Tipranavir contraindications
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ahmed Zaghw, M.D. [2]
Contraindications
Hepatic Impairment
APTIVUS is contraindicated in patients with moderate or severe (Child-Pugh Class B or C, respectively) hepatic impairment.
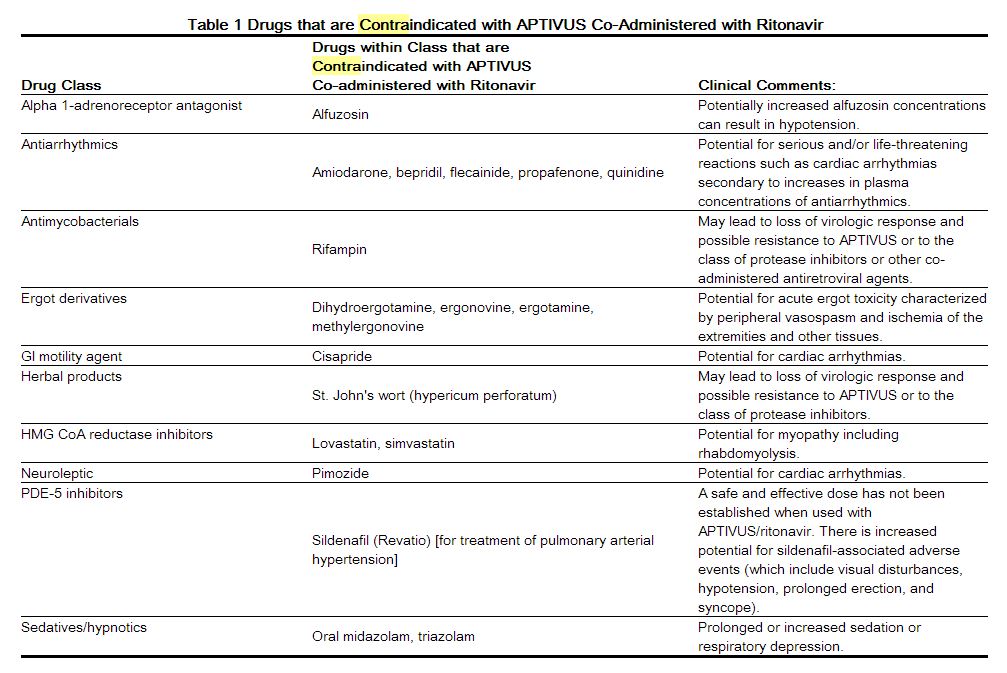
Drug Interactions
Co-administration of APTIVUS/ritonavir with drugs that are highly dependent on CYP 3A for clearance or are potent CYP 3A inducers are contraindicated (see Table 1). These recommendations are based on either drug interaction studies or they are predicted interactions due to the expected magnitude of interaction and potential for serious events or loss of efficacy. For information regarding clinical recommendations [see Drug Interactions (7.2)].
 |
Due to the need for co-administration of APTIVUS with ritonavir, please refer to the ritonavir prescribing information for a description of ritonavir contraindications.[1]
References
Adapted from the FDA Package Insert.