Praziquantel
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Deepika Beereddy, MBBS [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Praziquantel is an anthelmintic, antiparasitic agent that is FDA approved for the treatment of clonorchiasis, infection by opisthorchis viverrini, schistosomiasis. Common adverse reactions include cardiac dysrhythmia, rash, urticaria, abdominal pain, defecation urgency, diarrhea, hemorrhagic diarrhea, nausea, vomiting, dizziness, headache, acute CNS inflammatory disease, seizure, somnolence, fever, malaise.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Clonorchiasis
- Dosing Information
- The recommended dose for clonorchiasis or opisthorchiasis is 25 milligrams/kilogram/day 3 times a day as a 1-day treatment. The interval between doses should be not less than 4 and not more than 6 hours. The tablets should be taken with meals and washed down with water.
Infection by Opisthorchis viverrini
- Dosing Information
- The recommended dose for clonorchiasis or opisthorchiasis is 25 milligrams/kilogram 3 times a day as a 1-day treatment. The interval between doses should be not less than 4 and not more than 6 hours. The tablets should be taken with meals and washed down with water.
Schistosomiasis
- Dosing Information
- The recommended adult dose for Schistosoma hematobium, Schistosoma japonicum, Schistosoma mansoni, and Schistosoma mekongi is praziquantel 20 milligrams/kilogram 3 times a day as a 1-day treatment. The interval between the individual doses should not be less than 4 and not more than 6 hours.
- Praziquantel tablets should be swallowed whole (unchewed) and washed down with water during meals. If the tablets or parts of the tablets are kept in the mouth, a bitter taste (which can promote gagging or vomiting) may be experienced.
- Although single dose therapy is effective, the administration of larger doses has been reported in some studies to be associated with a higher incidence of adverse effects than seen with repeated smaller doses.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Praziquantel in adult patients.
Non–Guideline-Supported Use
- Infection by Paragonimus
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Clonorchiasis
- Dosing Information
- Safety of praziquantel in children under 4 years of age has not been established.
- The recommended dose for clonorchiasis or opisthorchiasis is 25 milligrams/kilogram 3 times a day as a 1-day treatment. The interval between the individual doses should not be less than 4 hours and not more than 6 hours.
- Praziquantel tablets should be swallowed whole (unchewed) and washed down with water during meals. If the tablets or parts of the tablets are kept in the mouth, a bitter taste (which can promote gagging or vomiting) may be experienced.
Infection by Opisthorchis viverrini
- Dosing Information
- Safety of praziquantel in children under 4 years of age has not been established.
- The recommended dose for clonorchiasis or opisthorchiasis is 25 milligrams/kilogram 3 times a day as a 1-day treatment. The interval between the individual doses should not be less than 4 hours and not more than 6 hours.
- Praziquantel tablets should be swallowed whole (unchewed) and washed down with water during meals. If the tablets or parts of the tablets are kept in the mouth, a bitter taste (which can promote gagging or vomiting) may be experienced.
Schistosomiasis
- Dosing Information
- Safety of praziquantel in children under 4 years of age has not been established.
- The recommended dose for Schistosoma infections in children 4 years of age or older is praziquantel 20 milligrams/kilogram 3 times a day as a 1-day treatment. The interval between the individual doses should not be less than 4 hours and not more than 6 hours.
- Praziquantel tablets should be swallowed whole (unchewed) and washed down with water during meals. If the tablets or parts of the tablets are kept in the mouth, a bitter taste (which can promote gagging or vomiting) may be experienced.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information regarding Off-Label Guideline-Supported Use of Praziquantel in pediatric patients.
Non–Guideline-Supported Use
- Hymenolepiasis
Contraindications
- BILTRICIDE is contraindicated in patients who previously have shown hypersensitivity to the drug or any of the excipients. Since parasite destruction within the eye may cause irreversible lesions, ocular cysticercosis must not be treated with this compound.
- Concomitant administration with strong Cytochrome P450 (P450) inducers, such as rifampin, is contraindicated since therapeutically effective blood levels of praziquantel may not be achieved (see PRECAUTIONS/Drug Interactions). In patients receiving rifampin who need immediate treatment for schistosomiasis, alternative agents for schistosomiasis should be considered. However, if treatment with praziquantel is necessary, rifampin should be discontinued 4 weeks before administration of praziquantel. Treatment with rifampin can then be restarted one day after completion of praziquantel treatment.
Warnings
- Therapeutically effective levels of BILTRICIDE may not be achieved when administered concomitantly with strong P450 inducers, such as rifampin.
Precautions
General
- Approximately 80% of a dose of praziquantel is excreted in the kidneys, almost exclusively (>99%) in the form of metabolites. Excretion might be delayed in patients with impaired renal function, but accumulation of unchanged drug would not be expected. Therefore, dose adjustment for renal impairment is not considered necessary. Nephrotoxic effects of praziquantel or its metabolites are not known.
- Caution should be exercised in the administration of the usual recommended dose of praziquantel to hepatosplenic schistosomiasis patients with moderate to severe liver impairment (Child-Pugh class B and C). Reduced metabolism of praziquantel by the liver in these patients may lead to considerably higher and longer lasting plasma concentrations of unmetabolized praziquantel.
- Minimal increases in liver enzymes have been reported in some patients.
- Patients suffering from cardiac irregularities should be monitored during treatment.
- As BILTRICIDE can exacerbate central nervous system pathology due to schistosomiasis, as a general rule this drug should not be administered to individuals reporting a history of epilepsy and/or other signs of potential central nervous systems involvement such as subcutaneous nodules suggestive of cysticercosis.
- When schistosomiasis or fluke infection is found to be associated with cerebral cysticercosis it is advised to hospitalize the patient for the duration of treatment.
Adverse Reactions
Clinical Trials Experience
- In general BILTRICIDE is very well tolerated. Side effects are usually mild and transient and do not require treatment. The following side effects were observed generally in order of severity: malaise, headache, dizziness, abdominal discomfort with or without nausea, rise in temperature and, rarely, urticaria. Such symptoms can, however, also result from the infection itself. Such side effects may be more frequent and/or serious in patients with a heavy worm burden.
Postmarketing Experience
- Additional adverse events reported from worldwide post marketing experience and from publications with praziquantel include: abdominal pain, allergic reaction (generalized hypersensitivity) including polyserositis, anorexia, arrhythmia (including bradycardia, ectopic rhythms, ventricular fibrillation, AV blocks), asthenia, bloody diarrhea, convulsion, eosinophilia, myalgia, pruritis, somnolence, vertigo and vomiting.
Drug Interactions
- Concomitant administration of rifampin, a strong P450 inducer, with praziquantel is contraindicated and must be avoided (see CONTRAINDICATIONS). In a crossover study with a 2-week washout period, 10 healthy subjects ingested a single 40 mg/kg dose of praziquantel following pre-treatment with oral rifampin (600 mg daily for 5 days). Plasma praziquantel concentrations were undetectable in 7 out of 10 subjects. When a single 40 mg/kg dose of praziquantel was administered to these healthy subjects two weeks after discontinuation of rifampin, the mean praziquantel AUC and Cmax were 23% and 35% lower, respectively, than when praziquantel was given alone. In patients receiving rifampin, for example, as part of a combination regimen for the treatment of tuberculosis, alternative agents for schistosomiasis should be considered. However, if treatment with praziquantel is necessary, treatment with rifampin should be discontinued 4 weeks before administration of praziquantel. Treatment with rifampin can then be restarted one day after completion of praziquantel treatment.
- Concomitant administration of other drugs that increase the activity of drug metabolizing liver enzymes (P450 inducers), for example, antiepileptic drugs (phenytoin, phenobarbital and carbamazepine), and dexamethasone, may also reduce plasma levels of praziquantel. Concomitant administration of drugs that decrease the activity of drug metabolizing liver enzymes (P 450 inhibitors), for example, cimetidine, ketoconazole, itraconazole, erythromycin may increase plasma levels of praziquantel.
- Chloroquine, when taken simultaneously, may lead to lower concentrations of praziquantel in blood. The mechanism of this drug-drug interaction is unclear.
- Grapefruit juice was reported to produce a 1.6-fold increase in the Cmax and a 1.9-fold increase in the AUC of praziquantel. However, the effect of this exposure increase on the therapeutic effect and safety of praziquantel has not been systematically evaluated.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): B Pregnancy Category B
- Reproduction studies have been performed in rats and rabbits at doses up to 40 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to praziquantel. There are, however, no adequate and well-controlled studies in pregnant women. An increase of the abortion rate was found in rats at three times the single human therapeutic dose. While animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
- There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Praziquantel in women who are pregnant.
Labor and Delivery
- There is no FDA guidance on use of Praziquantel during labor and delivery.
Nursing Mothers
- Praziquantel appeared in the milk of nursing women at a concentration of about 1/4 that of maternal serum although it is not known, whether a pharmacological effect is likely to occur in children. Women should not nurse on the day of BILTRICIDE treatment and during the subsequent 72 hours.
Pediatric Use
- Safety in children under 4 years of age has not been established.
Geriatic Use
- Clinical studies of praziquantel did not include a sufficient number of subjects ages 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older patients cannot be ruled out.
- This drug is known to be substantially excreted by the kidney. Because elderly patients are more likely to have decreased renal function, the risk of toxic reactions to this drug may be greater in these patients.
Gender
- There is no FDA guidance on the use of Praziquantel with respect to specific gender populations.
Race
- There is no FDA guidance on the use of Praziquantel with respect to specific racial populations.
Renal Impairment
- There is no FDA guidance on the use of Praziquantel in patients with renal impairment.
Hepatic Impairment
- There is no FDA guidance on the use of Praziquantel in patients with hepatic impairment.
Females of Reproductive Potential and Males
- There is no FDA guidance on the use of Praziquantel in women of reproductive potentials and males.
Immunocompromised Patients
- There is no FDA guidance one the use of Praziquantel in patients who are immunocompromised.
Administration and Monitoring
Administration
- The dosage recommended for the treatment of schistosomiasis is: 20 mg/kg bodyweight three times a day as a one day treatment, at intervals of not less than 4 hours and not more than 6 hours. The recommended dose for clonorchiasis and opisthorchiasis is: 25 mg/kg bodyweight three times a day as a one day treatment, at intervals of not less than 4 hours and not more than 6 hours. The tablets should be washed down unchewed with water during meals. Keeping the tablets or segments thereof in the mouth can reveal a bitter taste which can promote gagging or vomiting.
Monitoring
- There is limited information regarding Monitoring of Praziquantel in the drug label.
IV Compatibility
- There is limited information regarding IV Compatibility of Praziquantel in the drug label.
Overdosage
- In rats and mice the acute LD50 was about 2,500 mg/kg. No data are available in humans. In the event of overdose a fast-acting laxative should be given.
Pharmacology
Mechanism of Action
- Praziquantel induces a rapid contraction of schistosomes by a specific effect on the permeability of the cell membrane. The drug further causes vacuolization and disintegration of the schistosome tegument.
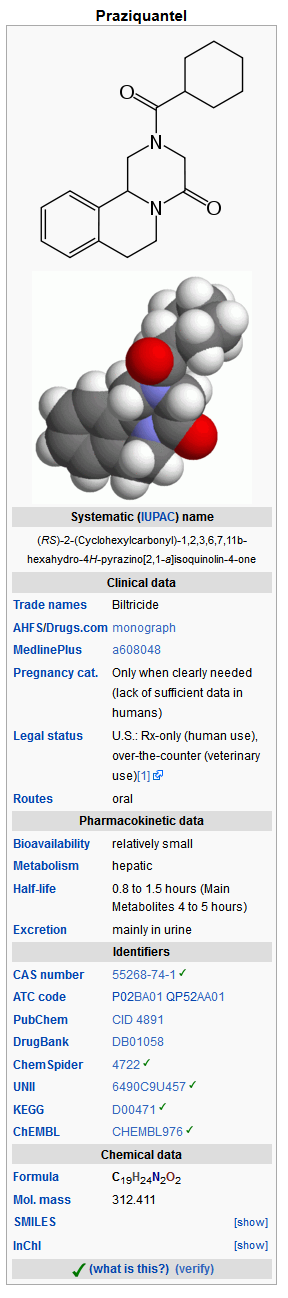
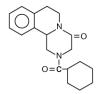
Structure
- BILTRICIDE® (praziquantel) is a trematodicide provided in tablet form for the oral treatment of schistosome infections and infections due to liver fluke.
- BILTRICIDE (praziquantel) is 2-(cyclohexylcarbonyl)-1,2,3,6,7, 11b-hexahydro-4H-pyrazino [2, 1-a] isoquinolin-4-one with the molecular formula; C19H24N2O2. The structural formula is as follows:
- Praziquantel is a white to nearly white crystalline powder of bitter taste. The compound is stable under normal conditions and melts at 136-140°C with decomposition. The active substance is hygroscopic. Praziquantel is easily soluble in chloroform and dimethylsulfoxide, soluble in ethanol and very slightly soluble in water.
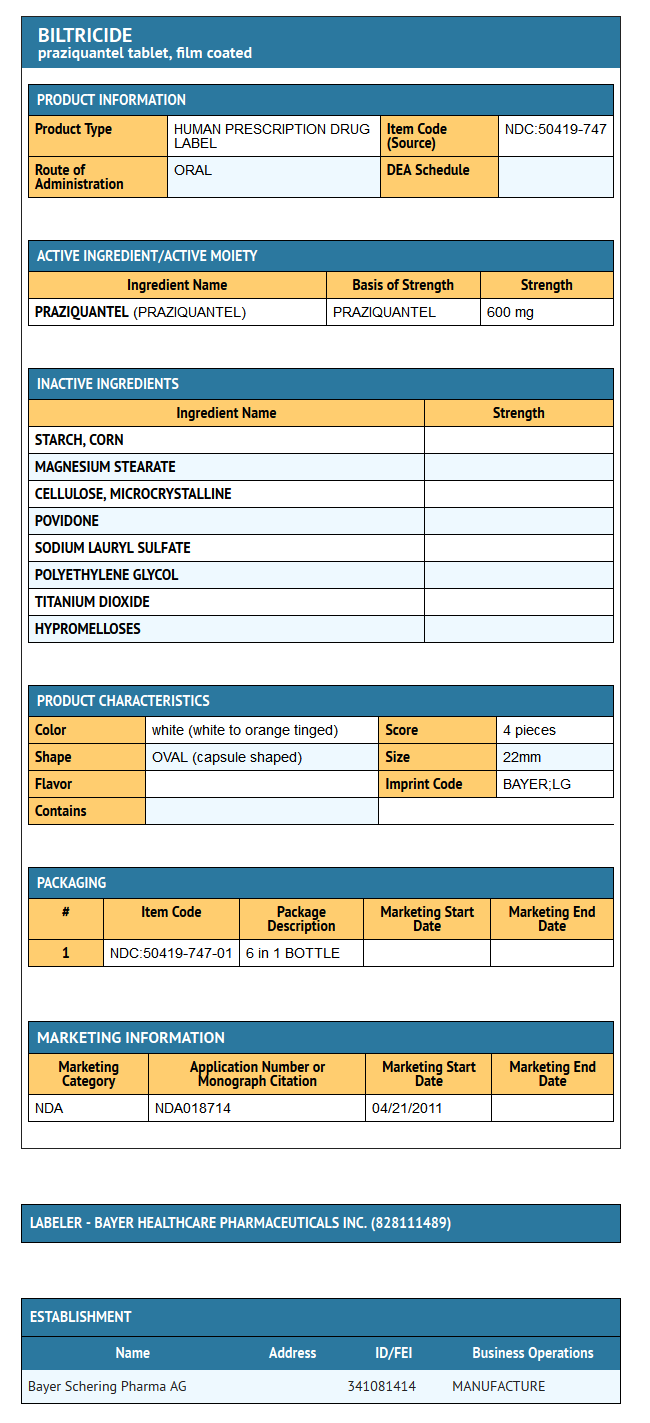
- BILTRICIDE tablets contain 600 mg of praziquantel. Inactive ingredients: corn starch, magnesium stearate, microcrystalline cellulose, povidone, sodium lauryl sulfate, polyethylene glycol, titanium dioxide and hypromellose.
Pharmacodynamics
- There is limited information regarding Pharmacodynamics of Praziquantel in the drug label.
Pharmacokinetics
- After oral administration BILTRICIDE is rapidly absorbed (80%), subjected to a first pass effect, metabolized and eliminated by the kidneys. Maximal serum concentration is achieved 1-3 hours after dosing. The half-life of praziquantel in serum is 0.8-1.5 hours.
Special Populations
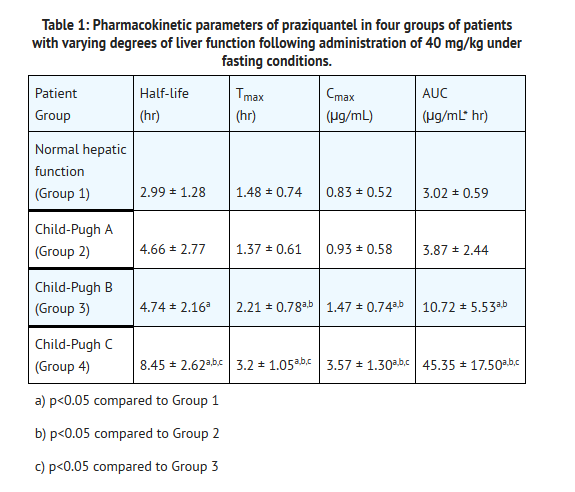
- The pharmacokinetics of praziquantel were studied in 40 patients with Schistosoma mansoni infections with varying degrees of hepatic dysfunction (See table1). In patients with schistosomiasis, the pharmacokinetic parameters did not differ significantly between those with normal hepatic function (Group 1) and those with mild (Child-Pugh class A) hepatic impairment. However, in patients with moderate-to-severe hepatic dysfunction (Child-Pugh class B and C), praziquantel half-life, Cmax, and AUC increased progressively with the degree of hepatic impairment. In Child-Pugh class B, the increases in mean half-life, Cmax, and AUC relative to Group 1 were 1.58-fold, 1.76-fold, and 3.55-fold, respectively. The corresponding increases in Child-Pugh class C patients were 2.82-fold, 4.29-fold, and 15-fold for half-life, Cmax, and AUC.
Nonclinical Toxicology
Mutagenesis, Carcinogenesis
- Mutagenic effects in Salmonella tests found by one laboratory have not been confirmed in the same tested strain by other laboratories. Long term carcinogenicity studies in rats and golden hamsters did not reveal any carcinogenic effect.
Clinical Studies
- There is limited information regarding Clinical Studies of Praziquantel in the drug label.
How Supplied
- BILTRICIDE is supplied as a 600 mg white to orange tinged, film-coated, oblong tablet with three scores. The tablet is coded with “BAYER” on one side and “LG” on the reverse side. When broken, each of the four segments contains 150 mg of active ingredient so that the dosage can be easily adjusted to the patient’s bodyweight.
- Segments are broken off by pressing the score (notch) with thumbnails. If 1/4 of a tablet is required, this is best achieved by breaking the segment from the outer end.
- BILTRICIDE is available in bottles of 6 tablets.

Storage
There is limited information regarding Praziquantel Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Praziquantel |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel

{{#ask: Label Page::Praziquantel |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Information for Patients
- Patients should be warned not to drive a car and not to operate machinery on the day of BILTRICIDE treatment and the following day.
Precautions with Alcohol
- Alcohol-Praziquantel interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Biltricide
Look-Alike Drug Names
- A® — B®[1]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Praziquantel
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Praziquantel |Label Name=Praziquantel11.png
}}
{{#subobject:
|Label Page=Praziquantel |Label Name=Praziquantel11.png
}}