis.
There have been rare reports of immune-mediated necrotizing myopathy associated with statin use [see Warnings and Precautions (5.1)].
Neurological: dysfunction of certain cranial nerves (including alteration of taste, impairment of extra-ocular movement, facial paresis), tremor, dizziness, vertigo, paresthesia, hypoesthesia, dysesthesia, peripheral neuropathy, peripheral nerve palsy.
There have been rare postmarketing reports of cognitive impairment (e.g., memory loss, forgetfulness, amnesia, memory impairment, confusion) associated with statin use. These cognitive issues have been reported for all statins. The reports are generally nonserious, and reversible upon statin discontinuation, with variable times to symptom onset (1 day to years) and symptom resolution (median of 3 weeks).
Psychiatric: anxiety, insomnia, depression, psychic disturbances
Hypersensitivity Reactions: An apparent hypersensitivity syndrome has been reported rarely which has included one or more of the following features: anaphylaxis, angioedema, lupus erythematosus-like syndrome, polymyalgia rheumatica, vasculitis, purpura, thrombocytopenia, leukopenia, hemolytic anemia, positive ANA, ESR (erythrocyte sedimentation rate) increase, eosinophilia, arthritis, arthralgia, urticaria, asthenia, photosensitivity reaction, fever,chills,flushing, malaise , dyspnea, toxic epidermal necrolysis, erythema multiforme, including Stevens-Johnson syndrome.
Gastrointestinal: pancreatitis, hepatitis, including chronic active hepatitis, cholestatic jaundice, fatty change in liver, cirrhosis, fulminant hepatic necrosis, hepatoma, anorexia,vomiting, fatal and non-fatal hepatic failure.
Skin: rash, dermatitis, including bullous dermatitis, eczema, alopecia,pruritus, a variety of skin changes (e.g. nodules, discoloration, dryness of skin/mucous membranes, changes to hair/nails).
Reproductive: gynecomastia, loss of libido, erectile dysfunction.
Eye: progression of cataracts (lens opacities), ophthalmoplegia.
Laboratory abnormalities: elevated transaminases, alkaline phosphatase, gamma-glutamyl transpeptidase and bilirubin; thyroid function abnormalities.
Drug Interactions
Cyclosporine
Cyclosporine coadministration increases fluvastatin exposure. Therefore, in patients taking cyclosporine, therapy should be limited to LESCOL 20 mg twice daily [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
Fluconazole
Administration of fluvastatin 40 mg single dose to healthy volunteers pre-treated with fluconazole for 4 days results in an increase of fluvastatin exposure. Therefore, in patients taking fluconazole, therapy should be limited to LESCOL 20 mg twice daily [see Clinical Pharmacology (12.3)].
Gemfibrozil
Due to an increased risk of myopathy/rhabdomyolysis when HMG-CoA reductase inhibitors are coadministered with gemfibrozil, concomitant administration of LESCOL/LESCOL XL with gemfibrozil should be avoided.
Other Fibrates
Because it is known that the risk of myopathy during treatment with HMG-CoA reductase inhibitors is increased with concurrent administration of other fibrates, LESCOL/LESCOL XL should be administered with caution when used concomitantly with other fibrates [see Warnings and Precautions (5.1) and Clinical Pharmacology (12.3)].
Niacin
The risk of skeletal muscle effects may be enhanced when LESCOL is used in combination with lipid-modifying doses (≥1 g/day) of niacin; a reduction in LESCOL dosage should be considered in this setting [see Warnings and Precautions (5.1)].
Glyburide
Concomitant administration of fluvastatin and glyburide increased glyburide exposures. Patients on concomitant therapy of glyburide and fluvastatin should continue to be monitored appropriately [see Clinical Pharmacology (12.3)].
Phenytoin
Concomitant administration of fluvastatin and phenytoin increased phenytoin exposures. Patients should continue to be monitored appropriately when fluvastatin therapy is initiated or when fluvastatin dose is changed [see Clinical Pharmacology (12.3)].
Warfarin
Bleeding and/or increased prothrombin times have been reported in patients taking coumarin anticoagulants concomitantly with other HMG-CoA reductase inhibitors. Therefore, patients receiving warfarin-type anticoagulants should have their prothrombin times closely monitored when fluvastatin sodium is initiated or the dosage of fluvastatin sodium is changed.
Colchicine
Cases of myopathy, including rhabdomyolysis, have been reported with fluvastatin coadministered with colchicine, and caution should be exercised when prescribing fluvastatin with colchicine.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): X LESCOL/LESCOL XL is contraindicated in women who are or may become pregnant [see Contraindications (4)].
Lipid lowering drugs are contraindicated during pregnancy, because cholesterol and cholesterol derivatives are needed for normal fetal development. Serum cholesterol and triglycerides increase during normal pregnancy. Atherosclerosisis a chronic process, and discontinuation of lipid-lowering drugs during pregnancy should have little impact on long-term outcomes of primary hypercholesterolemia therapy.
There are no adequate and well-controlled studies of use with LESCOL/LESCOL XL during pregnancy. Rare reports of congenital anomalies have been received following intrauterine exposure to other statins. In a review2 of about 100 prospectively followed pregnancies in women exposed to other statins, the incidences of congenital anomalies, spontaneous abortions, and fetal deaths/stillbirths did not exceed the rate expected in the general population. The number of cases is adequate only to exclude a 3- to 4-fold increase in congenital anomalies over background incidence. In 89% of prospectively followed pregnancies, drug treatment was initiated prior to pregnancy and was discontinued at some point in the first trimester when pregnancy was identified.
Teratology studies with fluvastatin in rats and rabbits showed maternal toxicity at high dose levels, but there was no evidence of embryotoxic or teratogenic potential [see Non-Clinical Toxicology (13)].
LESCOL or LESCOL XL should be administered to women of child-bearing potential only when such patients are highly unlikely to conceive and have been informed of the potential hazards. If a woman becomes pregnant while taking LESCOL or LESCOL XL, the drug should be discontinued and the patient advised again as to the potential hazards to the fetus.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Fluvastatin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Fluvastatin during labor and delivery.
Nursing Mothers
Based on animal data, fluvastatin is present in breast milk in a 2:1 ratio (milk:plasma). Because of the potential for serious adverse reactions in nursing infants, nursing women should not take LESCOL or LESCOL XL [see Contraindications (4)].
Pediatric Use
The safety and efficacy of LESCOL and LESCOL XL in children and adolescent patients 9-16 years of age with heterozygous familial hypercholesterolemia have been evaluated in open-label, uncontrolled clinical trials for a duration of two years. The most common adverse events observed were influenza and infections. In these limited uncontrolled studies, there was no detectable effect on growth or sexual maturation in the adolescent boys or on menstrual cycle length in girls [see Clinical Studies (14.2), Adverse Reactions (6.3) and Dosage and Administration (2.2)]. Adolescent females should be counseled on appropriate contraceptive methods while on LESCOL therapy [see Contraindications (4)].
Geriatic Use
Fluvastatin exposures were not significantly different between the nonelderly and elderly populations (age ≥ 65 years) [see Clinical Pharmacology (12.3)]. Since advanced age (greater than 65 years) is a predisposing factor for myopathy, LESCOL/LESCOL XL should be prescribed with caution in the elderly.
Gender
In a study evaluating the effect of age and gender on fluvastatin pharmacokinetics, there were no significant differences in fluvastatin exposures between males and females, except between younger females and younger males (both ages 21-49 years), where there was an approximate 30% increase in AUC in females. Adjusting for body weight decreases the magnitude of the differences seen. For LESCOL XL, the AUC increases 67% and 77% for women compared to men under fasted and high- fat meal fed conditions, respectively.
Race
There is no FDA guidance on the use of Fluvastatin with respect to specific racial populations.
Renal Impairment
Dose adjustments for mild to moderate renal impairment are not necessary. Fluvastatin has not been studied at doses greater than 40 mg in patients with severe renal impairment; therefore caution should be exercised when treating such patients at higher doses [see Clinical Pharmacology(12.3)].
Hepatic Impairment
LESCOL and LESCOL XL are contraindicated in patients with active liver disease or unexplained, persistent elevations in serum transaminases [see Clinical Pharmacology (12.3)].
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Fluvastatin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Fluvastatin in patients who are immunocompromised.
Administration and Monitoring
Administration
Oral
Monitoring
There is limited information about the drug monitoring.
IV Compatibility
FDA Package Insert for Fluvastatin contains no information regarding IV compatibility.
Overdosage
To date, there has been limited experience with overdosage of fluvastatin. If an overdose occurs, it should be treated symptomatically with laboratory monitoring and supportive measures should be instituted as required. The dialyzability of fluvastatin sodium and of its metabolites in humans is not known at present [see Warnings and Precautions (5)].
In the pediatric population, there have been reports of overdosage with fluvastatin sodium in children including a 2 year-old and the other 3 years of age, either of whom may have possibly ingested fluvastatin sodium. The maximum amount of fluvastatin sodium that could have been ingested was 80 mg (4 x 20 mg capsules). Vomiting was induced by ipecac in both children and no capsules were noted in their emesis. Neither child experienced any adverse symptoms and both recovered from the incident without problems.
In the postmarketing experience there have been reports of accidental ingestion of LESCOL tablets in infants up to 3 years of age. In one case, increased serum CPK values were noted. There have been reports of intentional overdose in adolescents with the development of hepatic enzyme elevations, convulsions and gastroenteritis/vomiting/diarrhea. One case of intentional overdose as suicide attempt in a 15 year-old female reported ingestion of 2,800 mg LESCOL XL with hepatic enzyme elevation.
Pharmacology
Mechanism of Action
LESCOL is a competitive inhibitor of HMG-CoA reductase, the rate limiting enzyme that converts 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) to mevalonate, a precursor of sterols, including cholesterol. The inhibition of cholesterol biosynthesis reduces the cholesterol in hepatic cells, which stimulates the synthesis of LDL receptors and thereby increases the uptake of LDL particles. The end result of these biochemical processes is a reduction of the plasma cholesterol concentration.
Structure
LESCOL is a water-soluble cholesterol lowering agent which acts through the inhibition of 3-hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase.
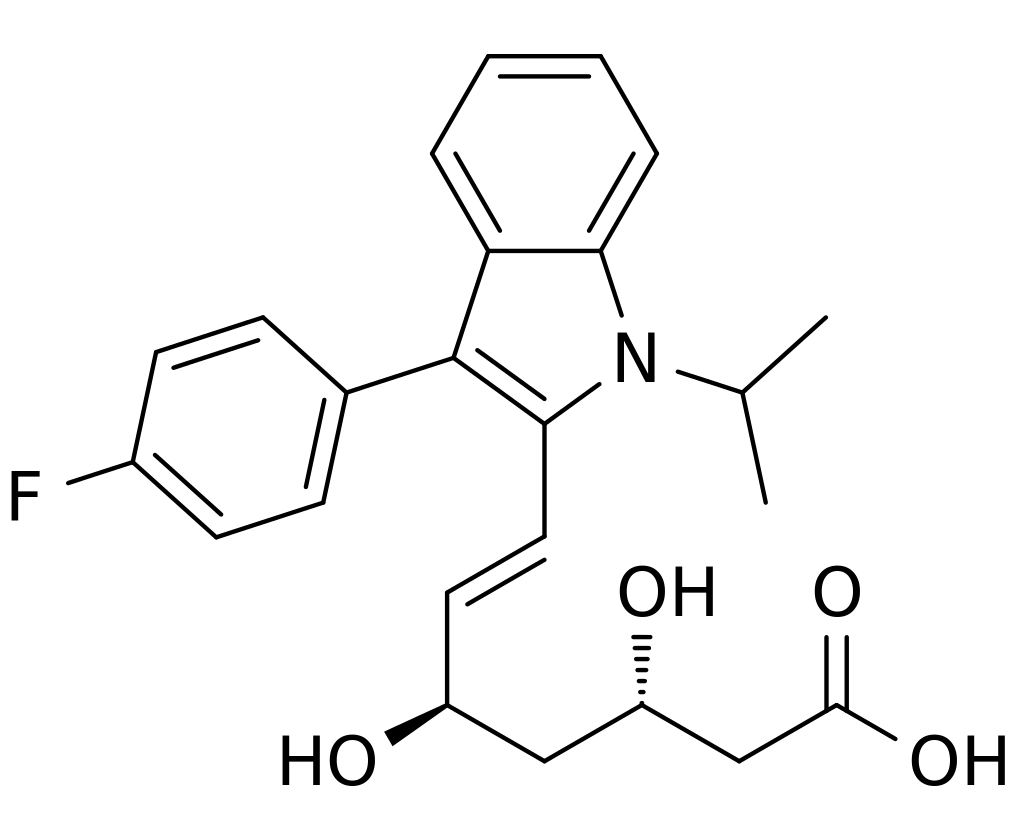
Fluvastatin sodium is [R*,S*-(E)]-(±)-7-[3-(4-fluorophenyl)-1-(1-methylethyl)-1H-indol-2-yl]-3,5-dihydroxy-6-heptenoic acid, monosodium salt. The empirical formula of fluvastatin sodium is C24H25FNO4•Na, its molecular weight is 433.46 and its structural formula is:

This molecular entity is the first entirely synthetic HMG-CoA reductase inhibitor, and is in part structurally distinct from the fungal derivatives of this therapeutic class.
Fluvastatin sodium is a white to pale yellow, hygroscopic powder soluble in water, ethanol and methanol. LESCOL is supplied as capsules containing fluvastatin sodium, equivalent to 20 mg or 40 mg of fluvastatin, for oral administration. LESCOL XL is supplied as extended-release tablets containing fluvastatin sodium, equivalent to 80 mg of fluvastatin, for oral administration.
Active Ingredient: fluvastatin sodium
Inactive Ingredients in capsules: calcium carbonate, gelatin, magnesium stearate, microcrystalline cellulose, pregelatinized starch (corn), red iron oxide, sodium bicarbonate, talc, titanium dioxide, yellow iron oxide, and other ingredients.
Capsules may also include: benzyl alcohol, black iron oxide, butylparaben, carboxymethylcellulose sodium, edetate calcium disodium, methylparaben, propylparaben, silicon dioxide, sodium lauryl sulfate, and sodium propionate.
Inactive Ingredients in extended-release tablets: microcrystalline cellulose, hydroxypropyl cellulose, hydroxypropyl methyl cellulose, potassium bicarbonate, povidone, magnesium stearate, yellow iron oxide, titanium dioxide and polyethylene glycol 8000.
Pharmacodynamics
FDA Package Insert for Fluvastatin contains no information regarding pharmacodynamics.
Pharmacokinetics
Absorption:
Following oral administration of the capsule, fluvastatin reaches peak concentrations in less than 1 hour. The absolute bioavailability is 24% (range 9%-50%) after administration of a 10 mg dose.
At steady state, administration of fluvastatin with the evening meal results in a 50% decrease in Cmax, an 11% decrease in AUC, and a more than two-fold increase in tmaxas compared to administration 4 hours after the evening meal. No significant differences in the lipid-lowering effects were observed between the two administrations. After single or multiple doses above 20 mg, fluvastatin exhibits saturable first-pass metabolism resulting in more than dose proportional plasma fluvastatin concentrations.
Fluvastatin administered as LESCOL XL 80 mg tablets reaches peak concentration in approximately 3 hours under fasting conditions, after a low-fat meal, or 2.5 hours after a low-fat meal. The mean relative bioavailability of the XL tablet is approximately 29% (range: 9%-66%) compared to that of the LESCOL immediate-release capsule administered under fasting conditions. Administration of a high-fat meal delayed the absorption (Tmax: 6h) and increased the bioavailability of the XL tablet by approximately 50%. However, the maximum concentration of LESCOL XL seen after a high-fat meal is less than the peak concentration following a single dose or twice daily dose of the 40 mg LESCOL capsule.
Distribution:
Fluvastatin is 98% bound to plasma proteins. The mean volume of distribution (VDss) is estimated at 0.35 L/kg. At therapeutic concentrations, the protein binding of fluvastatin is not affected by warfarin, salicylic acid and glyburide.
Metabolism:
Fluvastatin is metabolized in the liver, primarily via hydroxylation of the indole ring at the 5- and 6-positions. N-dealkylation and beta-oxidation of the side-chain also occurs. The hydroxy metabolites have some pharmacologic activity, but do not circulate in the blood. Fluvastatin has two enantiomers. Both enantiomers of fluvastatin are metabolized in a similar manner.
In vitro data indicate that fluvastatin metabolism involves multiple Cytochrome P450 (CYP) isozymes. CYP2C9 isoenzyme is primarily involved in the metabolism of fluvastatin (approximately 75%), while CYP2C8 and CYP3A4 isoenzymes are involved to a much less extent, i.e. approximately 5% and approximately 20%, respectively.
Excretion:
Following oral administration, fluvastatin is primarily (about 90%) excreted in the feces as metabolites, with less than 2% present as unchanged drug. Approximately 5% of a radiolabeled oral dose were recovered in urine. The elimination half-life (t1/2) of fluvastatin is approximately 3 hours.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
A 2-year study was performed in rats at dose levels of 6, 9, and 18-24 (escalated after 1 year) mg/kg/day. These treatment levels represented plasma drug levels of approximately 9, 13, and 26-35 times the mean human plasma drug concentration after a 40 mg oral dose. A low incidence of forestomach squamous papillomas and 1 carcinoma of the forestomach at the 24 mg/kg/day dose level was considered to reflect the prolonged hyperplasia induced by direct contact exposure to fluvastatin sodium rather than to a systemic effect of the drug. In addition, an increased incidence of thyroid follicular cell adenomas and carcinomas was recorded for males treated with 18-24 mg/kg/day. The increased incidence of thyroid follicular cell neoplasm in male rats with fluvastatin sodium appears to be consistent with findings from other HMG-CoA reductase inhibitors. In contrast to other HMG-CoA reductase inhibitors, no hepatic adenomas or carcinomas were observed.
The carcinogenicity study conducted in mice at dose levels of 0.3, 15 and 30 mg/kg/day revealed, as in rats, a statistically significant increase in forestomach squamous cell papillomas in males and females at 30 mg/kg/day and in females at 15 mg/kg/day. These treatment levels represented plasma drug levels of approximately 0.05, 2, and 7 times the mean human plasma drug concentration after a 40 mg oral dose.
No evidence of mutagenicity was observed in vitro, with or without rat-liver metabolic activation, in the following studies: microbial mutagen tests using mutant strains of Salmonella typhimurium or Escherichia coli; malignant transformation assay in BALB/3T3 cells; unscheduled DNA synthesis in rat primary hepatocytes; chromosomal aberrations in V79 Chinese Hamster cells; HGPRT V79 Chinese Hamster cells. In addition, there was no evidence of mutagenicity in vivo in either a rat or mouse micronucleus test.
In a study in rats at dose levels for females of 0.6, 2 and 6 mg/kg/day and at dose levels for males of 2, 10 and 20 mg/kg/day, fluvastatin sodium had no adverse effects on the fertility or reproductive performance.
Seminal vesicles and testes were small in hamsters treated for 3 months at 20 mg/kg/day (approximately three times the 40 mg human daily dose based on surface area, mg/m2). There was tubular degeneration and aspermatogenesis in testes as well as vesiculitis of seminal vesicles. Vesiculitis of seminal vesicles and edema of the testes were also seen in rats treated for 2 years at 18 mg/kg/day (approximately 4 times the human Cmax achieved with a 40 mg daily dose).
Fluvastatin sodium produced delays in skeletal development in rats at doses of 12 mg/kg/day and in rabbits at doses of 10 mg/kg/day. Malaligned thoracic vertebrae were seen in rats at 36 mg/kg, a dose that produced maternal toxicity. These doses resulted in 2 times (rat at 12 mg/kg) or 5 times (rabbit at 10 mg/kg) the 40 mg human exposure based on mg/m2 surface area. A study in which female rats were dosed during the third trimester at 12 and 24 mg/kg/day resulted in maternal mortality at or near term and postpartum. In addition, fetal and neonatal lethality were apparent. No effects on the dam or fetus occurred at 2 mg/kg/day. A second study at levels of 2, 6, 12 and 24 mg/kg/day confirmed the findings in the first study with neonatal mortality beginning at 6 mg/kg. A modified Segment III study was performed at dose levels of 12 or 24 mg/kg/day with or without the presence of concurrent supplementation with mevalonic acid, a product of HMG-CoA reductase which is essential for cholesterol biosynthesis. The concurrent administration of mevalonic acid completely revented the maternal and neonatal mortality but did not prevent low body weights in pups at 24 mg/kg on days 0 and 7 postpartum.
Clinical Studies
Hypercholesterolemia (Heterozygous Familial and Nonfamilial) and Mixed Dyslipidemia
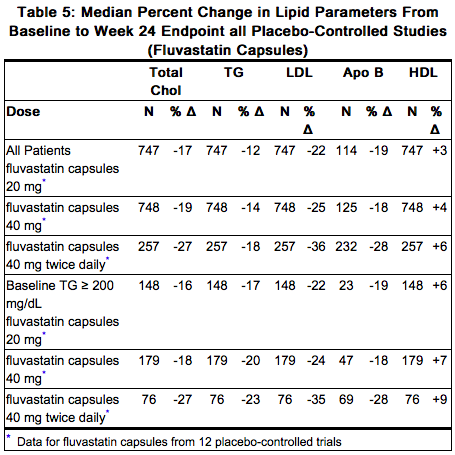
In 12 placebo-controlled studies in patients with primary hypercholesterolemia and mixed dyslipidemia, LESCOL was administered to 1621 patients in daily dose regimens of 20 mg, 40 mg, and 80 mg (40 mg twice daily) for at least 6 weeks duration (Table 5). After 24 weeks of treatment, treatment with LESCOL resulted in significantly reduced plasma LDL-C, TC, TG, and Apo B compared to placebo and was associated with variable increases in HDL-C across the dose range.
LESCOL XL has been studied in five controlled studies of patients with primary hypercholesterolemia and mixed dyslipidemia. LESCOL XL was administered to over 900 patients in trials from 4 to 26 weeks in duration. In the three largest of these studies, LESCOL XL given as a single daily dose of 80 mg significantly reduced Total-C, LDL-C, TG and Apo B and resulted in increases in HDL-C (Table 5).
In patients with primary mixed dyslipidemia as defined by baseline plasma TG levels ≥200 mg/dL and less than 400 mg/dL, treatment with LESCOL/LESCOL XL produced significant decreases in Total-C, LDL-C, TG and Apo B and variable increases in HDL-C (Table 5).
Heterozygous Familial Hypercholesterolemia in Pediatric Patients
LESCOL was studied in two open-label, uncontrolled, dose-titration studies. The first study enrolled 29 pre-pubertal boys, 9-12 years of age, who had an LDL-C level >90th percentile for age and one parent with primary hypercholesterolemia and either a family history of premature ischemic heart disease or tendon xanthomas. The mean baseline LDL-C was 226 mg/dL (range: 137-354 mg/dL). All patients were started on LESCOL capsules 20 mg daily with dose adjustments every 6 weeks to 40 mg daily then 80 mg daily (40 mg b.i.d.) to achieve an LDL-C goal between 96.7 – 123.7 mg/dL. Endpoint analyses were performed at Year 2. LESCOL decreased plasma levels of Total-C and LDL-C by 21% and 27%, respectively. The mean achieved LDL-C was 161 mg/dL (range: 74-336 mg/dL).
The second study enrolled 85 male and female patients, 10 to 16 years of age, who had an LDL-C >190 mg/dL or LDL-C >160 mg/dL and one or more risk factors for coronary heart disease, or LDL-C >160 mg/dL and a proven LDL-receptor defect. The mean baseline LDL-C was 225 mg/dL (range: 148-343 mg/dL). All patients were started on LESCOL capsules 20 mg daily with dose adjustments every 6 weeks to 40 mg daily then 80 mg daily (LESCOL 80 mg XL tablet) to achieve an LDL-C goal of less than 130 mg/dL. Endpoint analyses were performed at Week 114. LESCOL decreased plasma levels of Total-C and LDL-C by 22% and 28%, respectively. The mean achieved LDL-C was 159 mg/dL (range: 90-295 mg/dL).
The majority of patients in both studies (83% in the first study and 89% in the second study) were titrated to the maximum daily dose of 80 mg. At study endpoint, 26% to 30% of patients in both studies achieved a targeted LDL-C goal of less than 130 mg/dL. The long-term efficacy of LESCOL or LESCOL XL therapy in childhood to reduce morbidity and mortality in adulthood has not been established.
14.3 Secondary Prevention of Cardiovascular Disease
In the LESCOL Intervention Prevention Study (LIPS), the effect of LESCOL 40 mg administered twice daily on the risk of recurrent cardiac events (time to first occurrence of cardiac death, nonfatal myocardial infarction, or revascularization) was assessed in 1677 patients with CHD who had undergone a percutaneous coronary intervention (PCI) procedure (mean time from PCI to randomization=3 days). In this multicenter, randomized, double-blind, placebo-controlled study, patients were treated with dietary/lifestyle counseling and either LESCOL 40 mg (n=844) or placebo (n=833) given twice daily for a median of 3.9 years. The study population was 84% male, 98% Caucasian, with 37% greater than 65 years of age. Mean baseline lipid concentrations were: total cholesterol 201 mg/dL, LDL-C 132 mg/dL, triglycerides 70 mg/dL and HDL-C 39 mg/dL.
LESCOL significantly reduced the risk of recurrent cardiac events (Figure 1) by 22% (p=0.013, 181 patients in the LESCOL group vs. 222 patients in the placebo group). Revascularization procedures comprised the majority of the initial recurrent cardiac events (143 revascularization procedures in the LESCOL group and 171 in the placebo group). Consistent trends in risk reduction were observed in patients greater than 65 years of age.
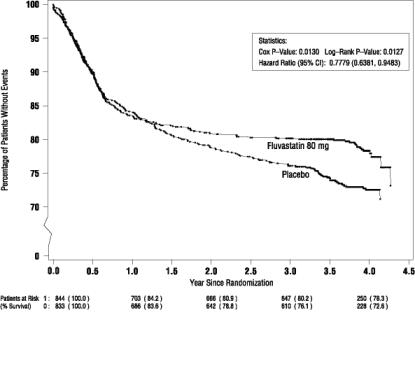
Figure 1 Primary Endpoint – Recurrent Cardiac Events (Cardiac Death, Nonfatal MI or Revascularization Procedure) (ITT Population)
Outcome data for the LESCOL Intervention Prevention Study are shown in Figure 2. After exclusion of revascularization procedures (CABG and repeat PCI) occurring within the first 6 months of the initial procedure involving the originally instrumental site, treatment with LESCOL was associated with a 32% (p=0.002) reduction in risk of late revascularization procedures (CABG or PCI occurring at the original site greater than 6 months after the initial procedure, or at another site).
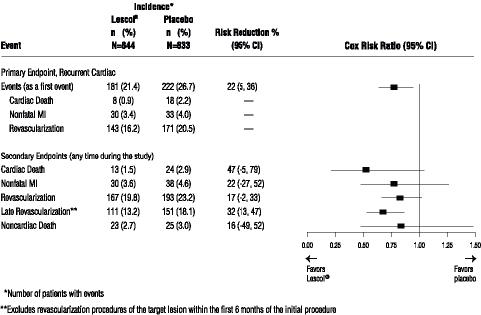
Figure 2 LESCOL® Intervention Prevention Study - Primary and Secondary Endpoints
In the Lipoprotein and Coronary Atherosclerosis Study (LCAS), the effect of LESCOL therapy on coronary atherosclerosis was assessed by quantitative coronary angiography (QCA) in patients with CAD and mild to moderate hypercholesterolemia (baseline LDL-C range 115-190 mg/dL). In this randomized double-blind, placebo- controlled trial, 429 patients were treated with conventional measures (Step 1 AHA Diet) and either LESCOL 40 mg/day or placebo. In order to provide treatment to patients receiving placebo with LDL-C levels ≥160 mg/dL at baseline, adjunctive therapy with cholestyramine was added after Week 12 to all patients in the study with baseline LDL-C values of ≥160 mg/dL which were present in 25% of the study population. Quantitative coronary angiograms were evaluated at baseline and 2.5 years in 340 (79%) angiographic evaluable patients.
Compared to placebo, LESCOL significantly slowed the progression of coronary atherosclerosis as measured by within-patient per-lesion change in minimum lumen diameter (MLD), the primary endpoint (Figure 3 below), percent diameter stenosis (Figure 4), and the formation of new lesions (13% of all fluvastatin patients versus 22% of all placebo patients). A significant difference in favor of LESCOL was found between all fluvastatin and all placebo patients in the distribution among the three categories of definite progression, definite regression, and mixed or no change. Beneficial angiographic results (change in MLD) were independent of patients’ gender and consistent across a range of baseline LDL-C levels.
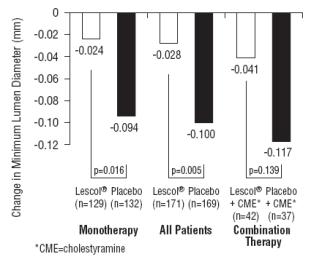

How Supplied
LESCOL® XL (fluvastatin sodium) Extended-Release Tablets
80 mg Yellow, round, slightly biconvex film-coated tablet with beveled edges debossed with “LESCOL XL” on one side and “80” on the other. Bottles of 30 tablets…………………………………………………………………………………...NDC 0078-0354-15 Bottles of 100 tablets…………………………………………………………………………………...NDC 0078-0354-05
Fluvastatin capsules USP
20 mg - hard gelatin capsules with ivory opaque body and pink opaque cap, filled with an off-white to yellowish powder with small agglomerates, body imprinted with “TEVA” and cap imprinted with “7442”, in bottles of 30 and 100. 40 mg - hard gelatin capsules with yellow opaque body and pink opaque cap, filled with an off-white to yellowish powder with small agglomerates, body imprinted with “TEVA” and cap imprinted with “7443”, in bottles of 30 and 100.
Storage
LESCOL® XL (fluvastatin sodium) Extended-Release Tablets
Store at 25ºC (77ºF); excursions permitted to 15 -30ºC (59 -86ºF) [see USP Controlled Room Temperature]. Dispense in a tight container. Protect from light.
Fluvastatin capsules USP
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required). Protect from light.
Images
Drug Images
{{#ask: Page Name::Fluvastatin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Fluvastatin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Information for Patients
Patients taking LESCOL/LESCOL XL should be advised that high cholesterol is a chronic condition and they should adhere to their medication along with their National Cholesterol Education Program (NCEP)-recommended diet, a regular exercise program, and periodic testing of a fasting lipid panel to determine goal attainment.
Patients should be advised about substances they should not take concomitantly with LESCOL/LESCOL XL [see Warnings and Precautions (5.1)]. Patients should also be advised to inform other healthcare professionals prescribing a new medication that they are taking LESCOL/LESCOL XL.
Muscle Pain
Patients starting therapy with LESCOL/LESCOL XL should be advised of the risk of myopathy and told to report promptly any unexplained muscle pain, tenderness or weakness, particularly if accompanied by malaise or fever or if these muscle signs or symptoms persist after discontinuing LESCOL/LESCOL XL.
Liver Enzymes
It is recommended that liver enzyme tests be performed before the initiation of LESCOL/LESCOL XL and if signs or symptoms of liver injury occur. All patients treated with LESCOL/LESCOL XL should be advised to report promptly any symptoms that may indicate liver injury, including fatigue, anorexia, right upper abdominal discomfort, dark urine or jaundice.
Pregnancy
Women of childbearing age should be advised to use an effective method of birth control to prevent pregnancy while using LESCOL/LESCOL XL. Discuss future pregnancy plans with your patients, and discuss when to stop taking LESCOL/LESCOL XL if they are trying to conceive. Patients should be advised that if they become pregnant they should stop taking LESCOL/LESCOL XL and call their healthcare professional.
Breastfeeding
Women who are breastfeeding should not use LESCOL/LESCOL XL. Patients who have a lipid disorder and are breastfeeding should be advised to discuss the options with their healthcare professional.
Precautions with Alcohol
Caution should be exercised when LESCOL is administered to patients with a history of liver disease or heavy alcohol ingestion [see Clinical Pharmacology (12.3)]. Such patients should be closely monitored.
Brand Names
There is limited information regarding Fluvastatin Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Fluvastatin Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ 1.0 1.1 1.2 1.3 1.4 Neuvonen, PJ; Backman, JT; Niemi, M (2008). "Pharmacokinetic comparison of the potential over-the-counter statins simvastatin, lovastatin, fluvastatin and pravastatin". Clinical Pharmacokinetics. 47 (7): 463–74. doi:10.2165/00003088-200847070-00003. PMID 18563955.
- ↑ 2.0 2.1 "Lescol, Lescol XR (fluvastatin) dosing, indications, interactions, adverse efects, and more". Medscape Reference. WebMD. Retrieved 18 March 2014.