Darunavir
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Darunavir is a protease inhibitor that is FDA approved for the treatment of human immunodeficiency virus (HIV-1) infection. Common adverse reactions include diarrhea, nausea, rash, headache, abdominal pain and vomiting.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Human Immunodeficiency Virus (HIV-1)
- Dosing Information
- Darunavir must be co-administered with ritonavir to exert its therapeutic effect. Failure to correctly co-administer Darunavir with ritonavir will result in plasma levels of darunavir that will be insufficient to achieve the desired antiviral effect and will alter some drug interactions.
- Treatment-Naïve Adult Patients
- The recommended oral dose of Darunavir is 800 mg (one 800 mg tablet or 8 mL of the oral suspension) taken with ritonavir 100 mg (one 100 mg tablet/capsule or 1.25 mL of a 80 mg/mL ritonavir oral solution) once daily and with food. An 8 mL darunavir dose should be taken as two 4 mL administrations with the included oral dosing syringe.
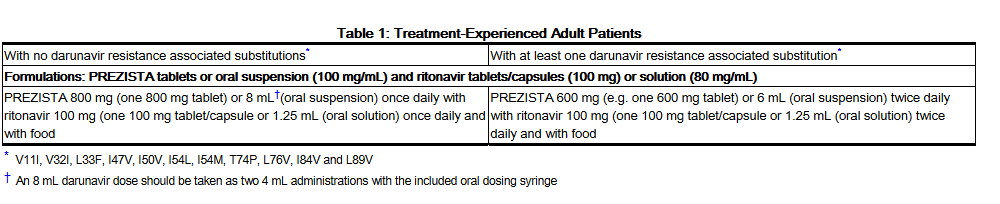
- Treatment-Experienced Adult Patients
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Darunavir in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Darunavir in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Human Immunodeficiency Virus (HIV-1)
- Dosing Information
- The indication for treatment-experienced pediatric patients 3 to less than 18 years of age is based on analyses of plasma HIV-1 RNA levels and CD4+ cell counts from two open-label Phase 2 trials in antiretroviral treatment-experienced pediatric subjects(24-week analysis for one trial in patients 6 to less than 18 years of age; 48-week analysis for one trial in patients 3 to less than 6 years of age) . The indication for treatment-naïve pediatric patients or antiretroviral treatment-experienced patients with no darunavir resistance associated substitutions is based on one open-label Phase 2 trial of 48 weeks duration in antiretroviral treatment-naïve subjects 12 to less than 18 years of age and pharmacokinetic modeling and simulation for patients 3 to less than 12 years of age.
- In treatment-experienced adult and pediatric patients, the following points should be considered when initiating therapy with Darunavir/ritonavir:
- Treatment history and, when available, genotypic or phenotypic testing should guide the use of Darunavir/ritonavir.
- The use of other active agents with Darunavir/ritonavir is associated with a greater likelihood of treatment response.
- Before prescribing Darunavir, children weighing greater than or equal to 15 kg should be assessed for the ability to swallow tablets. If a child is unable to reliably swallow a tablet, the use of Darunavir oral suspension should be considered.
- The recommended dose of Darunavir/ritonavir for pediatric patients (3 to less than 18 years of age and weighing at least 10 kg is based on body weight (see Tables 2, 3, 4, and 5) and should not exceed the recommended adult dose. Darunavir should be taken with ritonavir and with food.
- The recommendations for the Darunavir/ritonavir dosage regimens were based on the following:
- Twice daily dosing
- Results from two trials in treatment-experienced pediatric subjects 3 to less 18 years of age demonstrating similar darunavir plasma exposures, virologic response rate and safety profile compared to treatment-experienced adults.
- Twice daily dosing
- Once daily dosing
- Results from one trial in treatment-naive pediatric subjects 12 to less than 18 years of age demonstrating similar darunavir plasma exposures, virologic response rate and safety profile compared to treatment-naive adults.
- Results from population pharmacokinetic modeling and simulation in children 3 to less than 12 years of age predicting similar darunavir plasma exposures compared to treatment-naïve adults. Although no clinical trial was conducted to collect exposure-safety data, the predicted exposures from the once daily dosing is supported by exposures observed in a pediatric clinical trial where twice-daily dosing was administered.
- Dosing recommendations for treatment-naïve pediatric patients or antiretroviral treatment-experienced pediatric patients with no darunavir resistance associated substitutions
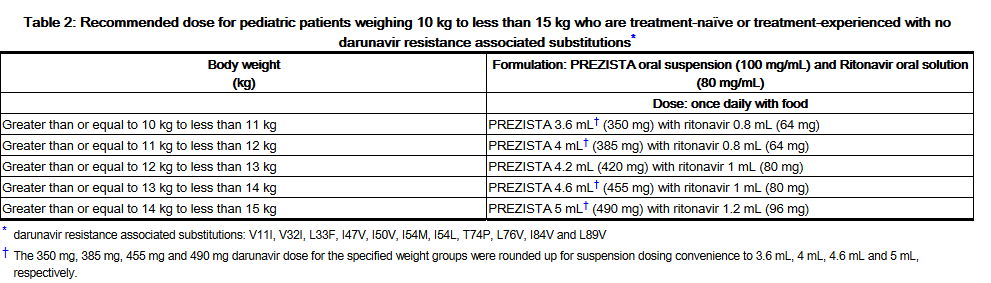
- Pediatric patients weighing at least 10 kg but less than 15 kg
- The weight-based dose in antiretroviral treatment-naïve pediatric patients or antiretroviral treatment-experienced pediatric patients with no darunavir resistance associated substitutions is Darunavir 35 mg/kg once daily with ritonavir 7 mg/kg once daily using the following table.
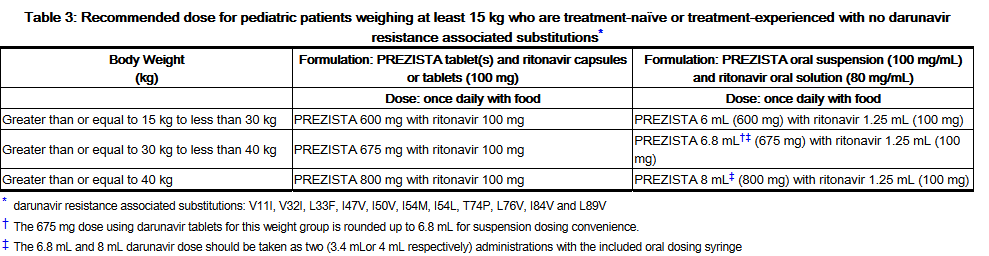
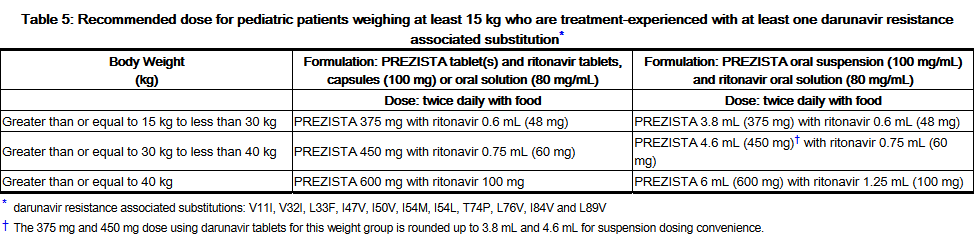
- Pediatric patients weighing at least 15 kg
- Pediatric patients weighing at least 15 kg can be dosed with Darunavir oral tablet(s) or suspension using the following table:
- Dosing recommendations for treatment-experienced pediatric patients with at least one darunavir resistance associated substitutions
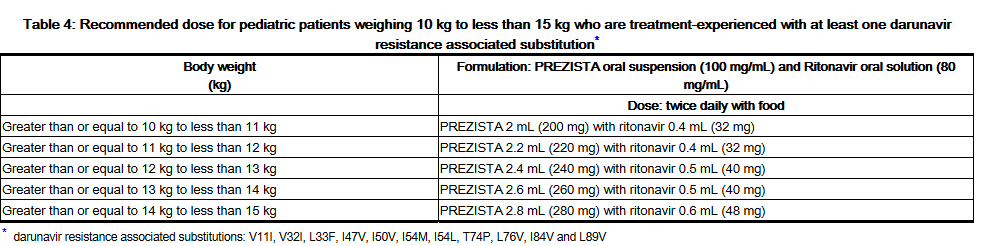
- Pediatric patients weighing at least 10 kg but less than 15 kg
- The weight-based dose in antiretroviral treatment-experienced pediatric patients with at least one darunavir resistance associated substitution is Darunavir 20 mg/kg twice daily with ritonavir 3 mg/kg twice daily using the following table:
- Pediatric patients weighing at least 15 kg
- Pediatric patients weighing at least 15 kg can be dosed with Darunavir oral tablet(s) or suspension using the following table:
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Darunavir in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Darunavir in pediatric patients.
Contraindications
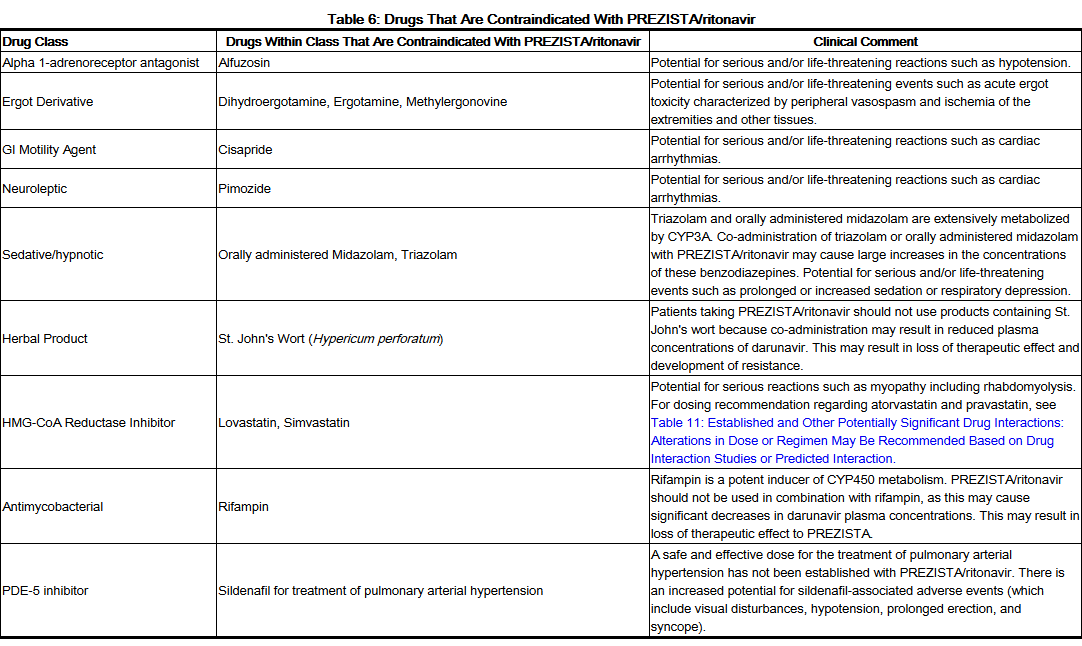
- Co-administration of Darunavir/ritonavir is contraindicated with drugs that are highly dependent on CYP3A for clearance and for which elevated plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). These drugs and other contraindicated drugs (which may lead to reduced efficacy of darunavir) are listed in Table 6
Warnings
Precautions
- General
- Darunavir must be co-administered with ritonavir and food to achieve the desired antiviral effect. Failure to administer Darunavir with ritonavir and food may result in a loss of efficacy of darunavir.
- Please refer to ritonavir prescribing information for additional information on precautionary measures.
- Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) has been reported with Darunavir/ritonavir. During the clinical development program (N=3063), hepatitis was reported in 0.5% of patients receiving combination therapy with Darunavir/ritonavir. Patients with pre-existing liver dysfunction, including chronic active hepatitis B or C, have an increased risk for liver function abnormalities including severe hepatic adverse events.
- Post-marketing cases of liver injury, including some fatalities, have been reported. These have generally occurred in patients with advanced HIV-1 disease taking multiple concomitant medications, having co-morbidities including hepatitis B or C co-infection, and/or developing immune reconstitution syndrome. A causal relationship with Darunavir/ritonavir therapy has not been established.
- Appropriate laboratory testing should be conducted prior to initiating therapy with Darunavir/ritonavir and patients should be monitored during treatment. Increased AST/ALT monitoring should be considered in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases, especially during the first several months of Darunavir/ritonavir treatment.
- Evidence of new or worsening liver dysfunction (including clinically significant elevation of liver enzymes and/or symptoms such as fatigue, anorexia, nausea, jaundice, dark urine, liver tenderness, hepatomegaly) in patients on Darunavir/ritonavir should prompt consideration of interruption or discontinuation of treatment.
- Severe Skin Reactions
- During the clinical development program (n=3063), severe skin reactions, accompanied by fever and/or elevations of transaminases in some cases, have been reported in 0.4% of subjects. Stevens-Johnson Syndrome was rarely (less than 0.1%) reported during the clinical development program. During post-marketing experience toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis have been reported. Discontinue Darunavir/ritonavir immediately if signs or symptoms of severe skin reactions develop. These can include but are not limited to severe rash or rash accompanied with fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, hepatitis and/or eosinophilia.
- Rash (all grades, regardless of causality) occurred in 10.3% of subjects treated with Darunavir/ritonavir. Rash was mostly mild-to-moderate, often occurring within the first four weeks of treatment and resolving with continued dosing. The discontinuation rate due to rash in subjects using Darunavir/ritonavir was 0.5%.
- Rash occurred more commonly in treatment-experienced subjects receiving regimens containing Darunavir/ritonavir + raltegravir compared to subjects receiving Darunavir/ritonavir without raltegravir or raltegravir without Darunavir/ritonavir. However, rash that was considered drug related occurred at similar rates for all three groups. These rashes were mild to moderate in severity and did not limit therapy; there were no discontinuations due to rash.
- Sulfa Allergy
- Darunavir contains a sulfonamide moiety. Darunavir should be used with caution in patients with a known sulfonamide allergy. In clinical studies with Darunavir/ritonavir, the incidence and severity of rash were similar in subjects with or without a history of sulfonamide allergy.
- Drug Interactions
- See Table 6 for a listing of drugs that are contraindicated for use with Darunavir/ritonavir due to potentially life-threatening adverse events, significant drug-drug interactions, or loss of therapeutic effect to Darunavir. Please refer to Table 11 for established and other potentially significant drug-drug interactions.
- Diabetes Mellitus / Hyperglycemia
- New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus, and hyperglycemia have been reported during postmarketing surveillance in HIV-infected patients receiving protease inhibitor (PI) therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases, diabetic ketoacidosis has occurred. In those patients who discontinued PI therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and causal relationships between PI therapy and these events have not been established.
- Fat Redistribution
- Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
- Immune Reconstitution Syndrome
- Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including Darunavir. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
- Autoimmune disorders (such as Graves' disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of antiretroviral treatment.
- There have been reports of increased bleeding, including spontaneous skin hematomas and hemarthrosis in patients with hemophilia type A and B treated with PIs. In some patients, additional factor VIII was given. In more than half of the reported cases, treatment with PIs was continued or reintroduced if treatment had been discontinued. A causal relationship between PI therapy and these episodes has not been established.
- Resistance/Cross-Resistance
- Because the potential for HIV cross-resistance among PIs has not been fully explored in Darunavir/ritonavir treated patients, the effect therapy with Darunavir will have on the activity of subsequently administered PIs is unknown.
- Pediatric Patients
- Do not administer Darunavir/ritonavir in pediatric patients below 3 years of age in view of toxicity and mortality observed in juvenile rats dosed with darunavir (from 20 mg/kg to 1000 mg/kg) up to days 23 to 26 of age.
Adverse Reactions
Clinical Trials Experience
Treatment-Naïve Adults
- Study TMC114-C211
- The safety assessment is based on all safety data from the Phase 3 trial TMC114-C211 comparing Darunavir/ritonavir 800/100 mg once daily versus lopinavir/ritonavir 800/200 mg per day in 689 antiretroviral treatment-naïve HIV-1-infected adult subjects. The total mean exposure for subjects in the Darunavir/ritonavir 800/100 mg once daily arm and in the lopinavir/ritonavir 800/200 mg per day arm was 162.5 and 153.5 weeks, respectively.
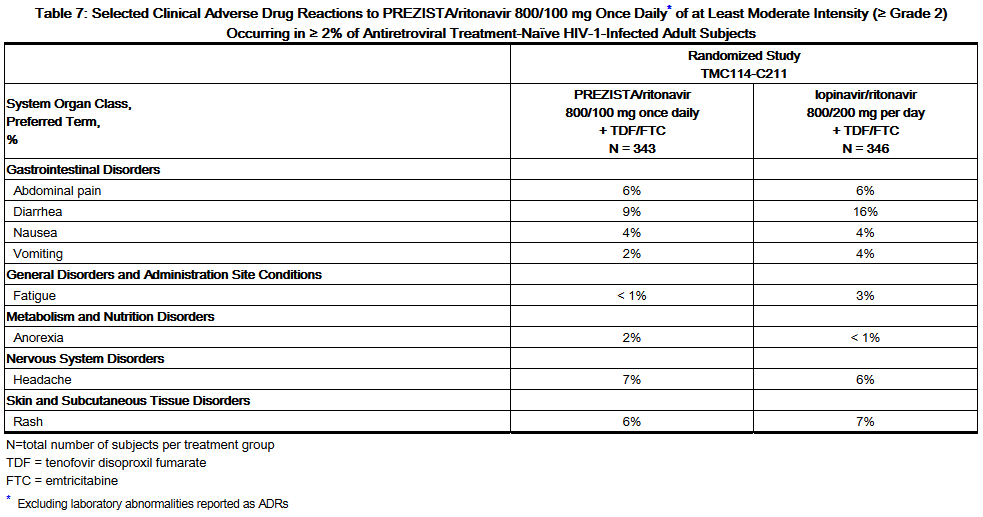
- The majority of the adverse drug reactions (ADRs) reported during treatment with Darunavir/ritonavir 800/100 mg once daily were mild in severity. The most common clinical ADRs to Darunavir/ritonavir 800/100 mg once daily (greater than or equal to 5%) of at least moderate intensity (greater than or equal to Grade 2) were diarrhea, headache, abdominal pain and rash. 2.3% of subjects in the Darunavir/ritonavir arm discontinued treatment due to ADRs.
- ADRs to Darunavir/ritonavir 800/100 mg once daily of at least moderate intensity (greater than or equal to Grade 2) in antiretroviral treatment-naïve HIV-1-infected adult subjects are presented in Table 7 and subsequent text below the table.
- Less Common Adverse Reactions
- Treatment-emergent ADRs of at least moderate intensity (greater than or equal to Grade 2) occurring in less than 2% of antiretroviral treatment-naïve subjects receiving Darunavir/ritonavir 800/100 mg once daily are listed below by body system:
Gastrointestinal Disorders
Acute pancreatitis, dyspepsia, flatulence
General Disorders and Administration Site Conditions
Hepatobiliary Disorders
Acute hepatitis (e.g., acute hepatitis, cytolytic hepatitis, hepatotoxicity)
Immune System Disorders
Hypersensitivity, immune reconstitution syndrome
Metabolism and Nutrition Disorders
Musculoskeletal and Connective Tissue Disorders
Psychiatric Disorders
Skin and Subcutaneous Tissue Disorders
Angioedema, pruritus, Stevens-Johnson Syndrome, urticaria
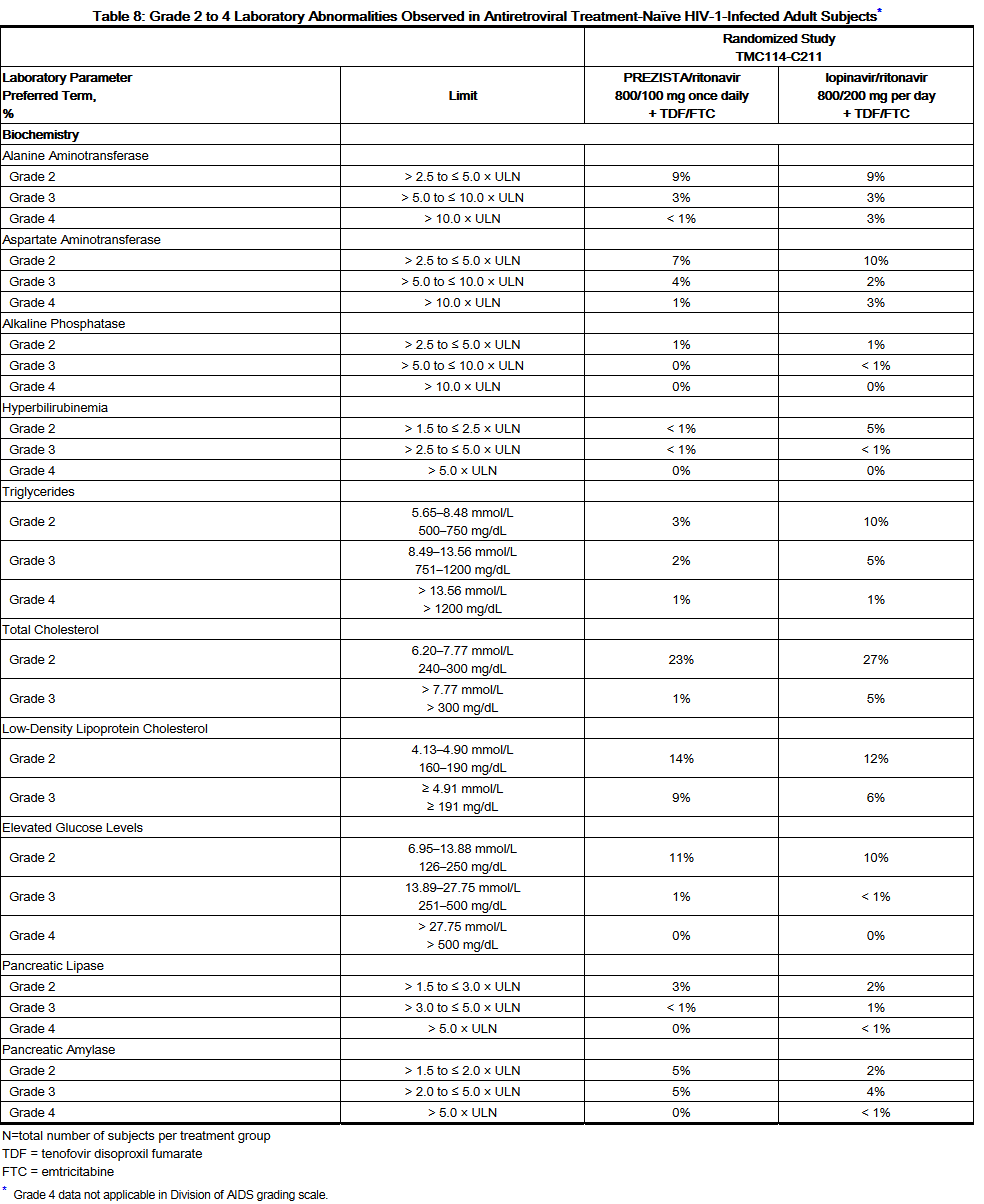
- Laboratory abnormalities:
- Selected Grade 2 to 4 laboratory abnormalities that represent a worsening from baseline observed in antiretroviral treatment-naïve adult subjects treated with Darunavir/ritonavir 800/100 mg once daily are presented in Table 8.
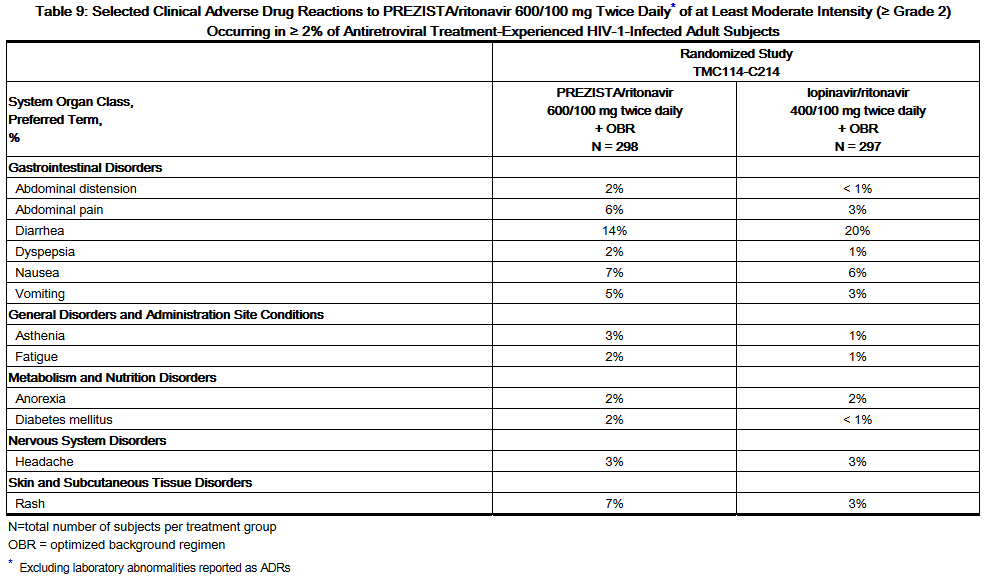
Treatment-Experienced Adults
- Study TMC114-C214
- The safety assessment is based on all safety data from the Phase 3 trial TMC114-C214 comparing Darunavir/ritonavir 600/100 mg twice daily versus lopinavir/ritonavir 400/100 mg twice daily in 595 antiretroviral treatment-experienced HIV-1-infected adult subjects. The total mean exposure for subjects in the Darunavir/ritonavir 600/100 mg twice daily arm and in the lopinavir/ritonavir 400/100 mg twice daily arm was 80.7 and 76.4 weeks, respectively.
- The majority of the ADRs reported during treatment with Darunavir/ritonavir 600/100 mg twice daily were mild in severity. The most common clinical ADRs to Darunavir/ritonavir 600/100 mg twice daily (greater than or equal to 5%) of at least moderate intensity (greater than or equal to Grade 2) were diarrhea, nausea, rash, abdominal pain and vomiting. 4.7% of subjects in the Darunavir/ritonavir arm discontinued treatment due to ADRs.
- ADRs to Darunavir/ritonavir 600/100 mg twice daily of at least moderate intensity (greater than or equal to Grade 2) in antiretroviral treatment-experienced HIV-1-infected adult subjects are presented in Table 9 and subsequent text below the table.
- Less Common Adverse Reactions
- Treatment-emergent ADRs of at least moderate intensity (greater than or equal to Grade 2) occurring in less than 2% of antiretroviral treatment-experienced subjects receiving Darunavir/ritonavir 600/100 mg twice daily are listed below by body system:
Gastrointestinal Disorders
Acute pancreatitis, flatulence
Musculoskeletal and Connective Tissue Disorders
Psychiatric Disorders
Skin and Subcutaneous Tissue Disorders
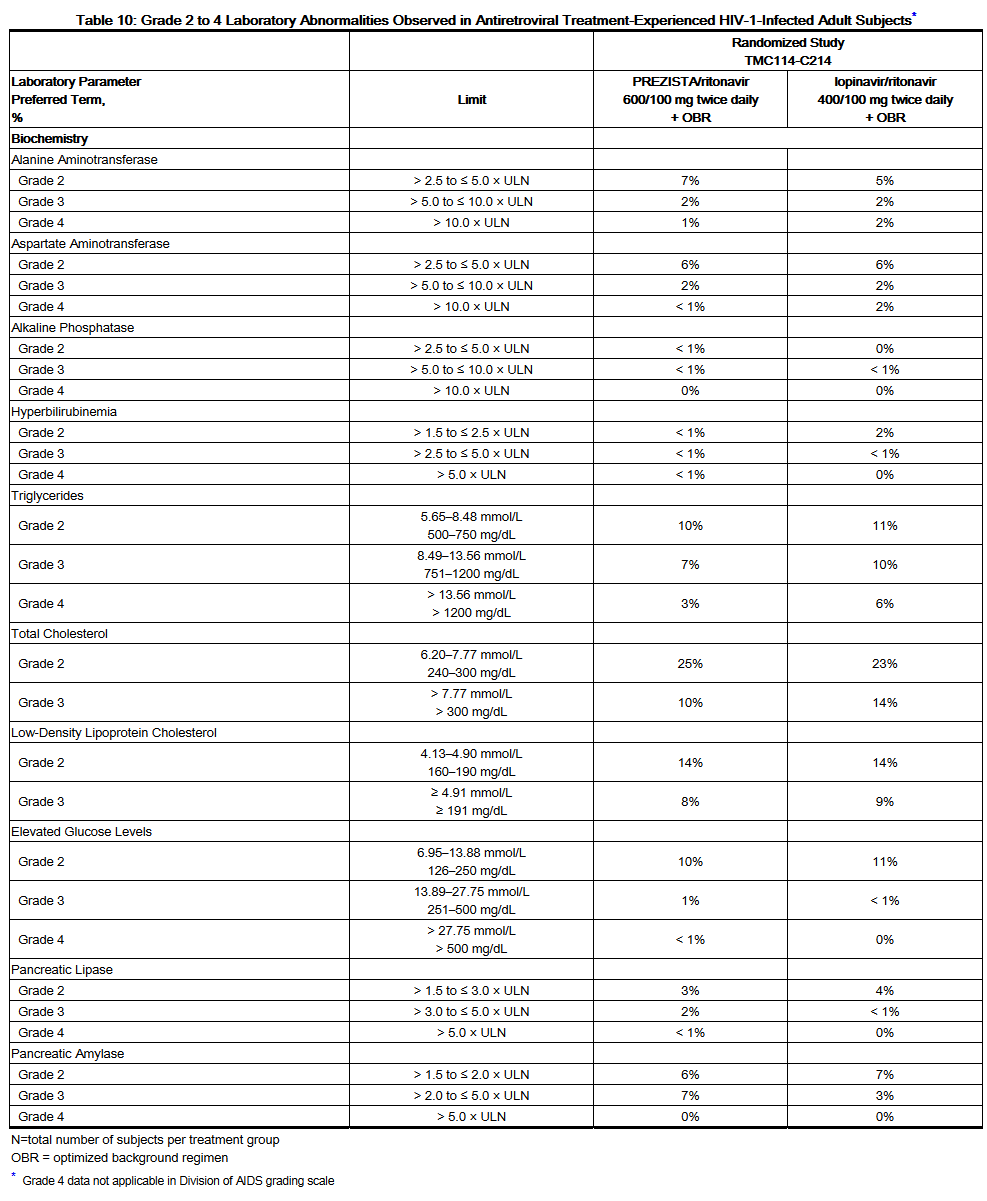
- Laboratory abnormalities:
- Selected Grade 2 to 4 laboratory abnormalities that represent a worsening from baseline observed in antiretroviral treatment-experienced adult subjects treated with Darunavir/ritonavir 600/100 mg twice daily are presented in Table 10.
Serious ADRs
- The following serious ADRs of at least moderate intensity (greater than or equal to Grade 2) occurred in the Phase 2b studies and Phase 3 studies with Darunavir/ritonavir: abdominal pain, acute hepatitis, acute pancreatitis, anorexia, asthenia, diabetes mellitus, diarrhea, fatigue, headache, hepatic enzyme increased, hypercholesterolemia, hyperglycemia, hypertriglyceridemia, immune reconstitution syndrome, low density lipoprotein increased, nausea, pancreatic enzyme increased, rash, Stevens-Johnson Syndrome, and vomiting.
Patients co-infected with hepatitis B and/or hepatitis C virus
- In subjects co-infected with hepatitis B or hepatitis C virus receiving Darunavir/ritonavir, the incidence of adverse events and clinical chemistry abnormalities was not higher than in subjects receiving Darunavir/ritonavir who were not co-infected, except for increased hepatic enzymes. The pharmacokinetic exposure in co-infected subjects was comparable to that in subjects without co-infection.
Clinical Trials Experience: Pediatric Patients
- Darunavir/ritonavir has been studied in combination with other antiretroviral agents in 3 Phase II trials. TMC114-C212, in which 80 antiretroviral treatment-experienced HIV-1-infected pediatric subjects 6 to less than 18 years of age and weighing at least 20 kg were included, TMC114-C228, in which 21 antiretroviral treatment-experienced HIV-1-infected pediatric subjects 3 to less than 6 years of age and weighing at least 10 kg were included, and TMC114-C230 in which 12 antiretroviral treatment-naïve HIV-1 infected pediatric patients aged from 12 to less than 18 years and weighing at least 40 kg were included. The TMC114-C212 and C228 trials evaluated Darunavir/ritonavir twice daily dosing and the TMC114-C230 trial evaluated Darunavir/ritonavir once daily dosing.
- Frequency, type, and severity of ADRs in pediatric subjects were comparable to those observed in adults.
- Study TMC114-C212
- Clinical ADRs to Darunavir/ritonavir (all grades, greater than or equal to 3%), were vomiting (13%), diarrhea (11%), abdominal pain (10%), headache (9%), rash (5%), nausea (4%) and fatigue (3%).
- Grade 3 or 4 laboratory abnormalities were ALT increased (Grade 3: 3%; Grade 4: 1%), AST increased (Grade 3: 1%), pancreatic amylase increased (Grade 3: 4%, Grade 4: 1%), pancreatic lipase increased (Grade 3: 1%), total cholesterol increased (Grade 3: 1%), and LDL increased (Grade 3: 3%).
- Study TMC114-C228
- Clinical ADRs to Darunavir/ritonavir (all grades, greater than or equal to 5%), were diarrhea (24%), vomiting (19%),rash (19%), abdominal pain (5%) and anorexia (5%).
- There were no Grade 3 or 4 laboratory abnormalities considered as ADRs in this study.
- Study TMC114-C230
- Clinical ADRs to Darunavir/ritonavir (all grades, greater than or equal to 3%), were vomiting (33%), nausea (25%), diarrhea (16.7%), abdominal pain (8.3%), decreased appetite (8.3%), pruritus (8.3%), and rash (8.3%).
- There were no Grade 3 or 4 laboratory abnormalities considered as ADRs in this study.
Postmarketing Experience
- The following events have been identified during post approval use of Darunavir. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Redistribution of body fat has been reported.
- Rarely, rhabdomyolysis (associated with co-administration with HMG-CoA reductase inhibitors and Darunavir/ritonavir) has been reported.
- In addition, toxic epidermal necrolysis,acute generalized exanthematous pustulosis and drug rash with eosinophilia and systemic symptoms have been reported rarely.
Drug Interactions
Potential for Darunavir/ritonavir to Affect Other Drugs
- Darunavir co-administered with ritonavir is an inhibitor of CYP3A and CYP2D6. Co-administration of Darunavir and ritonavir with drugs that are primarily metabolized by CYP3A and CYP2D6 may result in increased plasma concentrations of such drugs, which could increase or prolong their therapeutic effect and adverse events (see Table 11).
Potential for Other Drugs to Affect Darunavir
- Darunavir and ritonavir are metabolized by CYP3A. Drugs that induce CYP3A activity would be expected to increase the clearance of darunavir and ritonavir, resulting in lowered plasma concentrations of darunavir and ritonavir. Co-administration of darunavir and ritonavir and other drugs that inhibit CYP3A may decrease the clearance of darunavir and ritonavir and may result in increased plasma concentrations of darunavir and ritonavir (see Table 11).
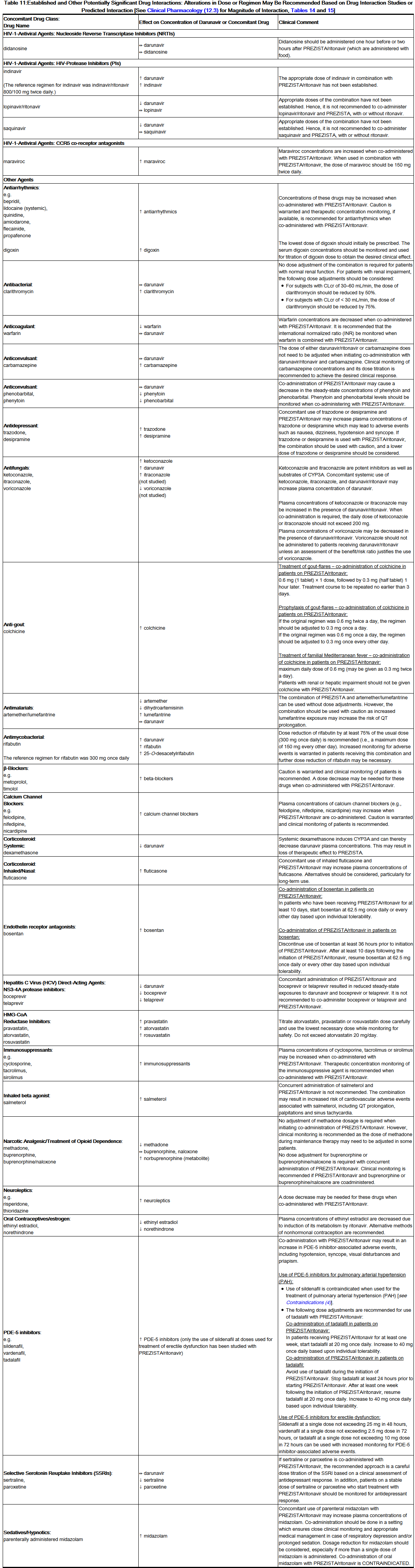
Established and Other Potentially Significant Drug Interactions
- Table 11 provides dosing recommendations as a result of drug interactions with Darunavir/ritonavir. These recommendations are based on either drug interaction studies or predicted interactions due to the expected magnitude of interaction and potential for serious adverse events or loss of efficacy.
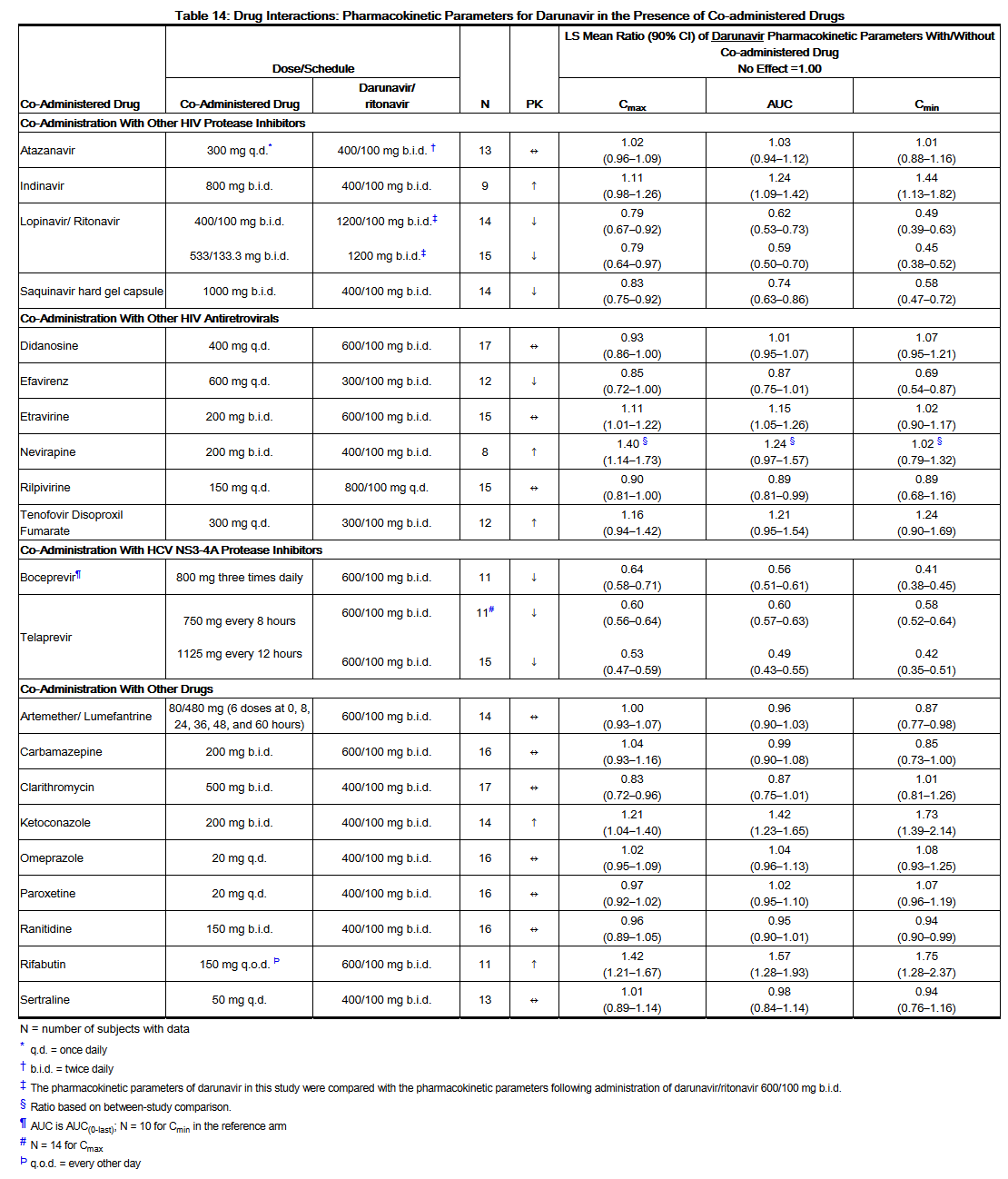
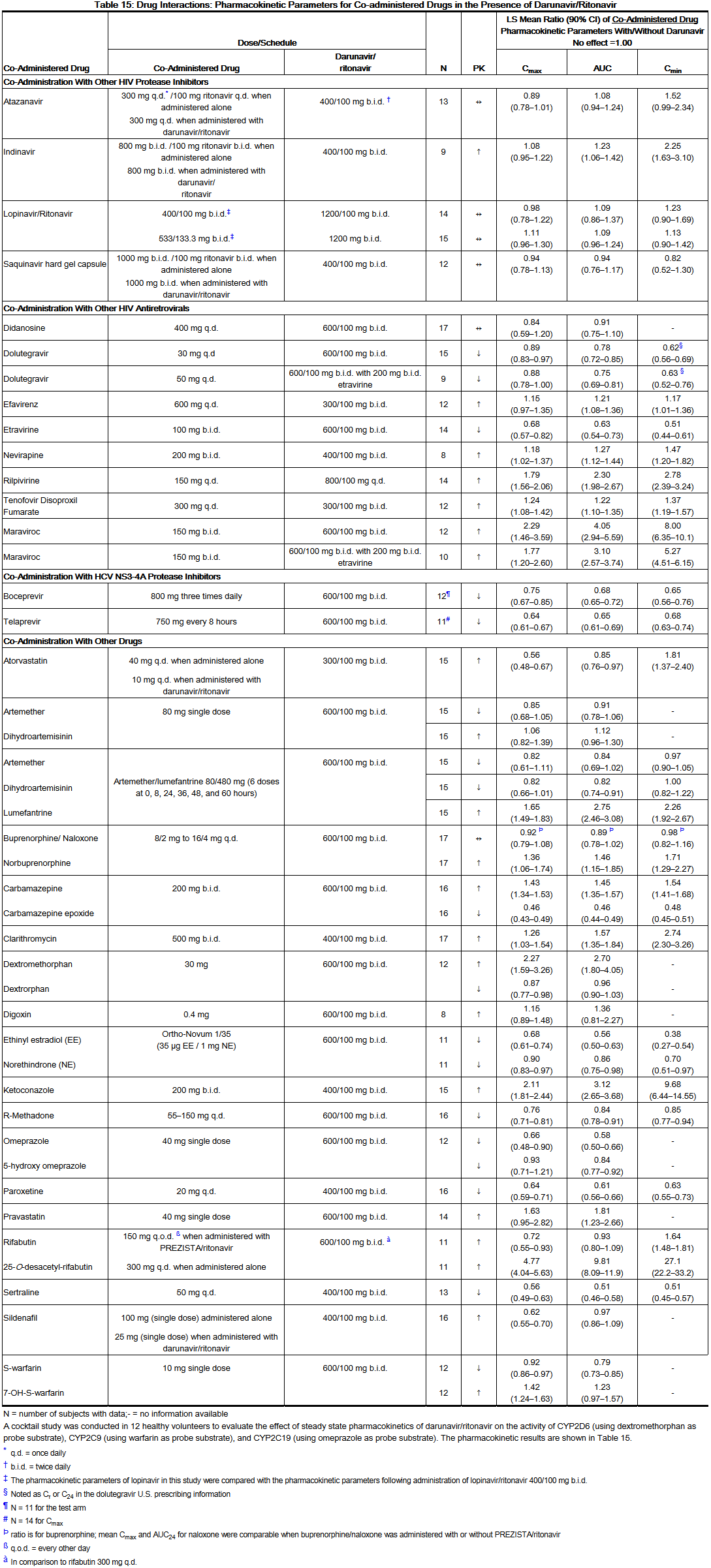
- In addition to the drugs included in Table 11, the interaction between Darunavir/ritonavir and the following drugs were evaluated in clinical studies and no dose adjustments are needed for either drug: atazanavir, efavirenz, etravirine, nevirapine, omeprazole, ranitidine, rilpivirine, and tenofovir disoproxil fumarate. Using cross-trial comparisons to historical pharmacokinetic data, dolutegravir did not appear to affect the pharmacokinetics of darunavir. Darunavir/ritonavir had no clinically significant effect on the pharmacokinetics of dolutegravir.
- Other nucleoside reverse transcriptase inhibitors (NRTIs):
- Based on the different elimination pathways of the other NRTIs (zidovudine, zalcitabine, emtricitabine, stavudine, lamivudine and abacavir) that are primarily renally excreted, no drug interactions are expected for these drugs and Darunavir/ritonavir.
- Other PIs:
- The co-administration of Darunavir/ritonavir and PIs other than lopinavir/ritonavir, saquinavir, atazanavir, and indinavir has not been studied. Therefore, such co-administration is not recommended.
- Integrase strand transfer inhibitors:
- Based on the pharmacokinetic data from literature references, either no clinically significant changes in darunavir concentrations or decreases in darunavir concentrations were observed with concomitant use of raltegravir. The potential decrease in darunavir concentrations does not appear to be clinically relevant. Darunavir/ritonavir and raltegravir can be used without dose adjustments.
Use in Specific Populations
Pregnancy
- Pregnancy Category C
- No adequate and well-controlled studies have been conducted in pregnant women. Reproduction studies conducted with darunavir showed no embryotoxicity or teratogenicity in mice and rats in the presence or absence of ritonavir as well as in rabbits with darunavir alone. In these studies, darunavir exposures (based on AUC) were higher in rats (3-fold), whereas in mice and rabbits, exposures were lower (less than 1-fold) compared to those obtained in humans at the recommended clinical dose of darunavir boosted with ritonavir.
- In the rat pre- and postnatal development study, a reduction in pup body weight gain was observed with darunavir alone or in combination with ritonavir during lactation. This was due to exposure of pups to drug substances via the milk. Sexual development, fertility and mating performance of offspring were not affected by maternal treatment with darunavir alone or in combination with ritonavir. The maximal plasma exposures achieved in rats were approximately 50% of those obtained in humans at the recommended clinical dose boosted with ritonavir.
- In the juvenile toxicity study where rats were directly dosed with darunavir, deaths occurred from post-natal day 5 through 11 at plasma exposure levels ranging from 0.1 to 1.0 of the human exposure levels. In a 4-week rat toxicology study, when dosing was initiated on post-natal day 23 (the human equivalent of 2 to 3 years of age), no deaths were observed with a plasma exposure (in combination with ritonavir) of 0.1 of the human plasma exposure levels.
- Antiretroviral Pregnancy Registry: To monitor maternal-fetal outcomes of pregnant women exposed to Darunavir, an Antiretroviral Pregnancy Registry has been established. Physicians are encouraged to register patients by calling 1-800-258-4263.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Darunavir in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Darunavir during labor and delivery.
Nursing Mothers
- The Centers for Disease Control and Prevention recommend that HIV-infected mothers in the United States not breastfeed their infants to avoid risking postnatal transmission of HIV. Although it is not known whether darunavir is secreted in human milk, darunavir is secreted into the milk of lactating rats. Because of both the potential for HIV transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breastfeed if they are receiving Darunavir.
Pediatric Use
- Do not administer Darunavir/ritonavir in pediatric patients below 3 years of age because of toxicity and mortality observed in juvenile rats dosed with darunavir (from 20 mg/kg to 1000 mg/kg) up to days 23 to 26 of age.
- The safety, pharmacokinetic profile, and virologic and immunologic responses of Darunavir/ritonavir were evaluated in treatment-experienced HIV-1-infected pediatric subjects 3 to less than 18 years of age and weighting at least 10 kg. These subjects were evaluated in clinical trials TMC114-C212 (80 subjects, 6 to less than 18 years of age) and TMC114-228 (21 subjects, 3 to less than 6 years of age). Frequency, type, and severity of adverse drug reactions in pediatric subjects were comparable to those observed in adults.
- In clinical trial TMC114-C230, the safety, pharmacokinetic profile and virologic and immunologic responses of Darunavir/ritonavir administered once daily were evaluated in treatment-naïve HIV-1 infected pediatric subjects 12 to less than 18 years of age (12 subjects). Frequency, type, and severity of adverse drug reactions in pediatric subjects were comparable to those observed in adults. Once daily dosing recommendations for pediatric patients 3 to less than 12 years of age were derived using population pharmacokinetic modeling and simulation. Although a Darunavir/ritonavir once daily dosing pediatric trial was not conducted in children less than 12 years of age, there is sufficient clinical safety data to support the predicted Darunavir exposures for the dosing recommendations in this age group.
Geriatic Use
- Clinical studies of Darunavir did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. In general, caution should be exercised in the administration and monitoring of Darunavir in elderly patients, reflecting the greater frequency of decreased hepatic function, and of concomitant disease or other drug therapy.
Gender
There is no FDA guidance on the use of Darunavir with respect to specific gender populations.
Race
There is no FDA guidance on the use of Darunavir with respect to specific racial populations.
Renal Impairment
- Population pharmacokinetic analysis showed that the pharmacokinetics of darunavir were not significantly affected in HIV-infected subjects with moderate renal impairment (CrCL between 30–60 mL/min, n=20). No pharmacokinetic data are available in HIV-1-infected patients with severe renal impairment or end stage renal disease; however, because the renal clearance of darunavir is limited, a decrease in total body clearance is not expected in patients with renal impairment. As darunavir and ritonavir are highly bound to plasma proteins, it is unlikely that they will be significantly removed by hemodialysis or peritoneal dialysis.
Hepatic Impairment
- No dose adjustment of Darunavir/ritonavir is necessary for patients with either mild or moderate hepatic impairment. No pharmacokinetic or safety data are available regarding the use of Darunavir/ritonavir in subjects with severe hepatic impairment. Therefore, Darunavir/ritonavir is not recommended for use in patients with severe hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Darunavir in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Darunavir in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
- Increased AST/ALT monitoring should be considered in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases, especially during the first several months of Darunavir/ritonavir treatment.
IV Compatibility
There is limited information regarding IV Compatibility of Darunavir in the drug label.
Overdosage
Acute Overdose
Signs and Symptoms
- Human experience of acute overdose with Darunavir/ritonavir is limited. Single doses up to 3200 mg of the oral solution of darunavir alone and up to 1600 mg of the tablet formulation of darunavir in combination with ritonavir have been administered to healthy volunteers without untoward symptomatic effects.
Management
- No specific antidote is available for overdose with Darunavir. Treatment of overdose with Darunavir consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. If indicated, elimination of unabsorbed active substance is to be achieved by emesis. Administration of activated charcoal may also be used to aid in removal of unabsorbed active substance. Since Darunavir is highly protein bound, dialysis is unlikely to be beneficial in significant removal of the active substance.
Chronic Overdose
There is limited information regarding Chronic Overdose of Darunavir in the drug label.
Pharmacology
Mechanism of Action
- Darunavir is an inhibitor of the HIV-1 protease. It selectively inhibits the cleavage of HIV-1 encoded Gag-Pol polyproteins in infected cells, thereby preventing the formation of mature virus particles.
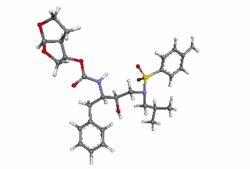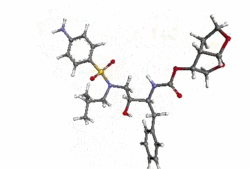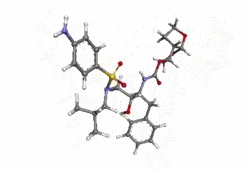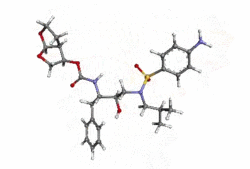
Structure
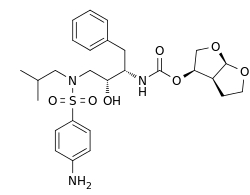
- Darunavir (darunavir) is an inhibitor of the human immunodeficiency virus (HIV-1) protease.
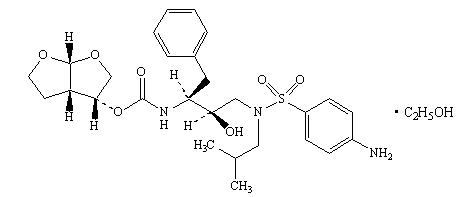
- Darunavir (darunavir), in the form of darunavir ethanolate, has the following chemical name: [(1S,2R)-3-[(4-aminophenyl)sulfonyl](2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]-carbamic acid (3R,3aS,6aR)-hexahydrofuro[2,3-b]furan-3-yl ester monoethanolate. Its molecular formula is C27H37N3O7S • C2H5OH and its molecular weight is 593.73. Darunavir ethanolate has the following structural formula:
- Darunavir ethanolate is a white to off-white powder with a solubility of approximately 0.15 mg/mL in water at 20°C.
- Darunavir 100 mg/mL oral suspension is available as a white to off-white opaque suspension for oral administration.
- Darunavir 75 mg tablets are available as white, caplet-shaped, film-coated tablets for oral administration. Darunavir 150 mg tablets are available as white, oval-shaped, film-coated tablets for oral administration. Darunavir 600 mg tablets are available as orange, oval-shaped, film-coated tablets for oral administration. Darunavir 800 mg tablets are available as dark red, oval-shaped, film-coated tablets for oral administration.
- Each mL of the oral suspension contains darunavir ethanolate equivalent to 100 mg darunavir. In addition, each mL contains the inactive ingredients hydroxypropyl cellulose, microcrystalline cellulose, sodium carboxymethylcellulose, methylparaben sodium, citric acid monohydrate, sucralose, masking flavor, strawberry cream flavor, hydrochloric acid (for pH adjustment) and purified water.
- Each 75 mg tablet contains darunavir ethanolate equivalent to 75 mg of darunavir. Each 150 mg tablet contains darunavir ethanolate equivalent to 150 mg of darunavir. Each 600 mg tablet contains darunavir ethanolate equivalent to 600 mg of darunavir. Each 800 mg tablet contains darunavir ethanolate equivalent to 800 mg of darunavir. During storage, partial conversion from ethanolate to hydrate may occur; however, this does not affect product quality or performance. Each tablet also contains the inactive ingredients colloidal silicon dioxide, crospovidone, magnesium stearate, and microcrystalline cellulose. The 800 mg tablet also contains hypromellose. The 75 and 150 mg tablet film coating, OPADRY® White, contains polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide. The 600 mg tablet film coating, OPADRY® Orange, contains FD&C Yellow No. 6, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide. The 800 mg tablet film coating, OPADRY® Dark Red, contains iron oxide red, polyethylene glycol 3350, polyvinyl alcohol-partially hydrolyzed, talc, and titanium dioxide.
- All dosages for Darunavir are expressed in terms of the free form of darunavir.
Pharmacodynamics
- In an open-label, randomized, placebo- and active-controlled, four-way crossover trial, 40 healthy subjects were administered supratheraputic doses of darunavir/ritonavir 1600/100 mg once daily and 800/100 mg twice daily for seven days.
- At the mean maximum darunavir concentration of 6599 ng/mL observed in this study, the mean increase in QTcF was 2.2 ms with a 90% two-sided confidence interval (CI) of -2.0 to 6.3 ms. When evaluating the 2-sided 90% CI on the time-matched mean changes in QTcF versus placebo control, the upper bounds of both darunavir/ritonavir groups never exceeded the 10 ms boundary. In the setting of this trial, darunavir/ritonavir did not appear to prolong the QTc interval.
Pharmacokinetics
Pharmacokinetics in Adults
- General
- Darunavir is primarily metabolized by CYP3A. Ritonavir inhibits CYP3A, thereby increasing the plasma concentrations of darunavir. When a single dose of Darunavir 600 mg was given orally in combination with 100 mg ritonavir twice daily, there was an approximate 14-fold increase in the systemic exposure of darunavir. Therefore, Darunavir should only be used in combination with 100 mg of ritonavir to achieve sufficient exposures of darunavir.
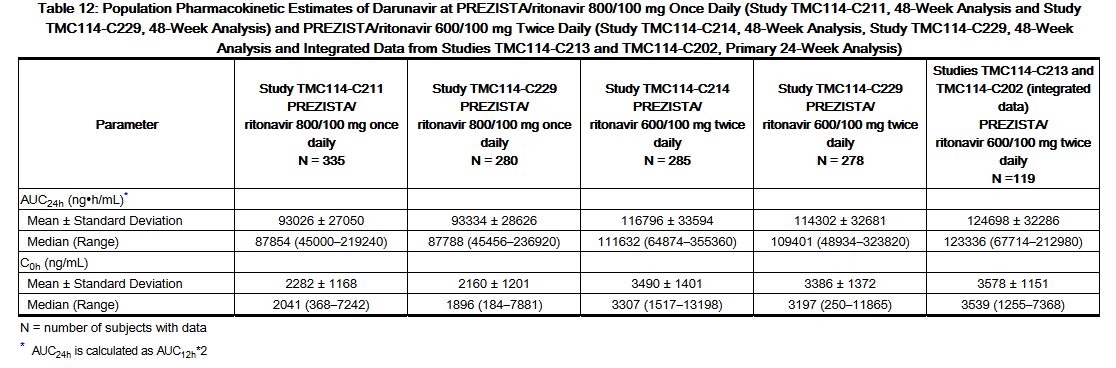
- The pharmacokinetics of darunavir, co-administered with low dose ritonavir (100 mg), has been evaluated in healthy adult volunteers and in HIV-1-infected subjects. Table 12 displays the population pharmacokinetic estimates of darunavir after oral administration of Darunavir/ritonavir 600/100 mg twice daily [based on sparse sampling in 285 patients in study TMC114-C214, 278 patients in Study TMC114-C229 and 119 patients (integrated data) from Studies TMC114-C202 and TMC114-C213] and Darunavir/ritonavir 800/100 mg once daily [based on sparse sampling in 335 patients in Study TMC114-C211 and 280 patients in Study TMC114-C229] to HIV-1-infected patients.
- Absorption and Bioavailability
- Darunavir, co-administered with 100 mg ritonavir twice daily, was absorbed following oral administration with a Tmax of approximately 2.5–4 hours. The absolute oral bioavailability of a single 600 mg dose of darunavir alone and after co-administration with 100 mg ritonavir twice daily was 37% and 82%, respectively. In vivo data suggest that darunavir/ritonavir is an inhibitor of the p-glycoprotein (p-gp) transporters.
- Effects of Food on Oral Absorption
- When Darunavir tablets were administered with food, the Cmax and AUC of darunavir, co-administered with ritonavir, is approximately 40% higher relative to the fasting state. Therefore, Darunavir tablets, co-administered with ritonavir, should always be taken with food. Within the range of meals studied, darunavir exposure is similar. The total caloric content of the various meals evaluated ranged from 240 Kcal (12 gms fat) to 928 Kcal (56 gms fat).
- Distribution
- Darunavir is approximately 95% bound to plasma proteins. Darunavir binds primarily to plasma alpha 1-acid glycoprotein (AAG).
- Metabolism
- In vitro experiments with human liver microsomes (HLMs) indicate that darunavir primarily undergoes oxidative metabolism. Darunavir is extensively metabolized by CYP enzymes, primarily by CYP3A. A mass balance study in healthy volunteers showed that after a single dose administration of 400 mg 14C-darunavir, co-administered with 100 mg ritonavir, the majority of the radioactivity in the plasma was due to darunavir. At least 3 oxidative metabolites of darunavir have been identified in humans; all showed activity that was at least 90% less than the activity of darunavir against wild-type HIV-1.
- Elimination
- A mass balance study in healthy volunteers showed that after single dose administration of 400 mg 14C-darunavir, co-administered with 100 mg ritonavir, approximately 79.5% and 13.9% of the administered dose of 14C-darunavir was recovered in the feces and urine, respectively. Unchanged darunavir accounted for approximately 41.2% and 7.7% of the administered dose in feces and urine, respectively. The terminal elimination half-life of darunavir was approximately 15 hours when co-administered with ritonavir. After intravenous administration, the clearance of darunavir, administered alone and co-administered with 100 mg twice daily ritonavir, was 32.8 L/h and 5.9 L/h, respectively.
- Special Populations
- Hepatic Impairment
- Darunavir is primarily metabolized by the liver. The steady-state pharmacokinetic parameters of darunavir were similar after multiple dose co-administration of Darunavir/ritonavir 600/100 mg twice daily to subjects with normal hepatic function (n=16), mild hepatic impairment (Child-Pugh Class A, n=8), and moderate hepatic impairment (Child-Pugh Class B, n=8). The effect of severe hepatic impairment on the pharmacokinetics of darunavir has not been evaluated.
- Hepatitis B or Hepatitis C Virus Co-infection
- The 48-week analysis of the data from Studies TMC114-C211 and TMC114-C214 in HIV-1-infected subjects indicated that hepatitis B and/or hepatitis C virus co-infection status had no apparent effect on the exposure of darunavir.
- Renal Impairment
- Results from a mass balance study with 14C-darunavir/ritonavir showed that approximately 7.7% of the administered dose of darunavir is excreted in the urine as unchanged drug. As darunavir and ritonavir are highly bound to plasma proteins, it is unlikely that they will be significantly removed by hemodialysis or peritoneal dialysis. Population pharmacokinetic analysis showed that the pharmacokinetics of darunavir were not significantly affected in HIV-1-infected subjects with moderate renal impairment (CrCL between 30–60 mL/min, n=20). There are no pharmacokinetic data available in HIV-1-infected patients with severe renal impairment or end stage renal disease.
- Gender
- Population pharmacokinetic analysis showed higher mean darunavir exposure in HIV-1-infected females compared to males. This difference is not clinically relevant.
- Race
- Population pharmacokinetic analysis of darunavir in HIV-1-infected subjects indicated that race had no apparent effect on the exposure to darunavir.
- Geriatric Patients
- Population pharmacokinetic analysis in HIV-1-infected subjects showed that darunavir pharmacokinetics are not considerably different in the age range (18 to 75 years) evaluated in HIV-1-infected subjects (n=12, age greater than or equal to 65).
- Pediatric Patients
- Darunavir/ritonavir administered twice daily:
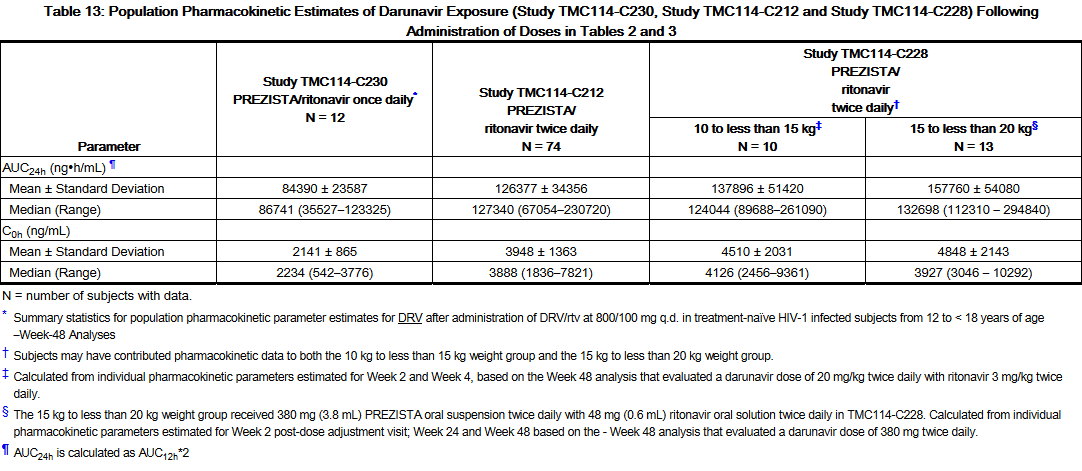
- The pharmacokinetics of darunavir in combination with ritonavir in 93 antiretroviral treatment-experienced HIV-1-infected pediatric subjects 3 to less than 18 years of age and weighing at least 10 kg showed that the administered weight-based dosages resulted in similar darunavir exposure when compared to the darunavir exposure achieved in treatment-experienced adults receiving Darunavir/ritonavir 600/100 mg twice daily.
- Darunavir/ritonavir administered once daily:
- The pharmacokinetics of darunavir in combination with ritonavir in 12 antiretroviral treatment-naïve HIV-1-infected pediatric subjects 12 to less than 18 years of age and weighing at least 40 kg receiving Darunavir/ritonavir 800/100 mg once daily resulted in similar darunavir exposures when compared to the darunavir exposure achieved in treatment-naive adults receiving Darunavir/ritonavir 800/100 mg once daily.
- Based on population pharmacokinetic modeling and simulation, the proposed Darunavir/ritonavir once daily dosing regimens for pediatric patients 3 to less than 12 years of age is predicted to result in similar darunavir exposures when compared to the darunavir exposures achieved in treatment-naïve adults receiving Darunavir/ritonavir 800/100 mg once daily.
- The population pharmacokinetic parameters in pediatric subjects with Darunavir/ritonavir administered once or twice daily are summarized in the table below.
- Drug Interactions
- Darunavir co-administered with ritonavir is an inhibitor of CYP3A and CYP2D6. Co-administration of darunavir and ritonavir with drugs primarily metabolized by CYP3A and CYP2D6 may result in increased plasma concentrations of such drugs, which could increase or prolong their therapeutic effect and adverse events.
- Darunavir and ritonavir are metabolized by CYP3A. Drugs that induce CYP3A activity would be expected to increase the clearance of darunavir and ritonavir, resulting in lowered plasma concentrations of darunavir and ritonavir. Co-administration of darunavir and ritonavir and other drugs that inhibit CYP3A may decrease the clearance of darunavir and ritonavir and may result in increased plasma concentrations of darunavir and ritonavir.
- Drug interaction studies were performed with darunavir and other drugs likely to be co-administered and some drugs commonly used as probes for pharmacokinetic interactions. The effects of co-administration of darunavir on the AUC, Cmax, and Cmin values are summarized in Table 14 (effect of other drugs on darunavir) and Table 15 (effect of darunavir on other drugs).
- Several interaction studies have been performed with a dose other than the recommended dose of the co-administered drug or darunavir; however, the results are applicable to the recommended dose of the co-administered drug and/or darunavir.
Microbiology
- Mechanism of Action
- Darunavir is an inhibitor of the HIV-1 protease. It selectively inhibits the cleavage of HIV-1 encoded Gag-Pol polyproteins in infected cells, thereby preventing the formation of mature virus particles.
- Antiviral Activity
- Darunavir exhibits activity against laboratory strains and clinical isolates of HIV-1 and laboratory strains of HIV-2 in acutely infected T-cell lines, human peripheral blood mononuclear cells and human monocytes/macrophages with median EC50 values ranging from 1.2 to 8.5 nM (0.7 to 5.0 ng/mL). Darunavir demonstrates antiviral activity in cell culture against a broad panel of HIV-1 group M (A, B, C, D, E, F, G), and group O primary isolates with EC50 values ranging from less than 0.1 to 4.3 nM. The EC50 value of darunavir increases by a median factor of 5.4 in the presence of human serum. Darunavir did not show antagonism when studied in combination with the PIs amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, ritonavir, saquinavir, or tipranavir, the N(t)RTIs abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir, zalcitabine, or zidovudine, the NNRTIs delavirdine, rilpivirine, efavirenz, etravirine, or nevirapine, and the fusion inhibitor enfuvirtide.
- Resistance
- Cell Culture: HIV-1 isolates with a decreased susceptibility to darunavir have been selected in cell culture and obtained from subjects treated with darunavir/ritonavir. Darunavir-resistant virus derived in cell culture from wild-type HIV-1 had 21- to 88-fold decreased susceptibility to darunavir and developed 2 to 4 of the following amino acid substitutions S37D, R41E/T, K55Q, H69Q, K70E, T74S, V77I, or I85V in the protease. Selection in cell culture of darunavir resistant HIV-1 from nine HIV-1 strains harboring multiple PI resistance-associated mutations resulted in the overall emergence of 22 mutations in the protease gene, coding for amino acid substitutions L10F, V11I, I13V, I15V, G16E, L23I, V32I, L33F, S37N, M46I, I47V, I50V, F53L, L63P, A71V, G73S, L76V, V82I, I84V, T91A/S, and Q92R, of which L10F, V32I, L33F, S37N, M46I, I47V, I50V, L63P, A71V, and I84V were the most prevalent. These darunavir-resistant viruses had at least eight protease substitutions and exhibited 50- to 641-fold decreases in darunavir susceptibility with final EC50 values ranging from 125 nM to 3461 nM.
- Clinical studies of Darunavir/ritonavir in treatment-experienced subjects: In a pooled analysis of the 600/100 mg Darunavir/ritonavir twice daily arms of Studies TMC114-C213, TMC114-C202, TMC114-C215, and the control arms of etravirine studies TMC125-C206 and TMC125-C216, the amino acid substitutions V32I and I54L or M developed most frequently on Darunavir/ritonavir in 41% and 25%, respectively, of the treatment-experienced subjects who experienced virologic failure, either by rebound or by never being suppressed (less than 50 copies/mL). Other substitutions that developed frequently in Darunavir/ritonavir virologic failure isolates occurred at amino acid positions V11I, I15V, L33F, I47V, I50V, and L89V. These amino acid substitutions were associated with decreased susceptibility to darunavir; 90% of the virologic failure isolates had a greater than 7-fold decrease in susceptibility to darunavir at failure. The median darunavir phenotype (fold change from reference) of the virologic failure isolates was 4.3-fold at baseline and 85-fold at failure. Amino acid substitutions were also observed in the protease cleavage sites in the Gag polyprotein of some Darunavir/ritonavir virologic failure isolates. In Study TMC114-C212 of treatment-experienced pediatric subjects, the amino acid substitutions V32I, I54L and L89M developed most frequently in virologic failures on Darunavir/ritonavir.
- In the 96-week as-treated analysis of the Phase 3 Study TMC114-C214, the percent of virologic failures (never suppressed, rebounders and discontinued before achieving suppression) was 21% (62/298) in the group of subjects receiving Darunavir/ritonavir 600/100 mg twice daily compared to 32% (96/297) of subjects receiving lopinavir/ritonavir 400/100 mg twice daily. Examination of subjects who failed on Darunavir/ritonavir 600/100 mg twice daily and had post-baseline genotypes and phenotypes showed that 7 subjects (7/43; 16%) developed PI substitutions on darunavir/ritonavir treatment resulting in decreased susceptibility to darunavir. Six of the 7 had baseline PI resistance-associated substitutions and baseline darunavir phenotypes greater than 7. The most common emerging PI substitutions in these virologic failures were V32I, L33F, M46I or L, I47V, I54L, T74P and L76V. These amino acid substitutions were associated with 59- to 839-fold decreased susceptibility to darunavir at failure. Examination of individual subjects who failed in the comparator arm on lopinavir/ritonavir and had post-baseline genotypes and phenotypes showed that 31 subjects (31/75; 41%) developed substitutions on lopinavir treatment resulting in decreased susceptibility to lopinavir (greater than 10-fold) and the most common substitutions emerging on treatment were L10I or F, M46I or L, I47V or A, I54V and L76V. Of the 31 lopinavir/ritonavir virologic failure subjects, 14 had reduced susceptibility (greater than 10-fold) to lopinavir at baseline.
- In the 48-week analysis of the Phase 3 Study TMC114-C229, the number of virologic failures (including those who discontinued before suppression after Week 4) was 26% (75/294) in the group of subjects receiving Darunavir/ritonavir 800/100 mg once daily compared to 19% (56/296) of subjects receiving Darunavir/ritonavir 600/100 mg twice daily. Examination of isolates from subjects who failed on Darunavir/ritonavir 800/100 mg once daily and had post-baseline genotypes showed that 8 subjects (8/60; 13%) had isolates that developed IAS-USA defined PI resistance-associated substitutions compared to 5 subjects (5/39; 13%) on Darunavir/ritonavir 600/100 mg twice daily. Isolates from 2 subjects developed PI resistance associated substitutions associated with decreased susceptibility to darunavir; 1 subject isolate in the Darunavir/ritonavir 800/100 mg once daily arm, developed substitutions V32I, M46I, L76V and I84V associated with a 24-fold decreased susceptibility to darunavir, and 1 subject isolate in the Darunavir/ritonavir 600/100 mg twice daily arm developed substitutions L33F and I50V associated with a 40-fold decreased susceptibility to darunavir. In the Darunavir/ritonavir 800/100 mg once daily and Darunavir/ritonavir 600/100 mg twice daily groups, isolates from 7 (7/60, 12%) and 4 (4/42, 10%) virologic failures, respectively, developed decreased susceptibility to an NRTI included in the treatment regimen.
- Clinical studies of Darunavir/ritonavir in treatment-naive subjects: In the 192-week as-treated analysis censoring those who discontinued before Week 4 of the Phase 3 Study TMC114-C211, the percentage of virologic failures (never suppressed, rebounders and discontinued before achieving suppression) was 22% (64/288) in the group of subjects receiving Darunavir/ritonavir 800/100 mg once daily compared to 29% (76/263) of subjects receiving lopinavir/ritonavir 800/200 mg per day. In the Darunavir/ritonavir arm, emergent PI resistance-associated substitutions were identified in 11 of the virologic failures with post-baseline genotypic data (n=43). However, none of the darunavir virologic failures had a decrease in darunavir susceptibility (greater than 7-fold change) at failure. In the comparator lopinavir/ritonavir arm, emergent PI resistance-associated substitutions were identified in 17 of the virologic failures with post-baseline genotypic data (n=53), but none of the lopinavir/ritonavir virologic failures had decreased susceptibility to lopinavir (greater than 10-fold change) at failure. The reverse transcriptase M184V substitution and/or resistance to emtricitabine, which was included in the fixed background regimen, was identified in 4 virologic failures from the Darunavir/ritonavir arm and 7 virologic failures in the lopinavir/ritonavir arm.
- Cross-resistance
- Cross-resistance among PIs has been observed. Darunavir has a less than 10-fold decreased susceptibility in cell culture against 90% of 3309 clinical isolates resistant to amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, ritonavir, saquinavir and/or tipranavir showing that viruses resistant to these PIs remain susceptible to darunavir.
- Darunavir-resistant viruses were not susceptible to amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, ritonavir or saquinavir in cell culture. However, six of nine darunavir-resistant viruses selected in cell culture from PI-resistant viruses showed a fold change in EC50 values less than 3 for tipranavir, indicative of limited cross-resistance between darunavir and tipranavir. In Studies TMC114-C213, TMC114-C202, and TMC114-C215, 34% (64/187) of subjects in the darunavir/ritonavir arm whose baseline isolates had decreased susceptibility to tipranavir (tipranavir fold change greater than 3) achieved less than 50 copies/mL serum HIV-1 RNA levels at Week 96. Of the viruses isolated from subjects experiencing virologic failure on Darunavir/ritonavir 600/100 mg twice daily (greater than 7 fold change), 41% were still susceptible to tipranavir and 10% were susceptible to saquinavir while less than 2% were susceptible to the other protease inhibitors (amprenavir, atazanavir, indinavir, lopinavir or nelfinavir).
- In Study TMC114-C214, the 7 darunavir/ritonavir virologic failures with reduced susceptibility to darunavir at failure were also resistant to the approved PIs (fos)amprenavir, atazanavir, lopinavir, indinavir, and nelfinavir at failure. Six of these 7 were resistant to saquinavir and 5 were resistant to tipranavir. Four of these virologic failures were already PI-resistant at baseline.
- Cross-resistance between darunavir and nucleoside/nucleotide reverse transcriptase inhibitors, non-nucleoside reverse transcriptase inhibitors, fusion inhibitors, CCR5 co-receptor antagonists, or integrase inhibitors is unlikely because the viral targets are different.
- Baseline Genotype/Phenotype and Virologic Outcome Analyses
- Genotypic and/or phenotypic analysis of baseline virus may aid in determining darunavir susceptibility before initiation of Darunavir/ritonavir 600/100 mg twice daily therapy. The effect of baseline genotype and phenotype on virologic response at 96 weeks was analyzed in as-treated analyses using pooled data from the Phase 2b studies (Studies TMC114-C213, TMC114-C202, and TMC114-C215) (n=439). The findings were confirmed with additional genotypic and phenotypic data from the control arms of etravirine Studies TMC125-C206 and TMC125-C216 at Week 24 (n=591).
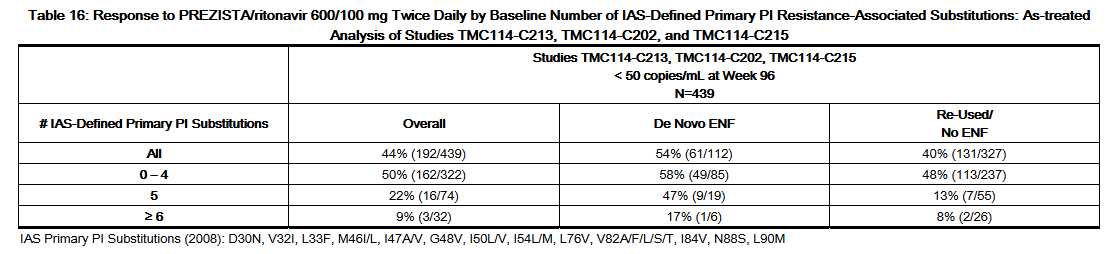
- Diminished virologic responses were observed in subjects with 5 or more baseline IAS-defined primary protease inhibitor resistance-associated substitutions (D30N, V32I, L33F, M46I/L, I47A/V, G48V, I50L/V, I54L/M, L76V, V82A/F/L/S/T, I84V, N88S, L90M) (see Table 16).
- The presence at baseline of two or more of the substitutions V11I, V32I, L33F, I47V, I50V, I54L or M, T74P, L76V, I84V or L89V was associated with a decreased virologic response to Darunavir/ritonavir. In subjects not taking enfuvirtide de novo, the proportion of subjects achieving viral load less than 50 plasma HIV-1 RNA copies/mL at 96 weeks was 59%, 29%, and 12% when the baseline genotype had 0–1, 2 and greater than or equal to 3 of these substitutions, respectively.
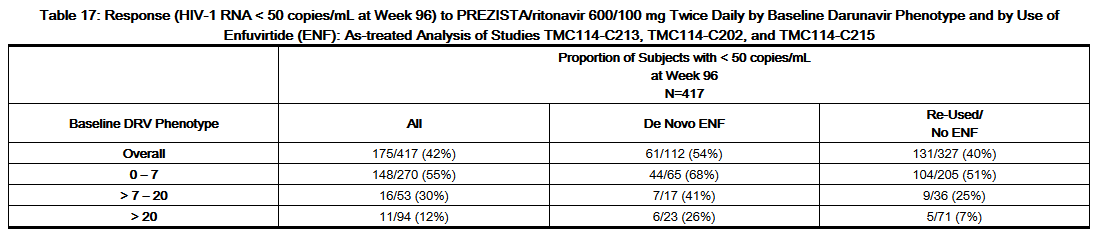
- Baseline darunavir phenotype (shift in susceptibility relative to reference) was shown to be a predictive factor of virologic outcome. Response rates assessed by baseline darunavir phenotype are shown in Table 17. These baseline phenotype groups are based on the select patient populations in the Studies TMC114-C213, TMC114-C202, and TMC114-C215, and are not meant to represent definitive clinical susceptibility breakpoints for Darunavir/ritonavir. The data are provided to give clinicians information on the likelihood of virologic success based on pre-treatment susceptibility to darunavir.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis and Mutagenesis
- Darunavir was evaluated for carcinogenic potential by oral gavage administration to mice and rats up to 104 weeks. Daily doses of 150, 450 and 1000 mg/kg were administered to mice and doses of 50, 150 and 500 mg/kg was administered to rats. A dose-related increase in the incidence of hepatocellular adenomas and carcinomas were observed in males and females of both species as well as an increase in thyroid follicular cell adenomas in male rats. The observed hepatocellular findings in rodents are considered to be of limited relevance to humans. Repeated administration of darunavir to rats caused hepatic microsomal enzyme induction and increased thyroid hormone elimination, which predispose rats, but not humans, to thyroid neoplasms. At the highest tested doses, the systemic exposures to darunavir (based on AUC) were between 0.4- and 0.7-fold (mice) and 0.7- and 1-fold (rats), relative to those observed in humans at the recommended therapeutic doses (600/100 mg twice daily or 800/100 mg once daily).
- Darunavir was not mutagenic or genotoxic in a battery of in vitro and in vivo assays including bacterial reserve mutation (Ames), chromosomal aberration in human lymphocytes and in vivo micronucleus test in mice.
Impairment of Fertility
- No effects on fertility or early embryonic development were observed with darunavir in rats and darunavir has shown no teratogenic potential in mice or rats (in the presence or absence of ritonavir), and rabbits.
Animal Toxicology and/or Pharmacology
- In juvenile rats single doses of darunavir (20 mg/kg to 160 mg/kg at ages 5–11 days) or multiple doses of darunavir (40 mg/kg to 1000 mg/kg at age 12 days) caused mortality. The mortalities were associated with convulsions in some of the animals. Within this age range exposures in plasma, liver and brain were dose and age dependent and were considerably greater than those observed in adult rats. These findings were attributed to the ontogeny of the CYP450 liver enzymes involved in the metabolism of darunavir and the immaturity of the blood-brain barrier. No treatment-related mortalities were noted in juvenile rats after a single dose of darunavir at 1000 mg/kg on day 26 of age or after repeat dosing at 500 mg/kg from day 23 to 50 of age. The exposures and toxicity profile in the older animals (day 23 or day 26) were comparable to those observed in adult rats. Due to uncertainties regarding the rate of development of the human blood-brain barrier and liver enzymes, do not administer Darunavir/ritonavir in pediatric patients below 3 years of age.
Clinical Studies
Description of Adult Clinical Studies
- The evidence of efficacy of Darunavir/ritonavir is based on the analyses of 192-week data from a randomized, controlled open-label Phase 3 trial in treatment-naïve (TMC114-C211) HIV-1-infected adult subjects and 96-week data from a randomized, controlled, open-label Phase 3 trial in antiretroviral treatment-experienced (TMC114-C214) HIV-1-infected adult subjects. In addition, 96-week data are included from 2 randomized, controlled Phase 2b trials, TMC114-C213 and TMC114-C202, in antiretroviral treatment-experienced HIV-1-infected adult subjects.
Treatment-Naïve Adult Subjects
- Study TMC114-C211
- Study TMC114-C211 is a randomized, controlled, open-label Phase 3 trial comparing Darunavir/ritonavir 800/100 mg once daily versus lopinavir/ritonavir 800/200 mg per day (given as a twice daily or as a once daily regimen) in antiretroviral treatment-naïve HIV-1-infected adult subjects. Both arms used a fixed background regimen consisting of tenofovir disoproxil fumarate 300 mg once daily (TDF) and emtricitabine 200 mg once daily (FTC).
- HIV-1-infected subjects who were eligible for this trial had plasma HIV-1 RNA greater than or equal to 5000 copies/mL. Randomization was stratified by screening plasma viral load (HIV-1 RNA less than 100,000 copies/mL or greater than or equal to 100,000 copies/mL) and screening CD4+ cell count (less than 200 cells/mm3 or greater than or equal to 200 cells/mm3). Virologic response was defined as a confirmed plasma HIV-1 RNA viral load less than 50 copies/mL. Analyses included 689 subjects in Study TMC114-C211 who had completed 192 weeks of treatment or discontinued earlier.
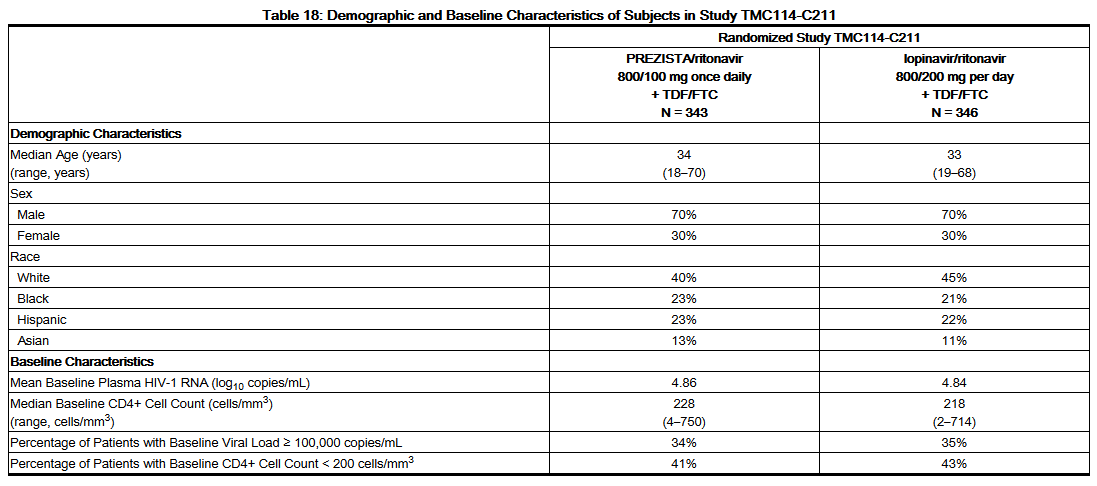
- Demographics and baseline characteristics were balanced between the Darunavir/ritonavir arm and the lopinavir/ritonavir arm (see Table 18). Table 18 compares the demographic and baseline characteristics between subjects in the Darunavir/ritonavir 800/100 mg once daily arm and subjects in the lopinavir/ritonavir 800/200 mg per day arm in Study TMC114-C211.
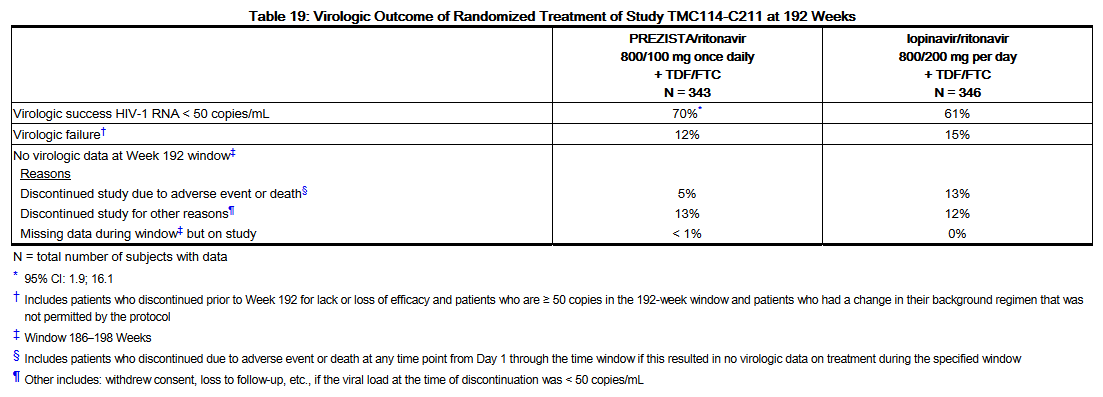
- Week 192 outcomes for subjects on Darunavir/ritonavir 800/100 mg once daily from Study TMC114-C211 are shown inTable 19.
- In Study TMC114-C211 at 192 weeks of treatment, the median increase from baseline in CD4+ cell counts was 258 cells/mm3 in the Darunavir/ritonavir 800/100 mg once daily arm and 263 cells/mm3 in the lopinavir/ritonavir 800/200 mg per day arm. Of the Darunavir/ritonavir subjects with a confirmed virologic response of < 50 copies/mL at Week 48, 81% remained undetectable at Week 192 versus 68% with lopinavir/ritonavir. In the 192 week analysis, statistical superiority of the Darunavir/ritonavir regimen over the lopinavir/ritonavir regimen was demonstrated for both ITT and OP populations.
Treatment-Experienced Adult Subjects
- Study TMC114-C229
- Study TMC114-C229 is a randomized, open-label trial comparing Darunavir/ritonavir 800/100 mg once daily to Darunavir/ritonavir 600/100 mg twice daily in treatment-experienced HIV-1-infected patients with screening genotype resistance test showing no darunavir resistance associated substitutions (i.e. V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V) and a screening viral load of greater than 1,000 HIV-1 RNA copies/mL. Both arms used an optimized background regimen consisting of greater than or equal to 2 NRTIs selected by the investigator.
- HIV-1-infected subjects who were eligible for this trial were on a highly active antiretroviral therapy regimen (HAART) for at least 12 weeks. Virologic response was defined as a confirmed plasma HIV-1 RNA viral load less than 50 copies/mL. Analyses included 590 subjects who had completed 48 weeks of treatment or discontinued earlier.
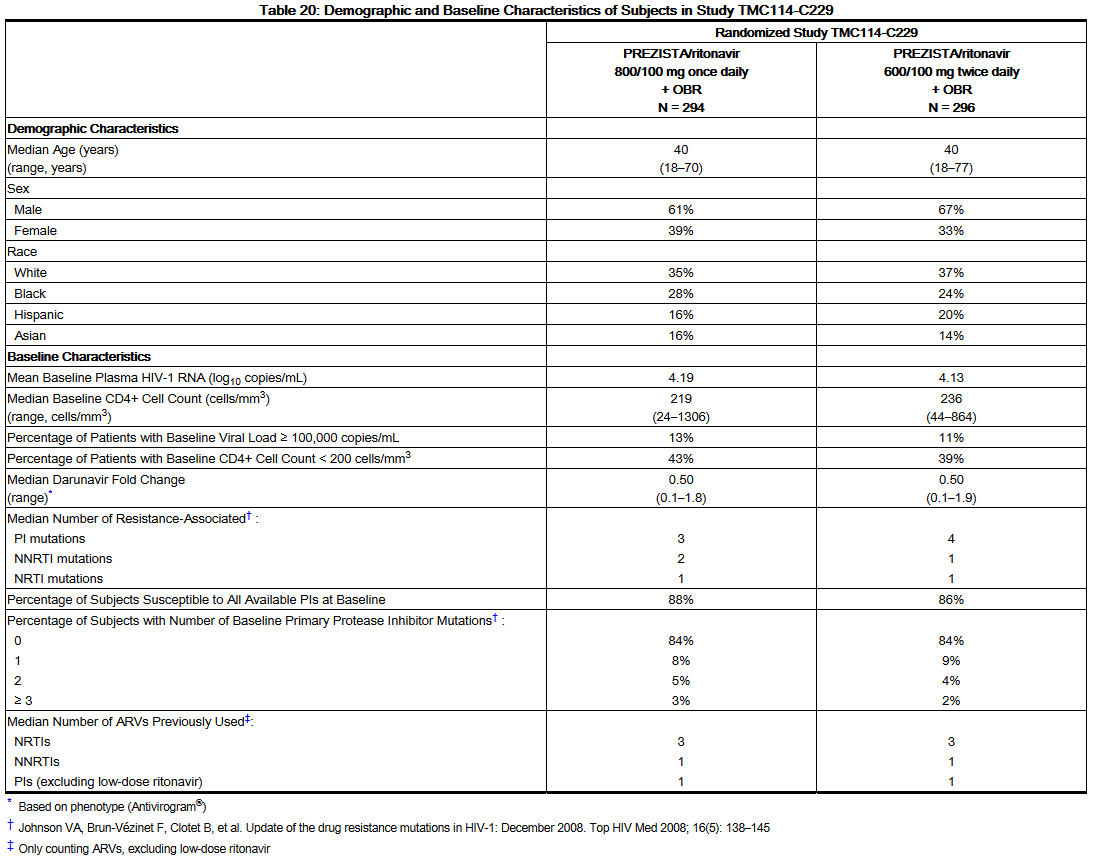
- Table 20 compares the demographic and baseline characteristics between subjects in the Darunavir/ritonavir 800/100 mg once daily arm and subjects in the Darunavir/ritonavir 600/100 mg twice daily arm in Study TMC114-C229. No imbalances between the 2 arms were noted.
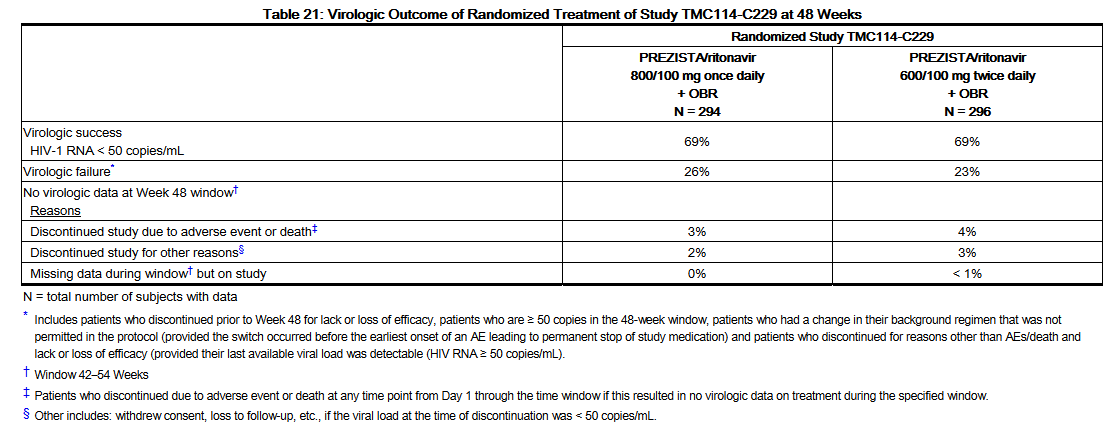
- Week 48 outcomes for subjects on Darunavir/ritonavir 800/100 mg once daily from Study TMC114-C229 are shown in Table 21.
- The mean increase from baseline in CD4+ cell counts was comparable for both treatment arms (108 cells/mm3 and 112 cells/mm3 in the Darunavir/ritonavir 800/100 mg once daily arm and the Darunavir/ritonavir 600/100 mg twice daily arm, respectively).
- Study TMC114-C214
- Study TMC114-C214 is a randomized, controlled, open-label Phase 3 trial comparing Darunavir/ritonavir 600/100 mg twice daily versus lopinavir/ritonavir 400/100 mg twice daily in antiretroviral treatment-experienced, lopinavir/ritonavir-naïve HIV-1-infected adult subjects. Both arms used an optimized background regimen (OBR) consisting of at least 2 antiretrovirals (NRTIs with or without NNRTIs).
- HIV-1-infected subjects who were eligible for this trial had plasma HIV-1 RNA greater than 1000 copies/mL and were on a highly active antiretroviral therapy regimen (HAART) for at least 12 weeks. Virologic response was defined as a confirmed plasma HIV-1 RNA viral load less than 400 copies/mL. Analyses included 595 subjects in Study TMC114-C214 who had completed 96 weeks of treatment or discontinued earlier.
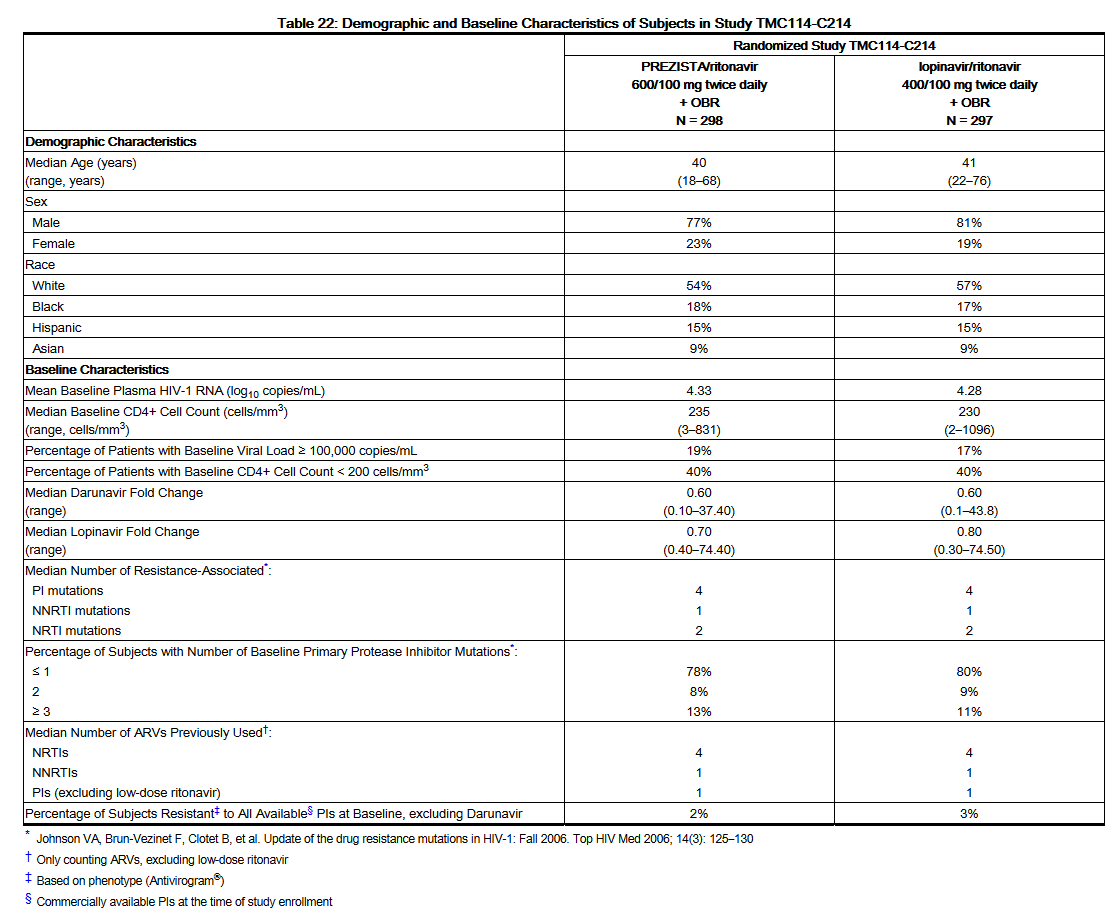
- Demographics and baseline characteristics were balanced between the Darunavir/ritonavir arm and the lopinavir/ritonavir arm (see Table 22). Table 22 compares the demographic and baseline characteristics between subjects in the Darunavir/ritonavir 600/100 mg twice daily arm and subjects in the lopinavir/ritonavir 400/100 mg twice daily arm in Study TMC114-C214.
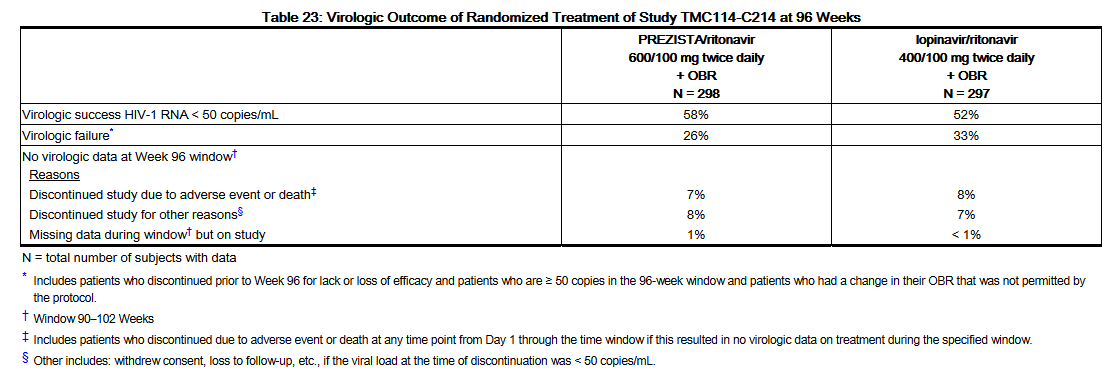
- Week 96 outcomes for subjects on Darunavir/ritonavir 600/100 mg twice daily from Study TMC114-C214 are shown inTable 23.
- In Study TMC114-C214 at 96 weeks of treatment, the median increase from baseline in CD4+ cell counts was 81 cells/mm3 in the Darunavir/ritonavir 600/100 mg twice daily arm and 93 cells/mm3 in the lopinavir/ritonavir 400/100 mg twice daily arm.
- Studies TMC114-C213 and TMC114-C202
- Studies TMC114-C213 and TMC114-C202 are randomized, controlled, Phase 2b trials in adult subjects with a high level of PI resistance consisting of 2 parts: an initial partially-blinded, dose-finding part and a second long-term part in which all subjects randomized to Darunavir/ritonavir received the recommended dose of 600/100 mg twice daily.
- HIV-1-infected subjects who were eligible for these trials had plasma HIV-1 RNA greater than 1000 copies/mL, had prior treatment with PI(s), NNRTI(s) and NRTI(s), had at least one primary PI mutation (D30N, M46I/L, G48V, I50L/V, V82A/F/S/T, I84V, L90M) at screening, and were on a stable PI-containing regimen at screening for at least 8 weeks. Randomization was stratified by the number of PI mutations, screening viral load, and the use of enfuvirtide.
- The virologic response rate was evaluated in subjects receiving Darunavir/ritonavir plus an OBR versus a control group receiving an investigator-selected PI(s) regimen plus an OBR. Prior to randomization, PI(s) and OBR were selected by the investigator based on genotypic resistance testing and prior ARV history. The OBR consisted of at least 2 NRTIs with or without enfuvirtide. Selected PI(s) in the control arm included: lopinavir in 36%, (fos)amprenavir in 34%, saquinavir in 35% and atazanavir in 17%; 98% of control subjects received a ritonavir boosted PI regimen out of which 23% of control subjects used dual-boosted PIs. Approximately 47% of all subjects used enfuvirtide, and 35% of the use was in subjects who were ENF-naïve. Virologic response was defined as a decrease in plasma HIV-1 RNA viral load of at least 1 log10 versus baseline.
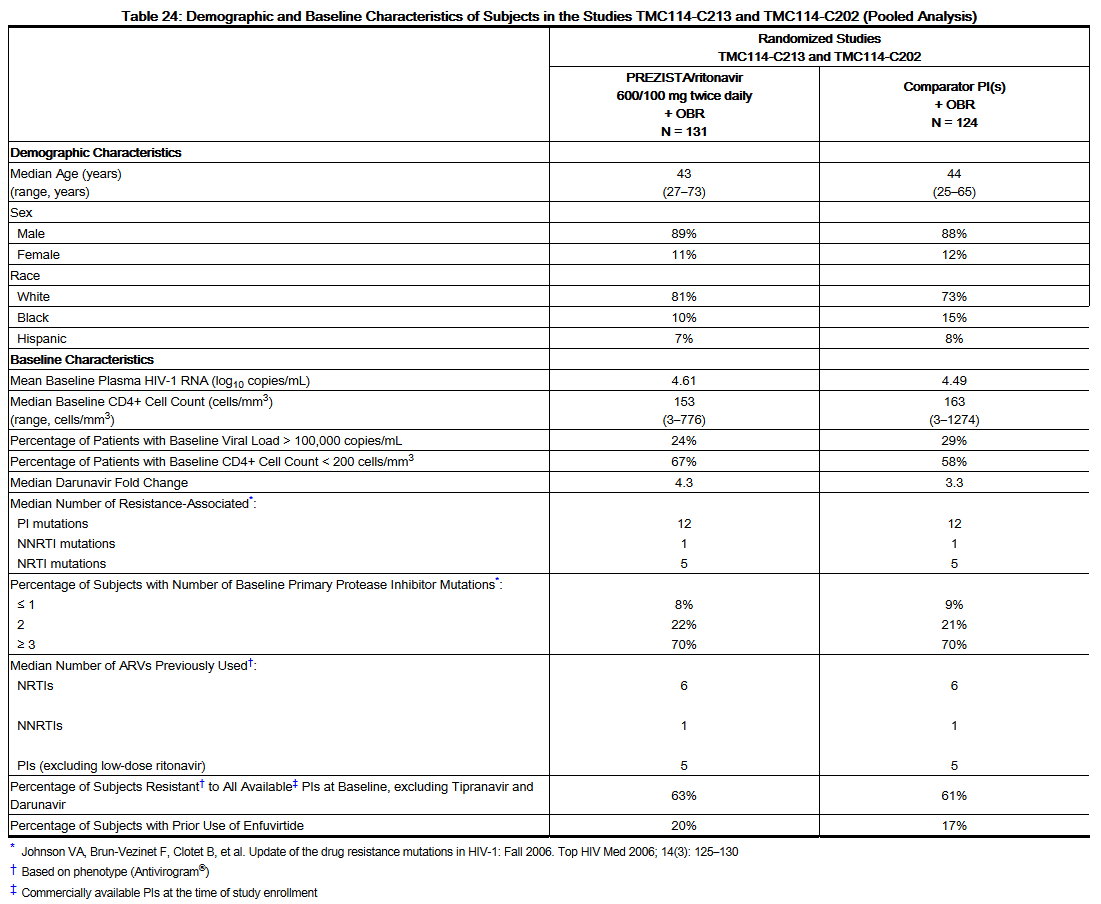
- In the pooled analysis for TMC114-C213 and TMC114-C202, demographics and baseline characteristics were balanced between the Darunavir/ritonavir arm and the comparator PI arm (see Table 24). Table 24 compares the demographic and baseline characteristics between subjects in the Darunavir/ritonavir 600/100 mg twice daily arm and subjects in the comparator PI arm in the pooled analysis of Studies TMC114-C213 and TMC114-C202.
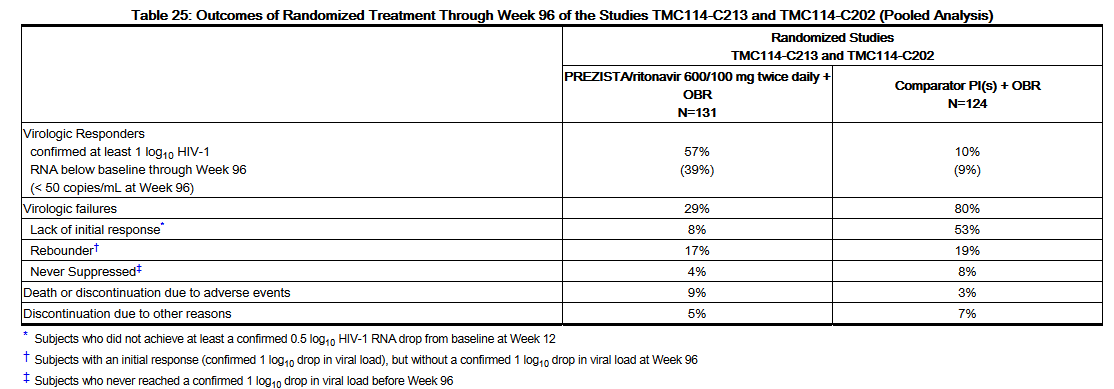
- Week 96 outcomes for subjects on the recommended dose Darunavir/ritonavir 600/100 mg twice daily from the pooled Studies TMC114-C213 and TMC114-C202 are shown inTable 25.
- In the pooled Studies TMC114-C213 and TMC114-C202 through 48 weeks of treatment, the proportion of subjects with HIV-1 RNA less than 400 copies/mL in the arm receiving Darunavir/ritonavir 600/100 mg twice daily compared to the comparator PI arm was 55.0% and 14.5%, respectively. In addition, the mean changes in plasma HIV-1 RNA from baseline were –1.69 log10 copies/mL in the arm receiving Darunavir/ritonavir 600/100 mg twice daily and –0.37 log10 copies/mL for the comparator PI arm. The mean increase from baseline in CD4+ cell counts was higher in the arm receiving Darunavir/ritonavir 600/100 mg twice daily (103 cells/mm3) than in the comparator PI arm (17 cells/mm3).
Pediatric Patients
- The pharmacokinetic profile, safety and antiviral activity of Darunavir/ritonavir were evaluated in 3 randomized, open-label, multicenter studies.
- Study TMC114-C212
- Treatment-experienced pediatric subjects between the ages of 6 and less than 18 years and weighing at least 20 kg were stratified according to their weight (greater than or equal to 20 kg to less than 30 kg, greater than or equal to 30 kg to less than 40 kg, greater than or equal to 40 kg) and received Darunavir tablets with either ritonavir capsules or oral solution plus background therapy consisting of at least two non-protease inhibitor antiretroviral drugs. Eighty patients were randomized and received at least one dose of Darunavir/ritonavir. Pediatric subjects who were at risk of discontinuing therapy due to intolerance of ritonavir oral solution (e.g., taste aversion) were allowed to switch to the capsule formulation. Of the 44 pediatric subjects taking ritonavir oral solution, 23 subjects switched to the 100 mg capsule formulation and exceeded the weight-based ritonavir dose without changes in observed safety.
- The 80 randomized pediatric subjects had a median age of 14 (range 6 to less than 18 years), and were 71% male, 54% Caucasian, 30% Black, 9% Hispanic and 8% other. The mean baseline plasma HIV-1 RNA was 4.64 log10 copies/mL, and the median baseline CD4+ cell count was 330 cells/mm3 (range: 6 to 1505 cells/mm3). Overall, 38% of pediatric subjects had baseline plasma HIV-1 RNA ≥ 100,000 copies/mL. Most pediatric subjects (79%) had previous use of at least one NNRTI and 96% of pediatric subjects had previously used at least one PI.
- Seventy-seven pediatric subjects (96%) completed the 24-week period. Of the patients who discontinued, one patient discontinued treatment due to an adverse event. An additional 2 patients discontinued for other reasons, one patient due to compliance and another patient due to relocation.
- The proportion of pediatric subjects with HIV-1 RNA less than 400 copies/mL and less than 50 copies/mL was 64% and 50%, respectively. The mean increase in CD4+ cell count from baseline was 117 cells/mm3.
- Study TMC114-C228
- Treatment-experienced pediatric subjects between the ages of 3 and less than 6 years and weighing greater than or equal to 10 kg to less than 20 kg received Darunavir oral suspension with ritonavir oral solution plus background therapy consisting of at least two active non-protease inhibitor antiretroviral drugs. Twenty-one subjects received at least one dose of Darunavir/ritonavir.
- The 21 subjects had a median age of 4.4 years (range 3 to less than 6 years), and were 48% male, 57% Black, 29%, Caucasian and 14% other. The mean baseline plasma HIV-1 was 4.34 log10 copies/mL, the median baseline CD4+ cell count was 927 × 106 cells/l (range: 209 to 2,429 × 106 cells/l) and the median baseline CD4+ percentage was 27.7% (range: 15.6% to 51.1%). Overall, 24% of subjects had a baseline plasma HIV-1 RNA greater than or equal to 100,000 copies/mL. All subjects had used greater than or equal to 2 NRTIs, 62% of subjects had used greater than or equal to 1 NNRTI and 76% had previously used at least one HIV PI.
- Twenty subjects (95%) completed the 48 week period. One subject prematurely discontinued treatment due to vomiting assessed as related to ritonavir.
- The proportion of subjects with HIV-1 RNA less than 50 copies/mL at Week 48 was 71%.. The mean increase in CD4+ percentage from baseline was 4%. The mean change in CD4+ cell count from baseline was 187 × 106 cells/L.
- Study TMC114-C230
- Treatment-naïve pediatric subjects between the ages of 12 and less than 18 years and weighing at least 40 kg received the adult recommended dose of Darunavir/ritonavir 800/100 mg once daily plus background therapy consisting of at least two non-protease inhibitor antiretroviral drugs.
- The 12 randomized pediatric subjects had a median age of 14.4 years (range 12.6 to 17.3 years), and were 33.3% male, 58.3% Caucasian and 41.7% Black. The mean baseline plasma HIV-1 RNA was 4.72 log10 copies/mL, and the median baseline CD4+ cell count was 282 cells/mm3 (range: 204 to 515 cells/mm3). Overall, 41.7% of pediatric subjects had baseline plasma HIV-1 RNA ≥ 100,000 copies/mL.
- All subjects completed the 48 week treatment period.
- The proportion of subjects with HIV-1 RNA less than 50 copies/mL and less than 400 copies/mL was 83.3% and 91.7%, respectively. The mean increase in CD4+ cell count from baseline was 221 × 106 cells/L.
How Supplied
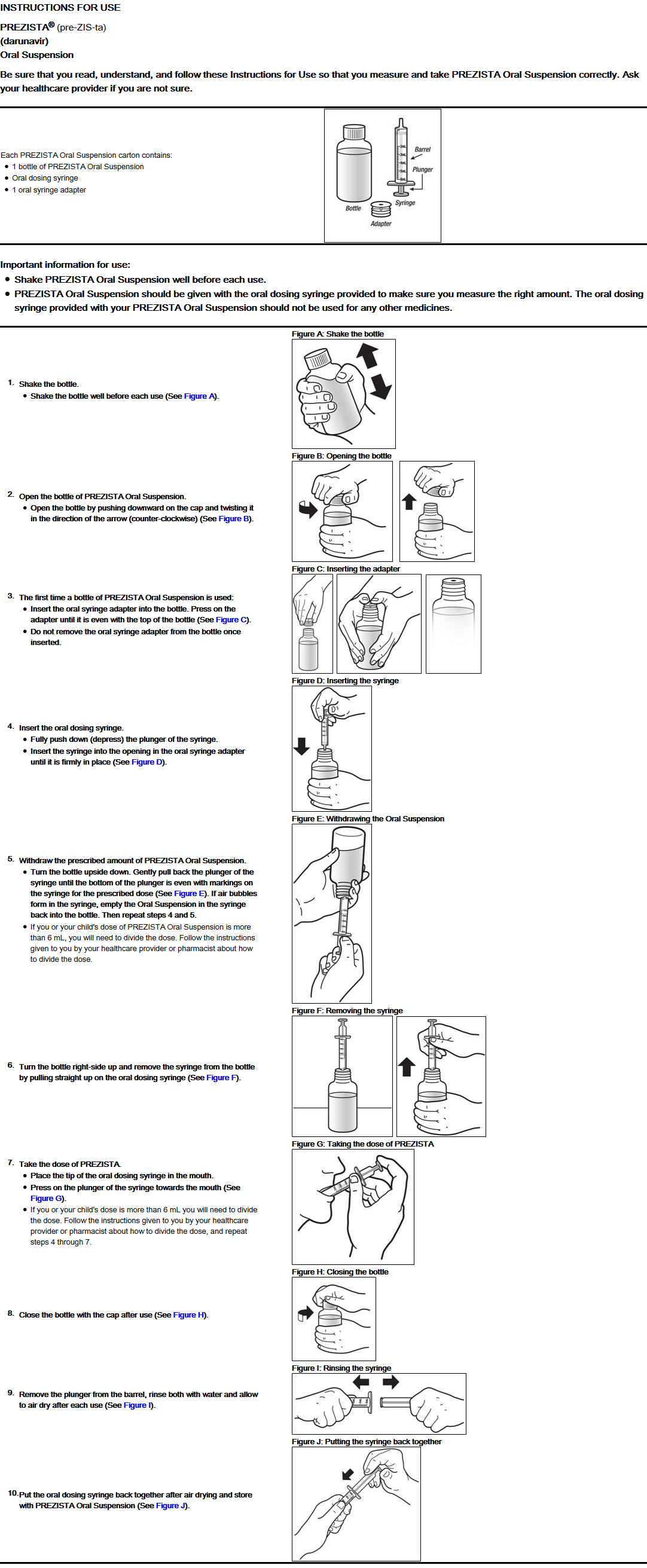
- Darunavir (darunavir) 100 mg/mL oral suspension is a white to off-white opaque liquid supplied in amber-colored multiple-dose bottles containing darunavir ethanolate equivalent to 100 mg of darunavir per mL packaged with a 6 mL oral dosing syringe with 0.2 mL gradations.
- Darunavir (darunavir) 75 mg tablets are supplied as white, caplet-shaped, film-coated tablets containing darunavir ethanolate equivalent to 75 mg of darunavir per tablet. Each tablet is debossed with "75" on one side and "TMC" on the other side.
- Darunavir (darunavir) 150 mg tablets are supplied as white, oval-shaped, film-coated tablets containing darunavir ethanolate equivalent to 150 mg of darunavir per tablet. Each tablet is debossed with "150" on one side and "TMC" on the other side.
- Darunavir (darunavir) 600 mg tablets are supplied as orange, oval-shaped, film-coated tablets containing darunavir ethanolate equivalent to 600 mg of darunavir per tablet. Each tablet is debossed with "600MG" on one side and "TMC" on the other side.
- Darunavir (darunavir) 800 mg tablets are supplied as dark red, oval-shaped, film-coated tablets containing darunavir ethanolate equivalent to 800 mg of darunavir per tablet. Each tablet is debossed with "800" on one side and "T" on the other side.
- Darunavir is packaged in bottles in the following configuration:
- 100 mg/mL oral suspension – 200 mL bottles (NDC 59676-565-01)
- 75 mg tablets—bottles of 480 (NDC 59676-563-01)
- 150 mg tablets—bottles of 240 (NDC 59676-564-01)
- 600 mg tablets—bottles of 60 (NDC 59676-562-01)
- 800 mg tablets—bottles of 30 (NDC 59676-566-30)
- Storage:
- Darunavir Oral Suspension
- Store at 25°C (77°F); with excursions permitted to 15°–30°C (59°–86°F).
- Do not refrigerate or freeze. Avoid exposure to excessive heat.
- Store in the original container.
- Shake well before each usage.
- Darunavir Tablets
- Store at 25°C (77°F); with excursions permitted to 15°–30°C (59°–86°F).
Storage
There is limited information regarding Darunavir Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Darunavir |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Darunavir |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Precautions with Alcohol
- Alcohol-Darunavir interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- PREZISTA®[1]
Look-Alike Drug Names
There is limited information regarding Darunavir Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "PREZISTA(darunavir) tablet, film coated Darunavir (darunavir) suspension [Janssen Products LP]". line feed character in
|title=at position 73 (help)
{{#subobject:
|Page Name=Darunavir
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Darunavir |Label Name=Darunavir27.png
}}
{{#subobject:
|Label Page=Darunavir |Label Name=Darunavir28.png
}}
{{#subobject:
|Label Page=Darunavir |Label Name=Darunavir29.png
}}
{{#subobject:
|Label Page=Darunavir |Label Name=Darunavir30.png
}}
{{#subobject:
|Label Page=Darunavir |Label Name=Darunavir31.png
}}
{{#subobject:
|Label Page=Darunavir |Label Name=Darunavir32.png
}}
{{#subobject:
|Label Page=Darunavir |Label Name=Darunavir33.png
}}