Emtricitabine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Gloria Picoy [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS AND POST TREATMENT EXACERBATION OF HEPATITIS B
See full prescribing information for complete Boxed Warning.
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination with other antiretrovirals.
Emtricitabine is not approved for the treatment of chronic hepatitis B virus (HBV) infection and the safety and efficacy of emtricitabine have not been established in patients coinfected with HBV and HIV-1. Severe acute exacerbations of hepatitis B have been reported in patients who have discontinued emtricitabine. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who are coinfected with HIV-1 and HBV and discontinue emtricitabine. If appropriate, initiation of anti-hepatitis B therapy may be warranted.
|
Overview
Emtricitabine is a nucleoside reverse transcriptase inhibitor that is FDA approved for the treatment of HIV-1 infection in combination with other antiretroviral agents. There is a Black Box Warning for this drug as shown here. Common adverse reactions include headache, diarrhea, nausea, fatigue, dizziness, depression, insomnia, abnormal dreams, rash, abdominal pain, asthenia, increased cough, rhinitis and skin hyperpigmentation (pediatric patients).
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Emtricitabine is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection.
- Dosage:
- Capsules: one 200 mg capsule administered once daily orally.
- Oral solution: 240 mg (24 mL) administered once daily orally.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Emtricitabine in adult patients.
Non–Guideline-Supported Use
- Dosage: 200 mg/day for 48 weeks [1]
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Emtricitabine is indicated in combination with other antiretroviral agents for the treatment of HIV-1 infection.
- Dosage in pediatric patients between 0–3 months of age
- Oral solution: 3 mg per kg administered once daily orally.
- Dosage in pediatric patients between 3 months through 17 years
- Oral solution: 6 mg per kg up to a maximum of 240 mg (24 mL) administered once daily orally.
- Capsules: for children weighing more than 33 kg who can swallow an intact capsule, one 200 mg capsule administered once daily orally.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Emtricitabine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Emtricitabine in pediatric patients.
Contraindications
- In patients with previously demonstrated hypersensitivity to any of the components of the products.
Warnings
|
WARNING: LACTIC ACIDOSIS/SEVERE HEPATOMEGALY WITH STEATOSIS AND POST TREATMENT EXACERBATION OF HEPATITIS B
See full prescribing information for complete Boxed Warning.
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination with other antiretrovirals.
Emtricitabine is not approved for the treatment of chronic hepatitis B virus (HBV) infection and the safety and efficacy of emtricitabine have not been established in patients coinfected with HBV and HIV-1. Severe acute exacerbations of hepatitis B have been reported in patients who have discontinued emtricitabine. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who are coinfected with HIV-1 and HBV and discontinue emtricitabine. If appropriate, initiation of anti-hepatitis B therapy may be warranted.
|
Lactic Acidosis/Severe Hepatomegaly with Steatosis
Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported with the use of nucleoside analogs alone or in combination, including emtricitabine and other antiretrovirals. A majority of these cases have been in women. Obesity and prolonged nucleoside exposure may be risk factors. Particular caution should be exercised when administering nucleoside analogs to any patient with known risk factors for liver diseases; however, cases have also been reported in patients with no known risk factors. Treatment with emtricitabine should be suspended in any patient who develops clinical or laboratory findings suggestive of lactic acidosis or pronounced hepatotoxicity (which may include hepatomegaly and steatosis even in the absence of marked transaminase elevations).
Patients Coinfected with HIV-1 and HBV
It is recommended that all patients with HIV-1 be tested for the presence of chronic Hepatitis B virus (HBV) before initiating antiretroviral therapy. Emtricitabine is not approved for the treatment of chronic HBV infection and the safety and efficacy of emtricitabine have not been established in patients coinfected with HBV and HIV-1. Severe acute exacerbations of Hepatitis B have been reported in patients after the discontinuation of emtricitabine. In some patients infected with HBV and treated with emtricitabine, the exacerbations of hepatitis B were associated with liver decompensation and liver failure. Hepatic function should be monitored closely with both clinical and laboratory follow-up for at least several months in patients who are coinfected with HIV-1 and HBV and discontinue emtricitabine. If appropriate, initiation of anti-Hepatitis B therapy may be warranted.
Coadministration with Related Products
Emtricitabine is a component of ATRIPLA (a fixed-dose combination of efavirenz, emtricitabine, and tenofovir disoproxil fumarate), COMPLERA (a fixed-dose combination of emtricitabine, rilpivirine, and tenofovir disoproxil fumarate), STRIBILD (a fixed-dose combination of elvitegravir, cobicistat, emtricitabine, and tenofovir disoproxil fumarate), and TRUVADA (a fixed-dose combination of emtricitabine and tenofovir disoproxil fumarate). Emtricitabine should not be coadministered with ATRIPLA, COMPLERA, STRIBILD, or TRUVADA. Due to similarities between emtricitabine and lamivudine, emtricitabine should not be coadministered with other drugs containing lamivudine, including Combivir (lamivudine/zidovudine), Epivir or Epivir-HBV (lamivudine), Epzicom (abacavir sulfate/lamivudine), or Trizivir (abacavir sulfate/lamivudine/zidovudine).
New Onset or Worsening Renal Impairment
Emtricitabine is principally eliminated by the kidney. Reduction of the dosage of emtricitabine is recommended for patients with impaired renal function.
Fat Redistribution
Redistribution/accumulation of body fat including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including emtricitabine. During the initial phase of combination antiretroviral treatment, patients whose immune system responds may develop an inflammatory response to indolent or residual opportunistic infections [such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia (PCP), or tuberculosis], which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, and Guillain-Barré syndrome) have also been reported to occur in the setting of immune reconstitution, however, the time to onset is more variable, and can occur many months after initiation of treatment.
Adverse Reactions
Clinical Trials Experience
Clinical Trials in Adult Subjects
More than 2,000 adult subjects with HIV-1 infection have been treated with emtricitabine alone or in combination with other antiretroviral agents for periods of 10 days to 200 weeks in clinical trials.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The most common adverse reactions (incidence greater than or equal to 10%, any severity) identified from any of the 3 large controlled clinical trials include headache, diarrhea, nausea, fatigue, dizziness, depression, insomnia, abnormal dreams, rash, abdominal pain, asthenia, increased cough, and rhinitis.
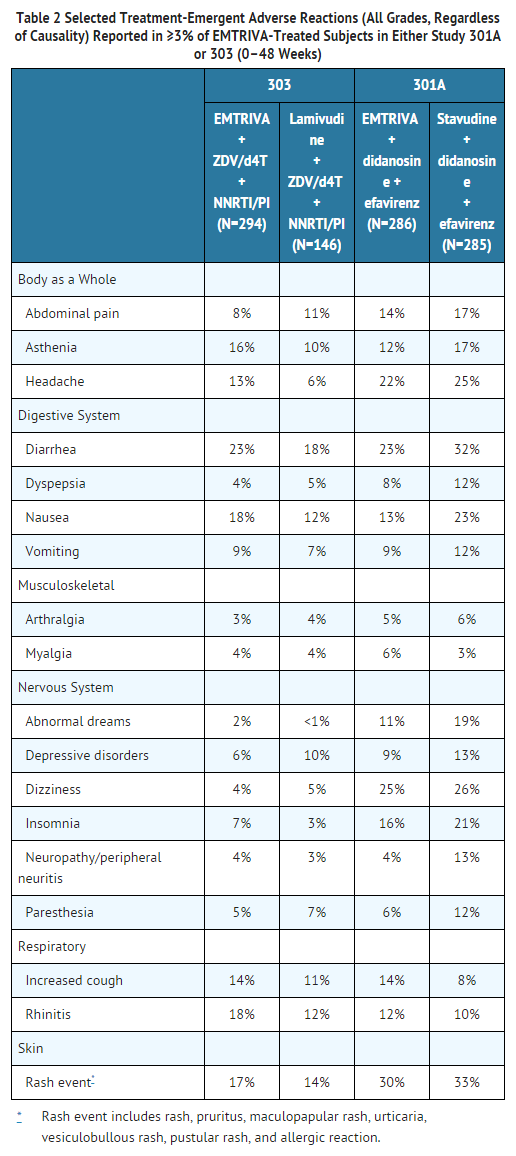
Studies 301A and 303 - Treatment Emergent Adverse Reactions: The most common adverse reactions that occurred in subjects receiving emtricitabine with other antiretroviral agents in clinical trials 301A and 303 were headache, diarrhea, nausea, and rash, which were generally of mild to moderate severity. Approximately 1% of subjects discontinued participation in the clinical trials due to these events. All adverse reactions were reported with similar frequency in emtricitabine and control treatment groups with the exception of skin discoloration which was reported with higher frequency in the emtricitabine treated group.
Skin discoloration, manifested by hyperpigmentation on the palms and/or soles was generally mild and asymptomatic. The mechanism and clinical significance are unknown.
A summary of emtricitabine treatment-emergent clinical adverse reactions in Studies 301A and 303 is provided in Table 2.
Studies 301A and 303 - Laboratory Abnormalities: Laboratory abnormalities in these trials occurred with similar frequency in the emtricitabine and comparator groups. A summary of Grades 3–4 laboratory abnormalities is provided in Table 3 below.

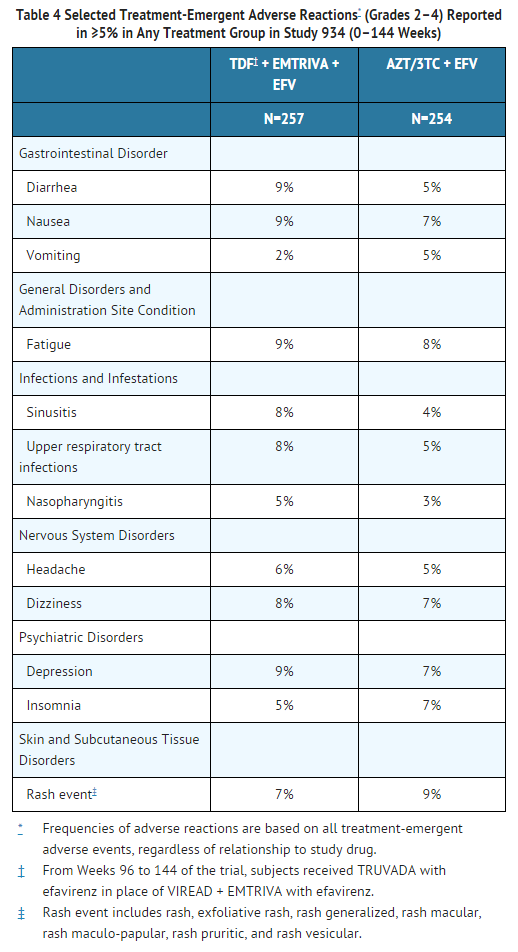
Study 934 - Treatment Emergent Adverse Reactions: In Study 934, 511 antiretroviral-naïve subjects received either VIREAD® + emtricitabine administered in combination with efavirenz (N=257) or zidovudine/lamivudine administered in combination with efavirenz (N=254). Adverse reactions observed in this trial were generally consistent with those seen in previous trials in treatment-experienced or treatment-naïve subjects (Table 4).
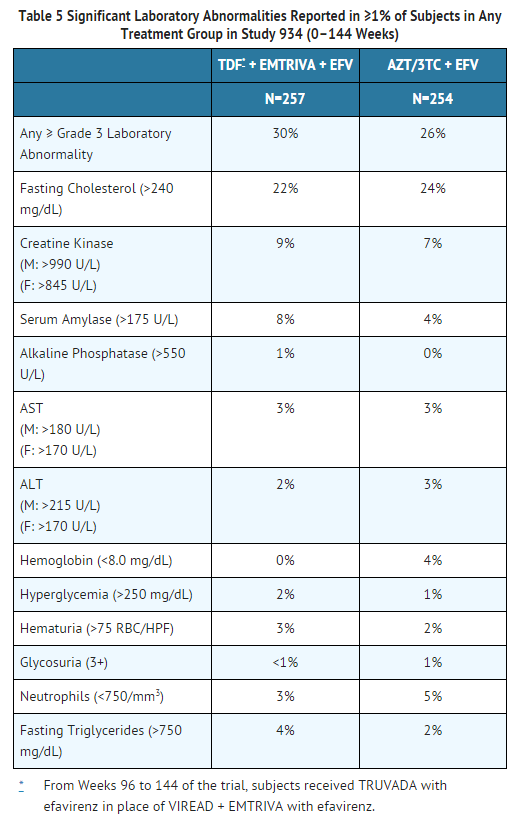
Study 934 – Laboratory Abnormalities: Significant laboratory abnormalities observed in this trial are shown in Table 5.
Clinical Trials in Pediatric Subjects
Assessment of adverse reactions is based on data from Study 203, an open label, uncontrolled trial of 116 HIV-1-infected pediatric subjects who received emtricitabine through 48 weeks. The adverse reaction profile in pediatric subjects was generally comparable to that observed in clinical trials of emtricitabine in adult subjects. Hyperpigmentation was more frequent in children. Additional adverse reactions identified from this trial include anemia.
Selected treatment-emergent adverse events, regardless of causality, reported in subjects during 48 weeks of treatment were the following: infection (44%), hyperpigmentation (32%), increased cough (28%), vomiting (23%), otitis media (23%), rash (21%), rhinitis (20%), diarrhea (20%), fever (18%), pneumonia (15%), gastroenteritis (11%), abdominal pain (10%), and anemia (7%). Treatment-emergent grades 3–4 laboratory abnormalities were experienced by 9% of pediatric subjects, including elevated amylase (>2.0 × ULN) (n=4), decreased neutrophils (<750/mm3) (n=3), elevated ALT (>5 × ULN) (n=2), elevated CPK (>4 × ULN) (n=2) and one subject each with elevated bilirubin (>3.0 × ULN), elevated GGT (>10 × ULN), elevated lipase (>2.5 × ULN), decreased hemoglobin (<7 g/dL), and decreased glucose (<40 mg/dL).
Postmarketing Experience
There is limited information regarding Emtricitabine Postmarketing Experience in the drug label.
Drug Interactions
The potential for drug interactions with emtricitabine has been studied in combination with zidovudine, indinavir, stavudine, famciclovir, and tenofovir disoproxil fumarate. There were no clinically significant drug interactions for any of these drugs. Drug interactions trials are described elsewhere in the labeling.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): B
The incidence of fetal variations and malformations was not increased in embryofetal toxicity studies performed with emtricitabine in mice at exposures (AUC) approximately 60-fold higher and in rabbits at approximately 120-fold higher than human exposures at the recommended daily dose. There are, however, no adequate and well-controlled trials in pregnant women. Because animal reproduction studies are not always predictive of human response, emtricitabine should be used during pregnancy only if clearly needed.
Pregnancy Category (AUS): B1
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Emtricitabine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Emtricitabine during labor and delivery.
Nursing Mothers
The Centers for Disease Control and Prevention recommend that HIV-1-infected mothers not breast-feed their infants to avoid risking postnatal transmission of HIV-1. Samples of breast milk obtained from five HIV-1 infected mothers show that emtricitabine is secreted in human milk. Breastfeeding infants whose mothers are being treated with emtricitabine may be at risk for developing viral resistance to emtricitabine. Other emtricitabine-associated risks in infants breastfed by mothers being treated with emtricitabine are unknown. Because of both the potential for HIV-1 transmission and the potential for serious adverse reactions in nursing infants, mothers should be instructed not to breast-feed if they are receiving emtricitabine.
Pediatric Use
The safety and efficacy of emtricitabine in patients between 3 months and 21 years of age is supported by data from three open-label, non-randomized clinical trials in which emtricitabine was administered to 169 HIV-1 infected treatment-naive and experienced (defined as virologically suppressed on a lamivudine containing regimen for which emtricitabine was substituted for lamivudine) subjects.
The pharmacokinetics of emtricitabine were studied in 20 neonates born to HIV-1-positive mothers. All neonates were HIV-1 negative at the end of the trial; the efficacy of emtricitabine in preventing or treating HIV-1 could not be determined.
Geriatic Use
Clinical trials of emtricitabine did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects. In general, dose selection for the elderly patient should be cautious, keeping in mind the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Gender
The pharmacokinetics of emtricitabine were similar in adult male and female subjects.
Race
No pharmacokinetic differences due to race have been identified.
Renal Impairment
It is recommended that the dose or dosing interval for emtricitabine be modified in patients with creatinine clearance less than 50 mL/min or in patients who require dialysis.
Hepatic Impairment
The pharmacokinetics of emtricitabine have not been studied in subjects with hepatic impairment, however, emtricitabine is not metabolized by liver enzymes, so the impact of liver impairment should be limited.
Females of Reproductive Potential and Males
Emtricitabine did not affect fertility in male rats at approximately 140-fold or in male and female mice at approximately 60-fold higher exposures (AUC) than in humans given the recommended 200 mg daily dose. Fertility was normal in the offspring of mice exposed daily from before birth (in utero) through sexual maturity at daily exposures (AUC) of approximately 60-fold higher than human exposures at the recommended 200 mg daily dose.
Immunocompromised Patients
There is no FDA guidance one the use of Emtricitabine in patients who are immunocompromised.
Administration and Monitoring
Administration
Oral
Monitoring
There is limited information regarding Emtricitabine Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Emtricitabine and IV administrations.
Overdosage
There is no known antidote for emtricitabine. Limited clinical experience is available at doses higher than the therapeutic dose of emtricitabine. In one clinical pharmacology trial single doses of emtricitabine 1200 mg were administered to 11 subjects. No severe adverse reactions were reported.
The effects of higher doses are not known. If overdose occurs the patient should be monitored for signs of toxicity, and standard supportive treatment applied as necessary.
Hemodialysis treatment removes approximately 30% of the emtricitabine dose over a 3-hour dialysis period starting within 1.5 hours of emtricitabine dosing (blood flow rate of 400 mL/min and a dialysate flow rate of 600 mL/min). It is not known whether emtricitabine can be removed by peritoneal dialysis.
Pharmacology
Mechanism of Action
Emtricitabine, a synthetic nucleoside analog of cytidine, is phosphorylated by cellular enzymes to form emtricitabine 5'-triphosphate. Emtricitabine 5'-triphosphate inhibits the activity of the HIV-1 reverse transcriptase by competing with the natural substrate deoxycytidine 5'-triphosphate and by being incorporated into nascent viral DNA which results in chain termination. Emtricitabine 5'-triphosphate is a weak inhibitor of mammalian DNA polymerase α, β, ε, and mitochondrial DNA polymerase γ.
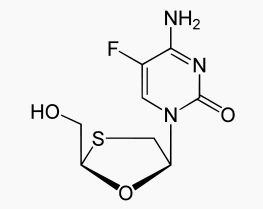
Structure
It has a molecular formula of C8H10FN3O3S and a molecular weight of 247.24. It has the following structural formula:
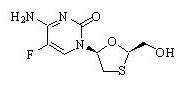
Pharmacodynamics
There is limited information regarding Emtricitabine Pharmacodynamics in the drug label.
Pharmacokinetics
The pharmacokinetics of emtricitabine were evaluated in healthy subjects and HIV-1-infected subjects. Emtricitabine pharmacokinetics are similar between these populations.
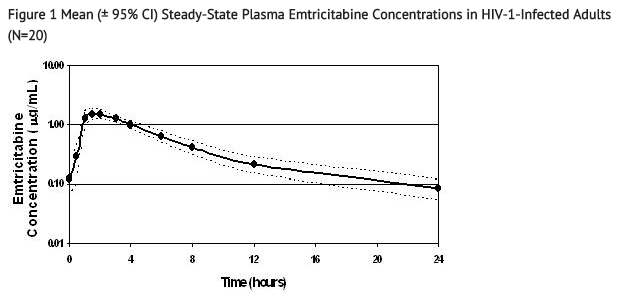
Figure 1 shows the mean steady-state plasma emtricitabine concentration-time profile in 20 HIV-1-infected subjects receiving emtricitabine capsules.
Absorption
Emtricitabine is rapidly and extensively absorbed following oral administration with peak plasma concentrations occurring at 1–2 hours post-dose. Following multiple dose oral administration of emtricitabine capsules to 20 HIV-1-infected subjects, the (mean ± SD) steady-state plasma emtricitabine peak concentration (Cmax) was 1.8 ± 0.7 µg/mL and the area-under the plasma concentration-time curve over a 24-hour dosing interval (AUC) was 10.0 ± 3.1 µg∙hr/mL. The mean steady state plasma trough concentration at 24 hours post-dose was 0.09 µg/mL. The mean absolute bioavailability of emtricitabine capsules was 93% while the mean absolute bioavailability of emtricitabine oral solution was 75%. The relative bioavailability of emtricitabine oral solution was approximately 80% of emtricitabine capsules.
The multiple dose pharmacokinetics of emtricitabine are dose proportional over a dose range of 25–200 mg.
Distribution
In vitro binding of emtricitabine to human plasma proteins was less than 4% and independent of concentration over the range of 0.02–200 µg/mL. At peak plasma concentration, the mean plasma to blood drug concentration ratio was ~1.0 and the mean semen to plasma drug concentration ratio was ~4.0.
Metabolism
In vitro studies indicate that emtricitabine is not an inhibitor of human CYP450 enzymes. Following administration of 14C-emtricitabine, complete recovery of the dose was achieved in urine (~86%) and feces (~14%). Thirteen percent (13%) of the dose was recovered in urine as three putative metabolites. The biotransformation of emtricitabine includes oxidation of the thiol moiety to form the 3'-sulfoxide diastereomers (~9% of dose) and conjugation with glucuronic acid to form 2'-O-glucuronide (~4% of dose). No other metabolites were identifiable.
Elimination
The plasma emtricitabine half-life is approximately 10 hours. The renal clearance of emtricitabine is greater than the estimated creatinine clearance, suggesting elimination by both glomerular filtration and active tubular secretion. There may be competition for elimination with other compounds that are also renally eliminated.
Nonclinical Toxicology
Antiviral Activity
The antiviral activity in cell culture of emtricitabine against laboratory and clinical isolates of HIV-1 was assessed in lymphoblastoid cell lines, the MAGI-CCR5 cell line, and peripheral blood mononuclear cells. The 50% effective concentration (EC50) value for emtricitabine was in the range of 0.0013–0.64 µM (0.0003–0.158 µg/mL). In drug combination trials of emtricitabine with nucleoside reverse transcriptase inhibitors (abacavir, lamivudine, stavudine, tenofovir, zalcitabine, zidovudine), non-nucleoside reverse transcriptase inhibitors (delavirdine, efavirenz, nevirapine), and protease inhibitors (amprenavir, nelfinavir, ritonavir, saquinavir), additive to synergistic effects were observed. Emtricitabine displayed antiviral activity in cell culture against HIV-1 clades A, B, C, D, E, F, and G (EC50 values ranged from 0.007–0.075 µM) and showed strain specific activity against HIV-2 (EC50 values ranged from 0.007–1.5 µM).
The in vivo activity of emtricitabine was evaluated in two clinical trials in which 101 subjects were administered 25–400 mg a day of emtricitabine as monotherapy for 10–14 days. A dose-related antiviral effect was observed, with a median decrease from baseline in plasma HIV-1 RNA of 1.3 log10 at a dose of 25 mg once daily and 1.7 log10 to 1.9 log10 at a dose of 200 mg once daily or twice daily.
Resistance
Emtricitabine-resistant isolates of HIV-1 have been selected in cell culture and in vivo. Genotypic analysis of these isolates showed that the reduced susceptibility to emtricitabine was associated with a substitution in the HIV-1 reverse transcriptase gene at codon 184 which resulted in an amino acid substitution of methionine by valine or isoleucine (M184V/I).
Emtricitabine-resistant isolates of HIV-1 have been recovered from some subjects treated with emtricitabine alone or in combination with other antiretroviral agents. In a clinical trial of treatment-naive subjects treated with emtricitabine, didanosine, and efavirenz, viral isolates from 37.5% of subjects with virologic failure showed reduced susceptibility to emtricitabine. Genotypic analysis of these isolates showed that the resistance was due to M184V/I substitutions in the HIV-1 reverse transcriptase gene.
In a clinical trial of treatment-naive subjects treated with either emtricitabine, VIREAD, and efavirenz or zidovudine/lamivudine and efavirenz, resistance analysis was performed on HIV-1 isolates from all confirmed virologic failure subjects with greater than 400 copies/mL of HIV-1 RNA at Week 144 or early discontinuation. Development of efavirenz resistance-associated substitutions occurred most frequently and was similar between the treatment arms. The M184V amino acid substitution, associated with resistance to emtricitabine and lamivudine, was observed in 2/19 analyzed subject isolates in the emtricitabine + VIREAD group and in 10/29 analyzed subject isolates in the lamivudine/zidovudine group. Through 144 weeks of Study 934, no subjects have developed a detectable K65R substitution in their HIV-1 as analyzed through standard genotypic analysis.
Cross Resistance
Cross-resistance among certain nucleoside analog reverse transcriptase inhibitors has been recognized. Emtricitabine-resistant isolates (M184V/I) were cross-resistant to lamivudine and zalcitabine but retained sensitivity in cell culture to didanosine, stavudine, tenofovir, zidovudine, and NNRTIs (delavirdine, efavirenz, and nevirapine). HIV-1 isolates containing the K65R substitution, selected in vivo by abacavir, didanosine, tenofovir, and zalcitabine, demonstrated reduced susceptibility to inhibition by emtricitabine. Viruses harboring substitutions conferring reduced susceptibility to stavudine and zidovudine (M41L, D67N, K70R, L210W, T215Y/F, K219Q/E) or didanosine (L74V) remained sensitive to emtricitabine. HIV-1 containing the K103N substitution associated with resistance to NNRTIs was susceptible to emtricitabine.
Carcinogenesis and Mutagenesis
In long-term oral carcinogenicity studies of emtricitabine, no drug-related increases in tumor incidence were found in mice at doses up to 750 mg/kg/day (26 times the human systemic exposure at the therapeutic dose of 200 mg/day) or in rats at doses up to 600 mg/kg/day (31 times the human systemic exposure at the therapeutic dose).
Emtricitabine was not genotoxic in the reverse mutation bacterial test (Ames test), mouse lymphoma or mouse micronucleus assays.
Clinical Studies
Treatment-Naive Adult Patients
Study 934
Data through 144 weeks are reported for Study 934, a randomized, open-label, active-controlled multicenter clinical trial comparing emtricitabine + tenofovir disoproxil fumarate (tenofovir DF) administered in combination with efavirenz versus zidovudine/lamivudine fixed-dose combination administered in combination with efavirenz in 511 antiretroviral-naive subjects. From Weeks 96 to 144 of the trial, subjects received emtricitabine/tenofovir DF fixed-dose combination with efavirenz in place of emtricitabine + tenofovir DF with efavirenz. Subjects had a mean age of 38 years (range 18–80), 86% were male, 59% were Caucasian and 23% were Black. The mean baseline CD4+ cell count was 245 cells/mm3 (range 2–1191) and median baseline plasma HIV-1 RNA was 5.01 log10 copies/mL (range 3.56–6.54). Subjects were stratified by baseline CD4+ cell count (< or ≥200 cells/mm3); 41% had CD4+ cell counts <200 cells/mm3 and 51% of subjects had baseline viral loads >100,000 copies/mL. Treatment outcomes through 48 and 144 weeks for those subjects who did not have efavirenz resistance at baseline are presented in Table 10.
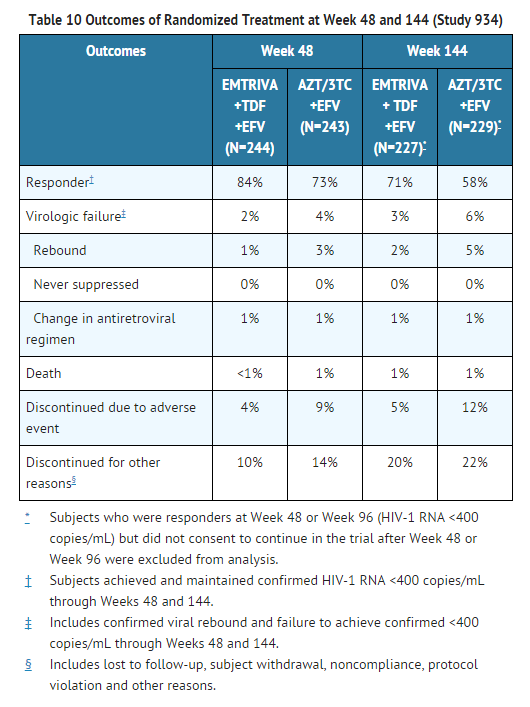
Through Week 48, 84% and 73% of subjects in the emtricitabine + tenofovir DF group and the zidovudine/lamivudine group, respectively, achieved and maintained HIV-1 RNA <400 copies/mL (71% and 58% through Week 144). The difference in the proportion of subjects who achieved and maintained HIV-1 RNA <400 copies/mL through 48 weeks largely results from the higher number of discontinuations due to adverse events and other reasons in the zidovudine/lamivudine group in this open-label trial. In addition, 80% and 70% of subjects in the emtricitabine + tenofovir DF group and the zidovudine/lamivudine group, respectively, achieved and maintained HIV-1 RNA <50 copies/mL through Week 48 (64% and 56% through Week 144). The mean increase from baseline in CD4+ cell count was 190 cells/mm3 in the emtricitabine + tenofovir DF group and 158 cells/mm3 in the zidovudine/lamivudine group at Week 48 (312 and 271 cells/mm3 at Week 144).
Through 48 weeks, 7 subjects in the emtricitabine + tenofovir DF group and 5 subjects in the zidovudine/lamivudine group experienced a new CDC Class C event (10 and 6 subjects through 144 weeks).
Study 301A
Study 301A was a 48 week double-blind, active-controlled multicenter clinical trial comparing emtricitabine (200 mg once daily) administered in combination with didanosine and efavirenz versus stavudine, didanosine and efavirenz in 571 antiretroviral naive adult subjects. Subjects had a mean age of 36 years (range 18–69), 85% were male, 52% Caucasian, 16% African-American and 26% Hispanic. Subjects had a mean baseline CD4+ cell count of 318 cells/mm3 (range 5–1317) and a median baseline plasma HIV-1 RNA of 4.9 log10 copies/mL (range 2.6–7.0). Thirty-eight percent of subjects had baseline viral loads >100,000 copies/mL and 31% had CD4+ cell counts <200 cells/mL. Treatment outcomes are presented in Table 11 below.

The mean increase from baseline in CD4+ cell count was 168 cells/mm3 for the emtricitabine arm and 134 cells/mm3 for the stavudine arm.
Through 48 weeks in the emtricitabine group, 5 subjects (1.7%) experienced a new CDC Class C event, compared to 7 subjects (2.5%) in the stavudine group.
Treatment-Experienced Adult Patients
Study 303
Study 303 was a 48 week, open-label, active-controlled multicenter clinical trial comparing emtricitabine (200 mg once daily) to lamivudine, in combination with stavudine or zidovudine and a protease inhibitor or NNRTI in 440 adult subjects who were on a lamivudine-containing triple-antiretroviral drug regimen for at least 12 weeks prior to trial entry and had HIV-1 RNA ≤400 copies/mL.
Subjects were randomized 1:2 to continue therapy with lamivudine (150 mg twice daily) or to switch to emtricitabine (200 mg once daily). All subjects were maintained on their stable background regimen. Subjects had a mean age of 42 years (range 22–80), 86% were male, 64% Caucasian, 21% African-American and 13% Hispanic. Subjects had a mean baseline CD4+ cell count of 527 cells/mm3 (range 37–1909), and a median baseline plasma HIV-1 RNA of 1.7 log10 copies/mL (range 1.7–4.0).
The median duration of prior antiretroviral therapy was 27.6 months. Treatment outcomes are presented in Table 12 below.

The mean increase from baseline in CD4+ cell count was 29 cells/mm3 for the emtricitabine arm and 61 cells/mm3 for the lamivudine arm.
Through 48 weeks, in the emtricitabine group 2 subjects (0.7%) experienced a new CDC Class C event, compared to 2 subjects (1.4%) in the lamivudine group.
Pediatric Patients
In three open-label, non-randomized clinical trials, emtricitabine was administered to 169 HIV-1 infected treatment-naive and experienced (defined as virologically suppressed on a lamivudine containing regimen for which emtricitabine was substituted for lamivudine) subjects between 3 months and 21 years of age. Subjects received once-daily emtricitabine oral solution (6 mg/kg to a maximum of 240 mg/day) or emtricitabine capsules (a single 200 mg capsule once daily) in combination with at least two other antiretroviral agents.
Subjects had a mean age of 7.9 years (range 0.3–21), 49% were male, 15% Caucasian, 61% Black and 24% Hispanic. Subjects had a median baseline HIV-1 RNA of 4.6 log10 copies/mL (range 1.7–6.4) and a mean baseline CD4+ cell count of 745 cells/mm3 (range 2–2650). Through 48 weeks of therapy, the overall proportion of subjects who achieved and sustained an HIV-1 RNA <400 copies/mL was 86%, and <50 copies/mL was 73%. The mean increase from baseline in CD4+ cell count was 232 cells/mm3 (-945, +1512). The adverse reaction profile observed during these clinical trials was similar to that of adult subjects, with the exception of the occurrence of anemia and higher frequency of hyperpigmentation in children.
The pharmacokinetics of emtricitabine were studied in 20 neonates born to HIV-1-positive mothers. Each mother received prenatal and intrapartum combination antiretroviral therapy. Neonates received up to 6 weeks of zidovudine prophylactically after birth. The neonates were administered two short courses of emtricitabine oral solution (each 3 mg/kg once daily × 4 days) during the first 3 months of life. Emtricitabine exposures in neonates were similar to the exposures achieved in subjects ages 3 months to 17 years. During the two short dosing periods on emtricitabine there were no safety issues identified in the treated neonates. All neonates were HIV-1 negative at the end of the trial; the efficacy of emtricitabine in preventing or treating HIV-1 could not be determined.
How Supplied
Emtricitabine capsules 200 mg
- Bottles of 30 capsules (NDC 61958–0601–1)
Emtricitabine oral solution 10 mg/mL
- Bottles of 170 mL (NDC 61958–0602–1)
Storage
- Capsules: Store at 25 °C (77 °F)
- Oral solution: Store refrigerated, 2°C–8°C (36 °F–46 °F)
Images
Drug Images
{{#ask: Page Name::Emtricitabine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Emtricitabine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Patients should be advised that:
- Emtricitabine is not a cure for HIV-1 infection and patients may continue to experience illnesses associated with HIV-1 infection, including opportunistic infections. Patients should remain under the care of a physician when using emtricitabine.
- Patients should avoid doing things that can spread HIV to others.
- Do not share needles or other injection equipment.
- Do not share personal items that can have blood or body fluids on them, like toothbrushes and razor blades.
- Do not have any kind of sex without protection: Always practice safer sex by using a latex or polyurethane condom to lower the chance of sexual contact with semen, vaginal secretions, or blood.
- Do not breastfeed: emtricitabine is secreted in breast milk. Mothers with HIV-1 should not breastfeed because HIV-1 can be passed to the baby in the breast milk.
- The long term effects of emtricitabine are unknown.
- Emtricitabine capsules and oral solution are for oral ingestion only.
- It is important to take emtricitabine with combination therapy on a regular dosing schedule to avoid missing doses.
- Lactic acidosis and severe hepatomegaly with steatosis, including fatal cases, have been reported. Treatment with emtricitabine should be suspended in any patient who develops clinical symptoms suggestive of lactic acidosis or pronounced hepatotoxicity (including nausea, vomiting, unusual or unexpected stomach discomfort, and weakness).
- Patients with HIV-1 should be tested for Hepatitis B virus (HBV) before initiating antiretroviral therapy.
- Severe acute exacerbations of Hepatitis B have been reported in patients who are coinfected with HBV and HIV-1 and have discontinued emtricitabine.
- Emtricitabine should not be coadministered with ATRIPLA, COMPLERA, STRIBILD, or TRUVADA; or with other drugs containing lamivudine, including Combivir (lamivudine/zidovudine), Epivir or Epivir-HBV (lamivudine), Epzicom (abacavir sulfate/lamivudine), or Trizivir (abacavir sulfate/lamivudine/zidovudine).
- Dose or dosing interval of emtricitabine may need adjustment in patients with renal impairment.
Precautions with Alcohol
Alcohol-Emtricitabine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Emtriva [2]
Look-Alike Drug Names
There is limited information regarding Emtricitabine Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Seng Gee Lim, Tay Meng Ng, Nelson Kung, Zahary Krastev, Miroslava Volfova, Petr Husa, Samuel S. Lee, Sing Chan, Mitchell L. Shiffman, Mary Kay Washington, Amy Rigney, Jane Anderson, Elsa Mondou, Andrea Snow, Jeff Sorbel, Richard Guan & Franck Rousseau (2006). "A double-blind placebo-controlled study of emtricitabine in chronic hepatitis B". Archives of internal medicine. 166 (1): 49–56. doi:10.1001/archinte.166.1.49. PMID 16401810. Unknown parameter
|month=ignored (help) - ↑ "FDA LABEL: EMTRIVA- emtricitabine capsule.EMTRIVA- emtricitabine solution".
{{#subobject:
|Label Page=Emtricitabine |Label Name=Emtricitabine capsules 200 mg.png
}}
{{#subobject:
|Label Page=Emtricitabine |Label Name=Emtricitabine oral solution 10 mg-ml.png
}}