Dilated cardiomyopathy
| Dilated cardiomyopathy | |
 | |
|---|---|
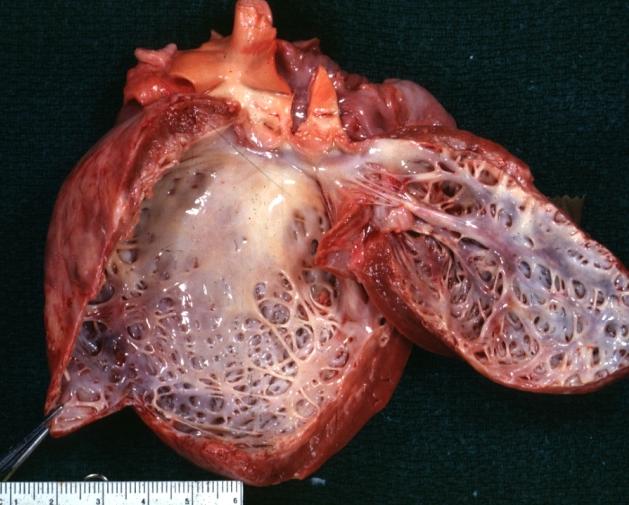
| Dilated Cardiomyopathy: Gross dilated left ventricle with marked endocardial sclerosis Image courtesy of Professor Peter Anderson DVM PhD and published with permission © PEIR, University of Alabama at Birmingham, Department of Pathology | |
| ICD-10 | I42.0 |
| ICD-9 | 425.4 |
| OMIM | 212110 |
| DiseasesDB | 3066 |
| MedlinePlus | 000168 |
| MeSH | D002311 |
|
Dilated cardiomyopathy Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Dilated cardiomyopathy On the Web |
|
American Roentgen Ray Society Images of Dilated cardiomyopathy |
|
Risk calculators and risk factors for Dilated cardiomyopathy |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor-in-Chief: Sachin Shah, M.D.
Synonyms and keywords: DCM; congestive cardiomyopathy
Overview
Causes
Clinical Presentation
Diagnosis
The diagnosis of dilated cardiomyopathy is based on clinical presentation and imaging findings. The most common imaging modality used to diagnose dilated cardiomyopathy is 2D-echocardiography. Echocardiographic findings of dilated cardiomyopathy include dilation of the left ventricle; however, may include dilation of all 4 cardiac chambers, LV (left ventricular) wall thickness usually is normal but given the dilation the LV mass is increased. In addition there is a global reduciton in systolic function. Occasionally there may also be focal wall motion abnormalities even in patients without flow limiting coronary artery disease.[1]
The diagnosis requires a dilated left ventricle and low ejection fraction.
In terms of determining the etiology a careful history is most instrumental. If the patient has CAD (coronary artery disease) risk factors, known CAD, or angina then a workup for CAD should be undertaken with coronary angiography. A viral prodrome such as viral URI or viral gastroenteritis may make viral myocarditis as a more likely cause. If the patient was exposed to chemotherapy such as anthracyclines then this would be the likely cause. Patients at risk for HIV should undergo testing as HIV can cause a dilated cardiomyopathy. Peripartum cardiomyopathy most often occurs 1 month prior to expected delivery or 5 months after delivery, so recent childbirth is important information. Often by 8 months gestational age pregnancy is physically apparent but it is important to rule out pregnancy in women of childbearing age with dilated cardiomyopathy. Screening questions regarding cocaine or alcohol abuse or other toxin exposure (such as cobalt) should be addressed.
A review of systems is also helpful in regards to connective tissue disease associated dilated cardiomyopathy (which can be related to SLE (systemic lupus erythematosis), rheumatoid arthritis, sarcoidosis, scleroderma, as well as other connective tissue diseases).
A family history also has a great importance in the diagnosis of dilated cardiomyopathy. It has been suggested that a portion of those patients labeled as "idiopathic" may have a familial form of the disease. The prevalence of this in the population of patients with dilated cardiomyopathy has been estimated as high as 25%.[2] The majority of these are thought to be related to autosomal dominant transmission, the remaining are thought to be transimtted in an autosomal recessive and X-linked fashion.[3] Mitochondrial inheritance of the disease has also been identified.[4]
Pericardial effusion may accompany myocarditis but this finding is not specific. Cardiac MRI as discussed above may be helpful in diagnosing myocarditis. Endomyocardial biopsy as discussed above has low sensitivy and the findings are also notoriously non-specific. The findings on biopsy usually involve findings of inflammation and specific pathogens are unlikely to be identified. There may be an increased yield to using MRI to target endomyocardial biopsy as described above. Viral titiers (serologies) are often unhelpful and not routinely ordered in clinical practice.
Prognosis
Treatment
Advanced Therapies
In patients with end stage heart failure or hemodynamic compromise vasopressor agents may be helpful. In patients with hemodynamic compromise medications such as dobutamine or dopamine may provide blood pressure support necessary. In addition parental agents such as milrinone (a phosphodiesterase inhibitor- somewhat specific for cardiac and vascular tissue) may be helpful in the short term or for end stage patients. There is no clear survival benefit and most trials with IV phosphodiesterase inhibitors are small trials of end stage heart failure patients. Oral milrinone has been studied and has been shown to increase mortality (as seen in the PROMISE trial).
In patients who have severe heart failure and are refractory to medical therapy cardiac transplantation may be an option (based upon patient characteristics such as age and comorbidities as well as disease characterisitics such as the etiology of dilated cardiomyopathy). As a bridge to cardiac transplantation multiple mechanical devices have been used. The most durable device is the LVAD (left ventricular assist device), certain patient populations not suitable for cardiac transplantation may also undergo LVAD placement by a cardiac surgeon as a "destination LVAD." This may be considered a palliative measure for end-stage heart failure patients. Other devices which may be more limited in the duration of use include the aortic balloon pump, Impella, and the PVAD (peripheral ventricular assist device) one of which is the Tandem Heart.
Implantable cardiac defibrillators in dilated cardiomyopathy
Implantable cardiac defibrillators (ICDs) have also been extensively studied in systolic heart failure. A survival benefit has also been shown in select patient populations. ICDs are implanted in patients with systolic heart failure for primary prevention of sudden cardiac death. The AHA/ACC (American Heart Association/American College of Cardiology) class I indications for ICDs for primary prevention are: 1. EF < 35% due to prior MI, 40 days post MI, NYHA class II or III. 2. EF < 35% NYHA class II or III non ischemic dilated cardiomyopathy. 3. EF < 30%, prior MI, 40 days post MI, NYHA I. 4. EF < 40%, prior MI, NSVT and inducible VT/VF at EP study.
There are four major studies that have helped shape these indication for an ICD. The MADIT I evaluated NYHA class I to III heart failure patients with an EF < or = to 35%, with NSVT and inducible VT or VF on EP study and randomized 196 patients to ICD versus conventional medical therapy. The patients who received ICDs had improved survival at 5 years. 60% of patients received shocks by 2 years.[5] The findings from this study are encompassed in the remaining three studies.
Madit II randomized 1232 patients who had a prior MI > 1 month prior to enrollment, an EF < or = to 30% to ICD or medical therapy and after a follow up of an average of 20 months there was a survival advantage in the ICD group. The number needed to treat was 18 patients. [6]
MUSTT randomized 704 patients with an EF < or = to 40%, NSVT and inducible VT/VF at EP study to arrhythmia therapy (medications and ICDs) or no antiarrhythmia. At 5 years there was a 27 risk reduction in mortality in the antiarrhythmia group. The relative risk of mortality when comparing ICD vs. no ICD was 0.24. There was no difference in mortality in the antiarrhythmic medication vs. no antiarrthymic medication subgroups.[7]
SCD-HeFT randomized 2521 patients with class II or III CHF and EF < or = to 35% (ischemic or non ischemic) to three arms: 1. conventional therapy, 2. conventional therapy plus amiodarone, and 3. conventional therapy plus an ICD. After a median follow up of 46 months there was a relative risk of death of 0.77 in the ICD arm. Findings were similar in the ischemic and non-ischemic subgroups.[8]
NYHA class IV heart failure is a contraindication to ICD placement.
Related chapters
Resources
References
- ↑ Mayo Clinic Cardiology. Concise Textbook. Murphy, Joseph G; Lloyd, Margaret A. Mayo Clinic Scientific Press. 2007.
- ↑ Ross J Jr. Dilated cardiomyopathy: concepts derived from gene deficient and transgenic animal models. Circ J. 2002;66:219-24. PMID 11922267
- ↑ Mestroni L; Rocco C; et al. Familial dilated cardiomyopathy: evidence for genetic and phenotypic heterogeneity. Heart Muscle Disease Study Group. J Am Coll Cardiol 1999 Jul;34(1):181-90.
- ↑ Schonberger J, Seidman CE. Many roads lead to a broken heart: the genetics of dilated cardiomyopathy. Am J Hum Genet. 2001;69:249-60. Epub 2001 Jul 6. PMID 11443548
- ↑ N Engl J Med. 1996. Dec 355(26)
- ↑ N Engl J Med. 2002. Mar 346(12)
- ↑ N Engl J Med. 1999 Dec 341:1882-1890.
- ↑ N Engl J Med. 2005. Jan 352(3).