Rivastigmine (oral): Difference between revisions
m (Aparna Vuppala moved page Rivastigmine to Rivastigmine (oral)) |
No edit summary |
||
| Line 5: | Line 5: | ||
<!--Overview--> | <!--Overview--> | ||
|genericName= | |genericName= | ||
|aOrAn= | |aOrAn= | ||
an | an | ||
|drugClass= | |drugClass= | ||
acetylcholinesterase inhibitor | acetylcholinesterase inhibitor | ||
|indication= | |indication= | ||
mild to moderate [[dementia]] of the [[alzheimer’s]] type and mild to moderate [[dementia]] associated with [[parkinson’s disease]] | mild to moderate [[dementia]] of the [[alzheimer’s]] type and mild to moderate [[dementia]] associated with [[parkinson’s disease]] | ||
|hasBlackBoxWarning= | |hasBlackBoxWarning= | ||
|adverseReactions= | |adverseReactions= | ||
| Line 29: | Line 21: | ||
<!--Black Box Warning--> | <!--Black Box Warning--> | ||
|blackBoxWarningTitle= | |blackBoxWarningTitle= | ||
Title | Title | ||
|blackBoxWarningBody= | |blackBoxWarningBody= | ||
<i><span style="color:#FF0000;">ConditionName: </span></i> | <i><span style="color:#FF0000;">ConditionName: </span></i> | ||
| Line 41: | Line 31: | ||
<!--FDA-Labeled Indications and Dosage (Adult)--> | <!--FDA-Labeled Indications and Dosage (Adult)--> | ||
|fdaLIADAdult= | |fdaLIADAdult= | ||
| Line 71: | Line 60: | ||
<!--Guideline-Supported Use (Adult)--> | <!--Guideline-Supported Use (Adult)--> | ||
|offLabelAdultGuideSupport= | |offLabelAdultGuideSupport= | ||
There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of | There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Rivastigmine in adult patients. | ||
<!--Non–Guideline-Supported Use (Adult)--> | <!--Non–Guideline-Supported Use (Adult)--> | ||
|offLabelAdultNoGuideSupport= | |offLabelAdultNoGuideSupport= | ||
| Line 91: | Line 78: | ||
<!--FDA-Labeled Indications and Dosage (Pediatric)--> | <!--FDA-Labeled Indications and Dosage (Pediatric)--> | ||
|fdaLIADPed= | |fdaLIADPed= | ||
There is limited information regarding <i>FDA-Labeled Use</i> of | There is limited information regarding <i>FDA-Labeled Use</i> of Rivastigmine in pediatric patients. | ||
<!--Off-Label Use and Dosage (Pediatric)--> | <!--Off-Label Use and Dosage (Pediatric)--> | ||
<!--Guideline-Supported Use (Pediatric)--> | <!--Guideline-Supported Use (Pediatric)--> | ||
|offLabelPedGuideSupport= | |offLabelPedGuideSupport= | ||
There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of | There is limited information regarding <i>Off-Label Guideline-Supported Use</i> of Rivastigmine in pediatric patients. | ||
<!--Non–Guideline-Supported Use (Pediatric)--> | <!--Non–Guideline-Supported Use (Pediatric)--> | ||
|offLabelPedNoGuideSupport= | |offLabelPedNoGuideSupport= | ||
There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of | There is limited information regarding <i>Off-Label Non–Guideline-Supported Use</i> of Rivastigmine in pediatric patients. | ||
<!--Contraindications--> | <!--Contraindications--> | ||
|contraindications= | |contraindications= | ||
| Line 121: | Line 104: | ||
<!--Warnings--> | <!--Warnings--> | ||
|warnings= | |warnings= | ||
| Line 162: | Line 144: | ||
<!--Clinical Trials Experience--> | <!--Clinical Trials Experience--> | ||
|clinicalTrials= | |clinicalTrials= | ||
| Line 181: | Line 162: | ||
:*The most common adverse reactions leading to discontinuation, defined as those occurring in at least 2% of patients and at twice the incidence seen in placebo patients, are shown in Table 1. | :*The most common adverse reactions leading to discontinuation, defined as those occurring in at least 2% of patients and at twice the incidence seen in placebo patients, are shown in Table 1. | ||
: [[File: | : [[File:Rivastigmine01.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
*Adverse Reactions Observed at an Incidence of at Least 2% | *Adverse Reactions Observed at an Incidence of at Least 2% | ||
| Line 188: | Line 169: | ||
:*No systematic effect of race or age could be determined from the incidence of adverse events in the controlled studies. [[Nausea]], [[vomiting]] and [[weight loss]] were more frequent in women than men. | :*No systematic effect of race or age could be determined from the incidence of adverse events in the controlled studies. [[Nausea]], [[vomiting]] and [[weight loss]] were more frequent in women than men. | ||
: [[File: | : [[File:Rivastigmine02.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
:*Nausea and Vomiting: In the controlled clinical trials, 47% of the patients treated with an Exelon dose in the therapeutic range of 6 mg to 12 mg per day (n=1189) developed [[nausea]] (compared with 12% in placebo). A total of 31% of Exelon-treated patients developed at least 1 episode of vomiting (compared with 6% for placebo). The rate of [[vomiting]] was higher during the titration phase (24% versus 3% for placebo) than in the maintenance phase (14% versus 3% for placebo). The rates were higher in women than men. Five percent of patients discontinued for vomiting, compared to less than 1% for patients on placebo. Vomiting was severe in 2% of Exelon-treated patients and was rated as mild or moderate each in 14% of patients. The rate of nausea was higher during the titration phase (43% versus 9% for placebo) than in the maintenance phase (17% versus 4% for placebo). | :*Nausea and Vomiting: In the controlled clinical trials, 47% of the patients treated with an Exelon dose in the therapeutic range of 6 mg to 12 mg per day (n=1189) developed [[nausea]] (compared with 12% in placebo). A total of 31% of Exelon-treated patients developed at least 1 episode of vomiting (compared with 6% for placebo). The rate of [[vomiting]] was higher during the titration phase (24% versus 3% for placebo) than in the maintenance phase (14% versus 3% for placebo). The rates were higher in women than men. Five percent of patients discontinued for vomiting, compared to less than 1% for patients on placebo. Vomiting was severe in 2% of Exelon-treated patients and was rated as mild or moderate each in 14% of patients. The rate of nausea was higher during the titration phase (43% versus 9% for placebo) than in the maintenance phase (17% versus 4% for placebo). | ||
| Line 209: | Line 190: | ||
:*In general, adverse reactions were less frequent later in the course of treatment. | :*In general, adverse reactions were less frequent later in the course of treatment. | ||
: [[File: | : [[File:Rivastigmine03.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
*Other Adverse Events Observed During Clinical Trials | *Other Adverse Events Observed During Clinical Trials | ||
| Line 341: | Line 322: | ||
<!--Postmarketing Experience--> | <!--Postmarketing Experience--> | ||
|postmarketing= | |postmarketing= | ||
| Line 359: | Line 339: | ||
<!--Drug Interactions--> | <!--Drug Interactions--> | ||
|drugInteractions= | |drugInteractions= | ||
| Line 366: | Line 345: | ||
<!--Use in Specific Populations--> | <!--Use in Specific Populations--> | ||
|useInPregnancyFDA= | |useInPregnancyFDA= | ||
* '''Pregnancy Category B''' | * '''Pregnancy Category B''' | ||
*There are no adequate and well-controlled studies in pregnant women. Reproduction studies conducted in pregnant rats and rabbits at oral doses up to 2.3 mg-base/kg/day, or 2 (rat) and 4 (rabbit) times the maximum recommended human dose (MRHD) of 12 mg per day on a mg/m2 basis, revealed no evidence of teratogenicity. Studies in pregnant rats showed slightly decreased fetal/pup weight at doses that were below the MRHD but usually associated with some maternal toxicity. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed. | *There are no adequate and well-controlled studies in pregnant women. Reproduction studies conducted in pregnant rats and rabbits at oral doses up to 2.3 mg-base/kg/day, or 2 (rat) and 4 (rabbit) times the maximum recommended human dose (MRHD) of 12 mg per day on a mg/m2 basis, revealed no evidence of teratogenicity. Studies in pregnant rats showed slightly decreased fetal/pup weight at doses that were below the MRHD but usually associated with some maternal toxicity. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed. | ||
|useInPregnancyAUS= | |useInPregnancyAUS= | ||
* '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | * '''Australian Drug Evaluation Committee (ADEC) Pregnancy Category''' | ||
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of | There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Rivastigmine in women who are pregnant. | ||
|useInLaborDelivery= | |useInLaborDelivery= | ||
There is no FDA guidance on use of | There is no FDA guidance on use of Rivastigmine during labor and delivery. | ||
|useInNursing= | |useInNursing= | ||
*Rivastigmine and its metabolites are excreted in rat milk following oral administration of rivastigmine; levels of rivastigmine plus metabolites in rat milk are approximately 2 times that in maternal plasma. It is not known whether rivastigmine is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from Exelon, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. | *Rivastigmine and its metabolites are excreted in rat milk following oral administration of rivastigmine; levels of rivastigmine plus metabolites in rat milk are approximately 2 times that in maternal plasma. It is not known whether rivastigmine is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from Exelon, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother. | ||
|useInPed= | |useInPed= | ||
*Safety and effectiveness in pediatric patients have not been established and therefore the use of rivastigmine in children and adolescents (below 18 years of age) is not recommended. | *Safety and effectiveness in pediatric patients have not been established and therefore the use of rivastigmine in children and adolescents (below 18 years of age) is not recommended. | ||
|useInGeri= | |useInGeri= | ||
*Of the total number of subjects in clinical studies of Exelon, 86 percent were 65 years and older while 46 percent were 75 years and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. | *Of the total number of subjects in clinical studies of Exelon, 86 percent were 65 years and older while 46 percent were 75 years and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out. | ||
|useInGender= | |useInGender= | ||
There is no FDA guidance on the use of | There is no FDA guidance on the use of Rivastigmine with respect to specific gender populations. | ||
|useInRace= | |useInRace= | ||
There is no FDA guidance on the use of | There is no FDA guidance on the use of Rivastigmine with respect to specific racial populations. | ||
|useInRenalImpair= | |useInRenalImpair= | ||
*In patients with moderate to severe renal impairment ([[glomerular filtration rate]] [GFR] <50 mL/min), clearance of oral rivastigmine was reduced. Accordingly, such patients may be able to only tolerate lower doses. | *In patients with moderate to severe renal impairment ([[glomerular filtration rate]] [GFR] <50 mL/min), clearance of oral rivastigmine was reduced. Accordingly, such patients may be able to only tolerate lower doses. | ||
|useInHepaticImpair= | |useInHepaticImpair= | ||
*In patients with mild or moderate [[hepatic impairment]] (Child-Pugh score 5 to 9), clearance of oral rivastigmine was reduced. Accordingly, such patients may be able to only tolerate lower doses. No data are available on the use of rivastigmine in patients with severe hepatic impairment. | *In patients with mild or moderate [[hepatic impairment]] (Child-Pugh score 5 to 9), clearance of oral rivastigmine was reduced. Accordingly, such patients may be able to only tolerate lower doses. No data are available on the use of rivastigmine in patients with severe hepatic impairment. | ||
|useInReproPotential= | |useInReproPotential= | ||
There is no FDA guidance on the use of | There is no FDA guidance on the use of Rivastigmine in women of reproductive potentials and males. | ||
|useInImmunocomp= | |useInImmunocomp= | ||
There is no FDA guidance one the use of | There is no FDA guidance one the use of Rivastigmine in patients who are immunocompromised. | ||
<!--Administration and Monitoring--> | <!--Administration and Monitoring--> | ||
|administration= | |administration= | ||
* Oral | * Oral | ||
|monitoring= | |monitoring= | ||
There is limited information regarding <i>Monitoring</i> of | There is limited information regarding <i>Monitoring</i> of Rivastigmine in the drug label. | ||
<!--IV Compatibility--> | <!--IV Compatibility--> | ||
|IVCompat= | |IVCompat= | ||
There is limited information regarding <i>IV Compatibility</i> of | There is limited information regarding <i>IV Compatibility</i> of Rivastigmine in the drug label. | ||
<!--Overdosage--> | <!--Overdosage--> | ||
|overdose= | |overdose= | ||
| Line 444: | Line 407: | ||
===Chronic Overdose=== | ===Chronic Overdose=== | ||
There is limited information regarding <i>Chronic Overdose</i> of | There is limited information regarding <i>Chronic Overdose</i> of Rivastigmine in the drug label. | ||
<!--Pharmacology--> | <!--Pharmacology--> | ||
<!--Drug box 2--> | <!--Drug box 2--> | ||
|drugBox= | |drugBox= | ||
| Line 508: | Line 470: | ||
<!--Mechanism of Action--> | <!--Mechanism of Action--> | ||
|mechAction= | |mechAction= | ||
| Line 514: | Line 475: | ||
<!--Structure--> | <!--Structure--> | ||
|structure= | |structure= | ||
| Line 521: | Line 481: | ||
*The distribution coefficient at 37°C in n-octanol/phosphate buffer solution pH 7 is 3.0. | *The distribution coefficient at 37°C in n-octanol/phosphate buffer solution pH 7 is 3.0. | ||
: [[File: | : [[File:Rivastigmine19.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
*Exelon Capsules contain rivastigmine tartrate, equivalent to 1.5 mg, 3 mg, 4.5 mg, and 6 mg of rivastigmine base for oral administration. Inactive ingredients are hydroxypropyl methylcellulose, magnesium stearate, microcrystalline cellulose, and silicon dioxide. Each hard-gelatin capsule contains gelatin, titanium dioxide and red and/or yellow iron oxides. | *Exelon Capsules contain rivastigmine tartrate, equivalent to 1.5 mg, 3 mg, 4.5 mg, and 6 mg of rivastigmine base for oral administration. Inactive ingredients are hydroxypropyl methylcellulose, magnesium stearate, microcrystalline cellulose, and silicon dioxide. Each hard-gelatin capsule contains gelatin, titanium dioxide and red and/or yellow iron oxides. | ||
| Line 528: | Line 488: | ||
<!--Pharmacodynamics--> | <!--Pharmacodynamics--> | ||
|PD= | |PD= | ||
| Line 536: | Line 495: | ||
<!--Pharmacokinetics--> | <!--Pharmacokinetics--> | ||
|PK= | |PK= | ||
| Line 583: | Line 541: | ||
<!--Nonclinical Toxicology--> | <!--Nonclinical Toxicology--> | ||
|nonClinToxic= | |nonClinToxic= | ||
| Line 596: | Line 553: | ||
<!--Clinical Studies--> | <!--Clinical Studies--> | ||
|clinicalStudies= | |clinicalStudies= | ||
| Line 615: | Line 571: | ||
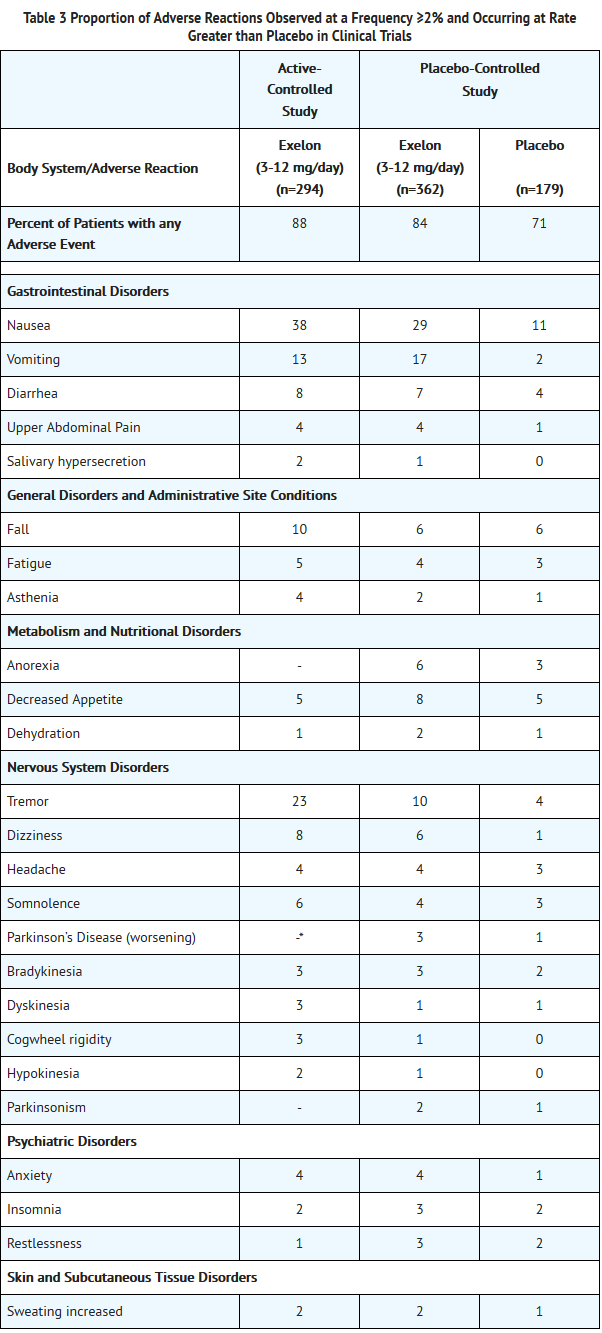
:*Figure 1 illustrates the time course for the change from baseline in ADAS-cog scores for all 3 dose groups over the 26 weeks of the study. At 26 weeks of treatment, the mean differences in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo were 1.9 and 4.9 units for the 1 mg to 4 mg and 6 mg to 12 mg treatments, respectively. Both treatments were statistically significantly superior to placebo and the 6 mg to 12 mg per day range was significantly superior to the 1 mg to 4 mg per day range. | :*Figure 1 illustrates the time course for the change from baseline in ADAS-cog scores for all 3 dose groups over the 26 weeks of the study. At 26 weeks of treatment, the mean differences in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo were 1.9 and 4.9 units for the 1 mg to 4 mg and 6 mg to 12 mg treatments, respectively. Both treatments were statistically significantly superior to placebo and the 6 mg to 12 mg per day range was significantly superior to the 1 mg to 4 mg per day range. | ||
: [[File: | : [[File:Rivastigmine05.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
:*Figure 2 illustrates the cumulative percentages of patients from each of the 3 treatment groups who had attained at least the measure of improvement in ADAS-cog score shown on the X-axis. Three change scores, (7-point and 4-point reductions from baseline or no change in score) have been identified for illustrative purposes, and the percent of patients in each group achieving that result is shown in the inset table. | :*Figure 2 illustrates the cumulative percentages of patients from each of the 3 treatment groups who had attained at least the measure of improvement in ADAS-cog score shown on the X-axis. Three change scores, (7-point and 4-point reductions from baseline or no change in score) have been identified for illustrative purposes, and the percent of patients in each group achieving that result is shown in the inset table. | ||
:*The curves demonstrate that both patients assigned to Exelon and placebo have a wide range of responses, but that the Exelon groups are more likely to show the greater improvements. A curve for an effective treatment would be shifted to the left of the curve for placebo, while an ineffective or deleterious treatment would be superimposed upon, or shifted to the right of the curve for placebo, respectively. | :*The curves demonstrate that both patients assigned to Exelon and placebo have a wide range of responses, but that the Exelon groups are more likely to show the greater improvements. A curve for an effective treatment would be shifted to the left of the curve for placebo, while an ineffective or deleterious treatment would be superimposed upon, or shifted to the right of the curve for placebo, respectively. | ||
: [[File: | : [[File:Rivastigmine06.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
:*Figure 3 is a histogram of the frequency distribution of CIBIC-Plus scores attained by patients assigned to each of the 3 treatment groups who completed 26 weeks of treatment. The mean Exelon-placebo differences for these groups of patients in the mean rating of change from baseline were 0.32 units and 0.35 units for 1 mg to 4 mg and 6 mg to 12 mg of Exelon, respectively. The mean ratings for the 6 mg to 12 mg per day and 1 mg to 4 mg per day groups were statistically significantly superior to placebo. The differences between the 6 mg to 12 mg per day and the 1 mg to 4 mg per day groups were statistically significant. | :*Figure 3 is a histogram of the frequency distribution of CIBIC-Plus scores attained by patients assigned to each of the 3 treatment groups who completed 26 weeks of treatment. The mean Exelon-placebo differences for these groups of patients in the mean rating of change from baseline were 0.32 units and 0.35 units for 1 mg to 4 mg and 6 mg to 12 mg of Exelon, respectively. The mean ratings for the 6 mg to 12 mg per day and 1 mg to 4 mg per day groups were statistically significantly superior to placebo. The differences between the 6 mg to 12 mg per day and the 1 mg to 4 mg per day groups were statistically significant. | ||
: [[File: | : [[File:Rivastigmine07.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
*Global 26-Week Study in Mild to Moderate Alzheimer’s Disease (Study 2) | *Global 26-Week Study in Mild to Moderate Alzheimer’s Disease (Study 2) | ||
| Line 630: | Line 586: | ||
:*Figure 4 illustrates the time course for the change from baseline in ADAS-cog scores for all 3 dose groups over the 26 weeks of the study. At 26 weeks of treatment, the mean differences in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo were 0.2 and 2.6 units for the 1 mg to 4 mg and 6 mg to 12 mg treatments, respectively. The 6 mg to 12 mg per day group was statistically significantly superior to placebo, as well as to the 1 mg to 4 mg per day group. The difference between the 1 mg to 4 mg per day group and placebo was not statistically significant. | :*Figure 4 illustrates the time course for the change from baseline in ADAS-cog scores for all 3 dose groups over the 26 weeks of the study. At 26 weeks of treatment, the mean differences in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo were 0.2 and 2.6 units for the 1 mg to 4 mg and 6 mg to 12 mg treatments, respectively. The 6 mg to 12 mg per day group was statistically significantly superior to placebo, as well as to the 1 mg to 4 mg per day group. The difference between the 1 mg to 4 mg per day group and placebo was not statistically significant. | ||
: [[File: | : [[File:Rivastigmine08.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
:*Figure 5 illustrates the cumulative percentages of patients from each of the 3 treatment groups who had attained at least the measure of improvement in ADAS-cog score shown on the X-axis. Similar to the U.S. 26-week study, the curves demonstrate that both patients assigned to Exelon and placebo have a wide range of responses, but that the 6 mg to 12 mg per day Exelon group is more likely to show the greater improvements. | :*Figure 5 illustrates the cumulative percentages of patients from each of the 3 treatment groups who had attained at least the measure of improvement in ADAS-cog score shown on the X-axis. Similar to the U.S. 26-week study, the curves demonstrate that both patients assigned to Exelon and placebo have a wide range of responses, but that the 6 mg to 12 mg per day Exelon group is more likely to show the greater improvements. | ||
: [[File: | : [[File:Rivastigmine09.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
:*Figure 6 is a histogram of the frequency distribution of CIBIC-Plus scores attained by patients assigned to each of the 3 treatment groups who completed 26 weeks of treatment. The mean Exelon-placebo differences for these groups of patients for the mean rating of change from baseline were 0.14 units and 0.41 units for 1 mg to 4 mg and 6 mg to 12 mg of Exelon, respectively. The mean ratings for the 6 mg to 12 mg per day group were statistically significantly superior to placebo. The comparison of the mean ratings for the 1 mg to 4 mg per day group and placebo group was not statistically significant. | :*Figure 6 is a histogram of the frequency distribution of CIBIC-Plus scores attained by patients assigned to each of the 3 treatment groups who completed 26 weeks of treatment. The mean Exelon-placebo differences for these groups of patients for the mean rating of change from baseline were 0.14 units and 0.41 units for 1 mg to 4 mg and 6 mg to 12 mg of Exelon, respectively. The mean ratings for the 6 mg to 12 mg per day group were statistically significantly superior to placebo. The comparison of the mean ratings for the 1 mg to 4 mg per day group and placebo group was not statistically significant. | ||
: [[File: | : [[File:Rivastigmine10.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
*U.S. Fixed-Dose Study in Mild to Moderate Alzheimer’s Disease (Study 3) | *U.S. Fixed-Dose Study in Mild to Moderate Alzheimer’s Disease (Study 3) | ||
| Line 653: | Line 609: | ||
:*Figure 7 illustrates the time course for the change from baseline in ADAS-cog scores for both treatment groups over the 24-week study. At 24 weeks of treatment, the mean difference in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo was 3.8 points. This treatment difference was statistically significant in favor of Exelon when compared to placebo. | :*Figure 7 illustrates the time course for the change from baseline in ADAS-cog scores for both treatment groups over the 24-week study. At 24 weeks of treatment, the mean difference in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo was 3.8 points. This treatment difference was statistically significant in favor of Exelon when compared to placebo. | ||
: [[File: | : [[File:Rivastigmine11.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
:*Figure 8 is a histogram of the distribution of patients’ scores on the ADCS-CGIC (Alzheimer’s Disease Cooperative Study - Clinician’s Global Impression of Change) at 24 weeks. The mean difference in change scores between the Exelon and placebo groups from baseline was 0.5 points. This difference was statistically significant in favor of Exelon treatment. | :*Figure 8 is a histogram of the distribution of patients’ scores on the ADCS-CGIC (Alzheimer’s Disease Cooperative Study - Clinician’s Global Impression of Change) at 24 weeks. The mean difference in change scores between the Exelon and placebo groups from baseline was 0.5 points. This difference was statistically significant in favor of Exelon treatment. | ||
: [[File: | : [[File:Rivastigmine12.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
:*Patients’ age, gender, or race did not predict clinical outcome of Exelon treatment. | :*Patients’ age, gender, or race did not predict clinical outcome of Exelon treatment. | ||
<!--How Supplied--> | <!--How Supplied--> | ||
|howSupplied= | |howSupplied= | ||
| Line 698: | Line 653: | ||
<!--Patient Counseling Information--> | <!--Patient Counseling Information--> | ||
|fdaPatientInfo= | |fdaPatientInfo= | ||
| Line 713: | Line 667: | ||
:*Caregivers and patients should be advised that [[cholinomimetics]], including rivastigmine, may exacerbate or induce extrapyramidal symptoms. Worsening in patients with [[Parkinson’s disease]], including an increased incidence or intensity of [[tremor]], has been observed. | :*Caregivers and patients should be advised that [[cholinomimetics]], including rivastigmine, may exacerbate or induce extrapyramidal symptoms. Worsening in patients with [[Parkinson’s disease]], including an increased incidence or intensity of [[tremor]], has been observed. | ||
: [[File: | : [[File:Rivastigmine04.png|thumb|none|600px|This image is provided by the National Library of Medicine.]] | ||
<!--Precautions with Alcohol--> | <!--Precautions with Alcohol--> | ||
|alcohol= | |alcohol= | ||
* Alcohol- | * Alcohol-Rivastigmine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication. | ||
<!--Brand Names--> | <!--Brand Names--> | ||
|brandNames= | |brandNames= | ||
| Line 728: | Line 680: | ||
<!--Look-Alike Drug Names--> | <!--Look-Alike Drug Names--> | ||
|lookAlike= | |lookAlike= | ||
<!--Drug Shortage Status--> | <!--Drug Shortage Status--> | ||
|drugShortage= | |drugShortage= | ||
}} | }} | ||
| Line 756: | Line 706: | ||
{{LabelImage | {{LabelImage | ||
|fileName= | |fileName=Rivastigmine13.png|This image is provided by the National Library of Medicine. | ||
}} | }} | ||
{{LabelImage | {{LabelImage | ||
|fileName= | |fileName=Rivastigmine14.png|This image is provided by the National Library of Medicine. | ||
}} | }} | ||
{{LabelImage | {{LabelImage | ||
|fileName= | |fileName=Rivastigmine15.png|This image is provided by the National Library of Medicine. | ||
}} | }} | ||
{{LabelImage | {{LabelImage | ||
|fileName= | |fileName=Rivastigmine16.png|This image is provided by the National Library of Medicine. | ||
}} | }} | ||
{{LabelImage | {{LabelImage | ||
|fileName= | |fileName=Rivastigmine17.png|This image is provided by the National Library of Medicine. | ||
}} | }} | ||
{{LabelImage | {{LabelImage | ||
|fileName= | |fileName=Rivastigmine18.png|This image is provided by the National Library of Medicine. | ||
}} | }} | ||
Revision as of 20:16, 7 May 2015
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Rivastigmine (oral) is an acetylcholinesterase inhibitor that is FDA approved for the {{{indicationType}}} of mild to moderate dementia of the alzheimer’s type and mild to moderate dementia associated with parkinson’s disease. Common adverse reactions include nausea, vomiting, anorexia, dyspepsia, and asthenia.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Alzheimer’s Disease
- The dosage of Exelon shown to be effective in controlled clinical trials in Alzheimer’s disease is 6 mg to 12 mg per day, administered twice a day (daily doses of 3 mg to 6 mg twice a day). There is evidence from the clinical trials that doses at the higher end of this range may be more beneficial.
- Initial Dose
- Initiate treatment with the 1.5 mg twice a day with Exelon.
- Dose Titration
- After a minimum of 2 weeks and if well tolerated, increase the dose to 3 mg twice a day. Subsequent increases to 4.5 mg twice a day and 6 mg twice a day should be attempted after a minimum of 2 weeks at the previous dose and if well tolerated. The maximum dose is 6 mg twice a day (12 mg per day).
Parkinson’s Disease Dementia
- The dosage of Exelon shown to be effective in the single controlled clinical trial conducted in dementia associated with Parkinson’s disease is 3 mg to 12 mg per day, administered twice a day (daily doses of 1.5 mg to 6 mg twice a day).
- Initial Dose
- Initiate treatment with the 1.5 mg twice a day with Exelon.
- Dose Titration
- After a minimum of 4 weeks and if well tolerated, increase the dose to 3 mg twice a day. Subsequent increases to 4.5 mg twice a day and 6 mg twice a day should be attempted after a minimum of 4 weeks at the previous dose and if well tolerated. The maximum dose is 6 mg twice a day (12 mg per day).
- Interruption of Treatment
- If adverse effects (e.g., nausea, vomiting, abdominal pain, loss of appetite) cause intolerance during treatment, the patient should be instructed to discontinue treatment for several doses and then restart at the same or next lower dose level.
- If dosing is interrupted for 3 days or fewer, restart treatment with the same or lower dose of Exelon. If dosing is interrupted for more than 3 days, treatment should be restarted with 1.5 mg twice a day and titrated as described above.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Rivastigmine in adult patients.
Non–Guideline-Supported Use
Multi-infarct dementia
- Rivastigmine 3 to 6 mg daily.
Senile dementia of the Lewy body type
- Rivastigmine in doses of 3 to 12 mg daily.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Rivastigmine in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Rivastigmine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Rivastigmine in pediatric patients.
Contraindications
- Exelon is contraindicated in patients with:
- known hypersensitivity to rivastigmine, other carbamate derivatives or other components of the formulation.
- a previous history of application site reaction with rivastigmine transdermal patch suggestive of allergic contact dermatitis, in the absence of negative allergy testing.
- Isolated cases of generalized skin reactions have been described in postmarketing experience.
Warnings
Precautions
- Gastrointestinal Adverse Reactions
- Exelon can cause gastrointestinal adverse reactions, including significant nausea, vomiting, diarrhea, anorexia/decreased appetite, and weight loss. Dehydration may result from prolonged vomiting or diarrhea and can be associated with serious outcomes. The incidence and severity of these reactions are dose-related. For this reason, patients should always be started at a dose of 1.5 mg twice a day and titrated to their maintenance dose.
- If treatment is interrupted for longer than 3 days, treatment should be reinitiated with the lowest daily dose to reduce the possibility of severe vomiting and its potentially serious sequelae (e.g., there has been one postmarketing report of severe vomiting with esophageal rupture following inappropriate reinitiation of treatment with a 4.5-mg dose after 8 weeks of treatment interruption).
- Inform caregivers to monitor for gastrointestinal adverse reactions and to inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than 3 days because of intolerance, the next dose should not be administered without contacting the physician regarding proper retitration.
- Hypersensitivity Reactions of the Skin
- There have been isolated postmarketing reports of patients experiencing disseminated hypersensitivity reactions of the skin when administered rivastigmine irrespective of the route of administration (oral or transdermal). Treatment should be discontinued if disseminated hypersensitivity reaction of the skin occurs. Patients and caregivers should be instructed accordingly.
- In patients who develop application site reactions suggestive of allergic contact dermatitis to Exelon Patch and who still require rivastigmine, treatment should be switched to oral rivastigmine only after negative allergy testing and under close medical supervision. It is possible that some patients sensitized to rivastigmine by exposure to rivastigmine patch may not be able to take rivastigmine in any form.
- Other Adverse Reactions from Increased Cholinergic Activity
- Neurologic Effects
- Extrapyramidal Symptoms: Cholinomimetics, including rivastigmine may exacerbate or induce extrapyramidal symptoms. Worsening of parkinsonian symptoms, particularly tremor, has been observed in patients with dementia associated with Parkinson’s disease who were treated with Exelon Capsules.
- Seizures: Drugs that increase cholinergic activity are believed to have some potential for causing seizures. However, seizure activity also may be a manifestation of Alzheimer's disease.
- Peptic Ulcers/Gastrointestinal Bleeding
- Cholinesterase inhibitors, including rivastigmine, may be expected to increase gastric acid secretion due to increased cholinergic activity. Monitor patients using Exelon for symptoms of active or occult gastrointestinal bleeding, especially those at increased risk for developing ulcers, e.g., those with a history of ulcer disease or those receiving concurrent nonsteroidal anti-inflammatory drugs (NSAIDs). Clinical studies of rivastigmine have shown no significant increase, relative to placebo, in the incidence of either peptic ulcer disease or gastrointestinal bleeding.
- Use with Anesthesia
- Rivastigmine, as a cholinesterase inhibitor, is likely to exaggerate succinylcholine-type muscle relaxation during anesthesia.
- Cardiac Conduction Effects
- Because rivastigmine increases cholinergic activity, use of rivastigmine may have vagotonic effects on heart rate (e.g., bradycardia). The potential for this action may be particularly important in patients with sick sinus syndrome or other supraventricular cardiac conduction conditions. In clinical trials, rivastigmine was not associated with any increased incidence of cardiovascular adverse events, heart rate or blood pressure changes, or ECG abnormalities. Syncopal episodes have been reported in 3% of patients receiving 6 mg to 12 mg per day of Exelon, compared to 2% of placebo patients.
- Genitourinary Effects
- Although not observed in clinical trials of rivastigmine, drugs that increase cholinergic activity may cause urinary obstruction.
- Pulmonary Effects
- Drugs that increase cholinergic activity, including rivastigmine, should be used with care in patients with a history of asthma or obstructive pulmonary disease.
- Impairment in Driving or Use of Machinery
- Dementia may cause gradual impairment of driving performance or compromise the ability to use machinery. The administration of rivastigmine may also result in adverse reactions that are detrimental to these functions. During treatment with the Exelon, routinely evaluate the patient’s ability to continue driving or operating machinery.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- Exelon has been administered to over 5,297 individuals during clinical trials worldwide. Of these, 4,326 patients have been treated for at least 3 months, 3,407 patients have been treated for at least 6 months, 2,150 patients have been treated for 1 year, 1,250 patients have been treated for 2 years, and 168 patients have been treated for over 3 years. With regard to exposure to the highest dose, 2,809 patients were exposed to doses of 10 mg to 12 mg, 2,615 patients treated for 3 months, 2,328 patients treated for 6 months, 1,378 patients treated for 1 year, 917 patients treated for 2 years, and 129 patients treated for over 3 years.
Mild to Moderate Alzheimer’s Disease
- Most Commonly Observed Adverse Reactions
- Gastrointestinal Adverse Reactions
- Exelon use is associated with significant nausea, vomiting, and weight loss.
- Discontinuation Rates
- The rate of discontinuation due to adverse events in controlled clinical trials of Exelon (rivastigmine tartrate) was 15% for patients receiving 6 mg to 12 mg per day compared to 5% for patients on placebo during forced weekly dose titration. While on a maintenance dose, the rates were 6% for patients on Exelon compared to 4% for those on placebo.
- The most common adverse reactions leading to discontinuation, defined as those occurring in at least 2% of patients and at twice the incidence seen in placebo patients, are shown in Table 1.
- Adverse Reactions Observed at an Incidence of at Least 2%
- Table 2 lists treatment-emergent signs and symptoms that were reported in at least 2% of patients in placebo-controlled trials and for which the rate of occurrence was greater for patients treated with Exelon doses of 6 mg to 12 mg per day than for those treated with placebo. The prescriber should be aware that these figures cannot be used to predict the frequency of adverse events in the course of usual medical practice when patient characteristics and other factors may differ from those prevailing during clinical studies. Similarly, the cited frequencies cannot be directly compared with figures obtained from other clinical investigations involving different treatments, uses, or investigators. An inspection of these frequencies, however, does provide the prescriber with one basis by which to estimate the relative contribution of drug and nondrug factors to the adverse event incidences in the population studied.
- In general, adverse reactions were less frequent later in the course of treatment.
- No systematic effect of race or age could be determined from the incidence of adverse events in the controlled studies. Nausea, vomiting and weight loss were more frequent in women than men.
- Nausea and Vomiting: In the controlled clinical trials, 47% of the patients treated with an Exelon dose in the therapeutic range of 6 mg to 12 mg per day (n=1189) developed nausea (compared with 12% in placebo). A total of 31% of Exelon-treated patients developed at least 1 episode of vomiting (compared with 6% for placebo). The rate of vomiting was higher during the titration phase (24% versus 3% for placebo) than in the maintenance phase (14% versus 3% for placebo). The rates were higher in women than men. Five percent of patients discontinued for vomiting, compared to less than 1% for patients on placebo. Vomiting was severe in 2% of Exelon-treated patients and was rated as mild or moderate each in 14% of patients. The rate of nausea was higher during the titration phase (43% versus 9% for placebo) than in the maintenance phase (17% versus 4% for placebo).
- Weight Decreased: In the controlled trials, approximately 26% of women on high doses of Exelon (greater than 9 mg per day) had weight loss equal to or greater than 7% of their baseline weight compared to 6% in the placebo-treated patients. About 18% of the males in the high-dose group experienced a similar degree of weight loss compared to 4% in placebo-treated patients. It is not clear how much of the weight loss was associated with anorexia, nausea, vomiting, and the diarrhea associated with the drug.
- Anorexia: In the controlled clinical trials, of the patients treated with an Exelon dose of 6 mg to 12 mg per day, 17% developed anorexia compared to 3% of the placebo patients. Neither the time course nor the severity of the anorexia is known.
Mild to Moderate Parkinson’s Disease Dementia
- Exelon has been administered to 779 individuals during clinical trials worldwide. Of these, 663 patients have been treated for at least 3 months, 476 patients have been treated for at least 6 months, and 313 patients have been treated for 1 year.
- Most Commonly Observed Adverse Reactions
- Discontinuation Rates
- The rate of discontinuation due to adverse events in the single placebo-controlled trial of Exelon was 18.2% for patients receiving 3 mg to 12 mg per day compared to 11.2% for patients on placebo during the 24-week study.
- The most frequent adverse events that led to discontinuation from this study, defined as those occurring in at least 1% of patients receiving Exelon and more frequent than those receiving placebo, were nausea (3.6% Exelon versus 0.6% placebo), vomiting (1.9% Exelon versus 0.6% placebo), and tremor (1.7% Exelon versus 0.0% placebo).
- Adverse Reactions Observed at an Incidence of at Least 2%
- Table 3 lists treatment-emergent signs and symptoms that were reported in at least 2% of patients in a single placebo-controlled trial and during the first 24 weeks of a 76-week open-label active-controlled trial for which the rate of occurrence was greater for patients treated with Exelon doses of 3 mg to 12 mg per day than for those treated with placebo in the placebo-controlled trial. The prescriber should be aware that these figures cannot be used to predict the frequency of adverse events in the course of usual medical practice when patient characteristics and other factors may differ from those prevailing during clinical studies. Similarly, the cited frequencies cannot be directly compared with figures obtained from other clinical investigations involving different treatments, uses, or investigators. An inspection of these frequencies, however, does provide the prescriber with 1 basis by which to estimate the relative contribution of drug and non-drug factors to the adverse event incidences in the population studied.
- In general, adverse reactions were less frequent later in the course of treatment.
- Other Adverse Events Observed During Clinical Trials
Mild to Moderate Alzheimer’s Disease
- Treatment-emergent signs and symptoms that occurred during 8 controlled clinical trials and 9 open-label trials in North America, Western Europe, Australia, South Africa, and Japan were recorded as adverse events by the clinical investigators using terminology of their own choosing. To provide an overall estimate of the proportion of individuals having similar types of events, the events were grouped into a smaller number of standardized categories using a modified WHO dictionary, and event frequencies were calculated across all studies. These categories are used in the listing below. The frequencies represent the proportion of 5,297 patients from these trials who experienced that event while receiving Exelon. All adverse events occurring in at least 6 patients (approximately 0.1%) are included, except for those already listed elsewhere in labeling, WHO terms too general to be informative, relatively minor events, or events unlikely to be drug-caused.
- Events are classified by body system and listed using the following definitions: frequent adverse events-those occurring in at least 1/100 patients; infrequent adverse events-those occurring in 1/100 to 1/1,000 patients. These adverse events are not necessarily related to Exelon treatment and in most cases were observed at a similar frequency in placebo-treated patients in the controlled studies.
Autonomic Nervous System
Infrequent: Increased saliva.
Body as a Whole
Frequent: Allergy, hot flushes.
Cardiovascular System
Frequent: Hypotension, postural hypotension.
Central and Peripheral Nervous System
Frequent: Abnormal gait, ataxia, paresthesia, convulsions. Infrequent: Dysphonia, hypoesthesia, migraine, nystagmus.
Gastrointestinal System
Frequent: Gastritis, constipation, flatulence. Infrequent: Gastric ulcer, gastroesophageal reflux, GI hemorrhage, hernia, melena, rectal hemorrhage, duodenal ulcer, hematemesis, pancreatitis.
Hearing and Vestibular Disorders
Frequent: Tinnitus.
Heart Rate and Rhythm Disorders
Frequent: Atrial fibrillation, bradycardia, palpitation. Infrequent: AV block, sick sinus syndrome, supraventricular tachycardia, extrasystoles, tachycardia.
Liver and Biliary System Disorders
Infrequent: Abnormal hepatic function tests.
Metabolic and Nutritional Disorders
Frequent: Dehydration, hypokalemia. Infrequent: Hyponatremia.
Musculoskeletal Disorders
Infrequent: Muscle weakness.
Myo-, Endo-, Pericardial and Valve Disorders
Frequent: Angina pectoris, myocardial infarction.
Psychiatric Disorders
Frequent: Confusion. Infrequent: Apathy, suicide attempt, increased libido, suicidal ideation.
Reproductive Disorders (Female and Male)
Infrequent: Breast pain.
Respiratory System
Infrequent: Bronchospasm.
Skin and Appendages
Frequent: Rashes of various kinds (maculopapular, eczema, bullous, exfoliative, psoriaform, erythematous). Infrequent: Urticaria, contact dermatitis.
Urinary System Disorders
Frequent: Hematuria. Infrequent: Acute renal failure, dysuria.
Vascular (extracardiac) Disorders
Infrequent: Peripheral ischemia, intracranial hemorrhage.
Vision Disorders
Frequent: Cataract. Infrequent: Diplopia, glaucoma.
White Cell and Resistance Disorders
Infrequent: Lymphadenopathy.
Mild to Moderate Parkinson’s Disease Dementia
- Additional treatment-emergent adverse events in patients with Parkinson’s disease dementia occurring in at least 1 patient (approximately 0.3%) are listed below, excluding events that are already listed above for the dementia of the Alzheimer’s type or elsewhere in labeling, WHO terms too general to be informative, relatively minor events, or events unlikely to be drug-caused. Events are classified by body system and listed using the following definitions: frequent adverse events– those occurring in at least 1/100 patients; infrequent adverse events–those occurring in 1/100 to 1/1,000 patients. These adverse events are not necessarily related to Exelon treatment and in most cases were observed at a similar frequency in placebo-treated patients in the controlled studies.
Central and Peripheral Nervous System
Frequent: Transient ischemic attack.
Gastrointestinal System
Frequent: Dyspepsia. Infrequent: Fecaloma, dysphagia.
General Disorders and Administration Site Conditions
Frequent: Chest pain.
Liver and Biliary System Disorders
Infrequent: Elevated gamma-glutamyltransferase level.
Musculoskeletal Disorders
Frequent: Back pain. Infrequent: Muscle stiffness.
Psychiatric Disorders
Frequent: Agitation, depression.
Respiratory System
Frequent: Dyspnea. Infrequent: Cough.
Urinary System Disorders
Infrequent: Urinary incontinence.
Vision Disorders
Infrequent: Blurred vision.
- Other Adverse Reactions Observed with Exelon Transdermal Patch
- The following additional adverse reactions have been observed with Exelon transdermal patch:
- Cerebrovascular accident, delirium, psychomotor hyperactivity, erythema, blister, dermatitis allergic.
Postmarketing Experience
- Voluntary reports of adverse events temporally associated with Exelon that have been received since market introduction that are not listed above, and that may or may not be causally related to the drug include the following:
Hepatobiliary Disorders
Psychiatric Disorders
Aggression.
Skin and Appendages
Stevens-Johnson syndrome, disseminated cutaneous hypersensitivity reactions.
Drug Interactions
- Cholinomimetic and Anticholinergic Drugs
- Increased cholinergic effects may be expected when rivastigmine is given with other cholinomimetic drugs. Rivastigmine may also interfere with the activity of anticholinergic medications. Avoid concomitant use of rivastigmine with drugs having these pharmacologic effects unless deemed clinically necessary.
Use in Specific Populations
Pregnancy
- Pregnancy Category B
- There are no adequate and well-controlled studies in pregnant women. Reproduction studies conducted in pregnant rats and rabbits at oral doses up to 2.3 mg-base/kg/day, or 2 (rat) and 4 (rabbit) times the maximum recommended human dose (MRHD) of 12 mg per day on a mg/m2 basis, revealed no evidence of teratogenicity. Studies in pregnant rats showed slightly decreased fetal/pup weight at doses that were below the MRHD but usually associated with some maternal toxicity. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Rivastigmine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Rivastigmine during labor and delivery.
Nursing Mothers
- Rivastigmine and its metabolites are excreted in rat milk following oral administration of rivastigmine; levels of rivastigmine plus metabolites in rat milk are approximately 2 times that in maternal plasma. It is not known whether rivastigmine is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from Exelon, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
- Safety and effectiveness in pediatric patients have not been established and therefore the use of rivastigmine in children and adolescents (below 18 years of age) is not recommended.
Geriatic Use
- Of the total number of subjects in clinical studies of Exelon, 86 percent were 65 years and older while 46 percent were 75 years and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and other reported clinical experience has not identified differences in responses between the elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Gender
There is no FDA guidance on the use of Rivastigmine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Rivastigmine with respect to specific racial populations.
Renal Impairment
- In patients with moderate to severe renal impairment (glomerular filtration rate [GFR] <50 mL/min), clearance of oral rivastigmine was reduced. Accordingly, such patients may be able to only tolerate lower doses.
Hepatic Impairment
- In patients with mild or moderate hepatic impairment (Child-Pugh score 5 to 9), clearance of oral rivastigmine was reduced. Accordingly, such patients may be able to only tolerate lower doses. No data are available on the use of rivastigmine in patients with severe hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Rivastigmine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Rivastigmine in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
There is limited information regarding Monitoring of Rivastigmine in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Rivastigmine in the drug label.
Overdosage
Acute Overdose
- Because strategies for the management of overdose are continually evolving, it is advisable to contact a Poison Control Center to determine the latest recommendations for the management of an overdose of any drug.
- As rivastigmine has a short plasma half-life of about 1 hour and a moderate duration of acetylcholinesterase inhibition of 8 to 10 hours, it is recommended that in cases of asymptomatic overdoses, no further dose of Exelon should be administered for the next 24 hours.
- As in any case of overdose, general supportive measures should be utilized. Overdosage with cholinesterase inhibitors can result in cholinergic crisis characterized by severe nausea, vomiting, salivation, sweating, bradycardia, hypotension, respiratory depression, collapse and convulsions. Increasing muscle weakness is a possibility and may result in death if respiratory muscles are involved. Atypical responses in blood pressure and heart rate have been reported with other drugs that increase cholinergic activity when coadministered with quaternary anticholinergics such as glycopyrrolate. Due to the short half-life of rivastigmine, dialysis (hemodialysis, peritoneal dialysis, or hemofiltration) would not be clinically indicated in the event of an overdose.
- In overdoses accompanied by severe nausea and vomiting, the use of antiemetics should be considered. In a documented case of a 46 mg overdose with Exelon, the patient experienced vomiting, incontinence, hypertension, psychomotor retardation, and loss of consciousness. The patient fully recovered within 24 hours and conservative management was all that was required for treatment
Chronic Overdose
There is limited information regarding Chronic Overdose of Rivastigmine in the drug label.
Pharmacology
| |
| |
Rivastigmine (oral)
| |
| Systematic (IUPAC) name | |
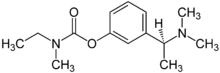
| (S)-3-[1-(dimethylamino)ethyl]phenyl N-ethyl-N-methylcarbamate | |
| Identifiers | |
| CAS number | |
| ATC code | N06 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 250.337 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | 60 to 72% |
| Protein binding | 40% |
| Metabolism | Hepatic, via pseudocholinesterase |
| Half life | 1.5 hours |
| Excretion | Renal, 97% |
| Therapeutic considerations | |
| Pregnancy cat. |
B(US) |
| Legal status |
[[Prescription drug|Template:Unicode-only]](US) |
| Routes | Oral, Transdermal |
Mechanism of Action
- Although the precise mechanism of action of rivastigmine is unknown, it is thought to exert its therapeutic effect by enhancing cholinergic function. This is accomplished by increasing the concentration of acetylcholine through reversible inhibition of its hydrolysis by cholinesterase. Therefore, the effect of rivastigmine may lessen as the disease process advances and fewer cholinergic neurons remain functionally intact. There is no evidence that rivastigmine alters the course of the underlying dementing process.
Structure
- Exelon (rivastigmine tartrate) is a reversible cholinesterase inhibitor and is known chemically as (S)-N-Ethyl-N-methyl-3-[1-(dimethylamino)ethyl]-phenyl carbamate hydrogen-(2R,3R)-tartrate. Rivastigmine tartrate is commonly referred to in the pharmacological literature as SDZ ENA 713 or ENA 713. It has an empirical formula of C14H22N2O2 • C4H6O6 (hydrogen tartrate salt-hta salt) and a molecular weight of 400.43 (hta salt). Rivastigmine tartrate is a white to off-white, fine crystalline powder that is very soluble in water, soluble in ethanol and acetonitrile, slightly soluble in n-octanol and very slightly soluble in ethyl acetate.
- The distribution coefficient at 37°C in n-octanol/phosphate buffer solution pH 7 is 3.0.
- Exelon Capsules contain rivastigmine tartrate, equivalent to 1.5 mg, 3 mg, 4.5 mg, and 6 mg of rivastigmine base for oral administration. Inactive ingredients are hydroxypropyl methylcellulose, magnesium stearate, microcrystalline cellulose, and silicon dioxide. Each hard-gelatin capsule contains gelatin, titanium dioxide and red and/or yellow iron oxides.
- Exelon Oral Solution is supplied as a solution containing rivastigmine tartrate, equivalent to 2 mg/mL of rivastigmine base for oral administration. Inactive ingredients are citric acid, D&C yellow #10, purified water, sodium benzoate and sodium citrate.
Pharmacodynamics
- After a 6-mg dose of rivastigmine, anticholinesterase activity is present in cerebrospinal fluid (CSF) for about 10 hours, with a maximum inhibition of about 60% 5 hours after dosing.
- In vitro and in vivo studies demonstrate that the inhibition of cholinesterase by rivastigmine is not affected by the concomitant administration of memantine, an N-methyl-D-aspartate receptor antagonist.
Pharmacokinetics
- Rivastigmine shows linear pharmacokinetics up to 3 mg twice a day but is nonlinear at higher doses. Doubling the dose from 3 mg to 6 mg twice a day results in a 3-fold increase in AUC. The elimination half-life is about 1.5 hours, with most elimination as metabolites via the urine.
- Absorption
- Rivastigmine is rapidly and completely absorbed. Peak plasma concentrations are reached in approximately 1 hour. Absolute bioavailability after a 3-mg dose is about 36%. Administration of Exelon with food delays absorption (tmax) by 90 minutes lowers Cmax by approximately 30% and increases AUC by approximately 30%.
- Distribution
- Rivastigmine is weakly bound to plasma proteins (approximately 40%) over the therapeutic range. It readily crosses the blood-brain barrier, reaching CSF peak concentrations in 1.4 to 2.6 hours. It has an apparent volume of distribution in the range of 1.8 to 2.7 L/kg.
- Metabolism
- Rivastigmine is rapidly and extensively metabolized, primarily via cholinesterase-mediated hydrolysis to the decarbamylated metabolite. Based on evidence from in vitro and animal studies, the major cytochrome P450 isozymes are minimally involved in rivastigmine metabolism. Consistent with these observations is the finding that no drug interactions related to cytochrome P450 have been observed in humans.
- Elimination
- The major pathway of elimination is via the kidneys. Following administration of 14C-rivastigmine to 6 healthy volunteers, total recovery of radioactivity over 120 hours was 97% in urine and 0.4% in feces. No parent drug was detected in urine. The sulfate conjugate of the decarbamylated metabolite is the major component excreted in urine and represents 40% of the dose. Mean oral clearance of rivastigmine is 1.8 ± 0.6 L/min after 6 mg twice a day.
- Renal Impairment
- Following a single 3-mg dose, mean oral clearance of rivastigmine is 64% lower in moderately impaired renal patients (n=8, GFR=10 to 50 mL/min) than in healthy subjects (n=10, GFR≥60 mL/min); Cl/F=1.7 L/min and 4.8 L/min, respectively. In patients with severe renal impairment (n=8, GFR <10 mL/min), mean oral clearance of rivastigmine is 43% higher than in healthy subjects (n=10, GFR ≥60 mL/min); Cl/F=6.9 L/min and 4.8 L/min, respectively. For unexplained reasons, the severely impaired renal patients had a higher clearance of rivastigmine than moderately impaired patients. Despite this finding, such patients may be able to only tolerate lower doses.
- Hepatic Impairment
- Following a single 3-mg dose, mean oral clearance of rivastigmine was 60% lower in hepatically impaired patients (n=10, biopsy proven) than in healthy subjects (n=10). After multiple 6-mg twice a day oral dosing, the mean clearance of rivastigmine was 65% lower in mild (n=7, Child-Pugh score 5 to 6) and moderate (n=3, Child-Pugh score 7 to 9) hepatically impaired patients (biopsy proven, liver cirrhosis) than in healthy subjects (n=10). Accordingly, such patients may be able to only tolerate lower doses.
- Body Weight
- A relationship between drug exposure at steady-state (rivastigmine and metabolite NAP226-90) and body weight was observed in Alzheimer’s dementia patients. Rivastigmine exposure is higher in subjects with low body weight. Compared to a patient with a body weight of 65 kg, the rivastigmine steady-state concentrations in a patient with a body weight of 35 kg would be approximately doubled, while for a patient with a body weight of 100 kg the concentrations would be approximately halved.
- Age
- Following a single 2.5-mg oral dose to elderly volunteers (60 years and older, n=24) and younger volunteers (n=24), mean oral clearance of rivastigmine was 30% lower in elderly (7 L/min) than in younger subjects (10 L/min).
- Gender or Race
- Population pharmacokinetic analysis of oral rivastigmine indicated that neither gender (n=277 males and 348 females) nor race (n=575 Caucasian, 34 Black, 4 Asian, and 12 Other) affected clearance of the drug.
- Smoking
- Population pharmacokinetic analysis showed that nicotine use increased the oral clearance of rivastigmine by 23% (n=75 smokers and 549 nonsmokers).
- Drug Interaction Studies
- Effect of Rivastigmine on the Metabolism of Other Drugs
- Rivastigmine is primarily metabolized through hydrolysis by esterases. Minimal metabolism occurs via the major cytochrome P450 isoenzymes. Based on in vitro studies, no pharmacokinetic drug interactions with drugs metabolized by the following isoenzyme systems are expected: CYP1A2, CYP2D6, CYP3A4/5, CYP2E1, CYP2C9, CYP2C8, CYP2C19, or CYP2B6.
- No pharmacokinetic interaction was observed between rivastigmine taken orally and digoxin, warfarin, diazepam or fluoxetine in studies in healthy volunteers. The increase in prothrombin time induced by warfarin is not affected by administration of rivastigmine.
- Effect of Other Drugs on the Metabolism of Rivastigmine
- Drugs that induce or inhibit CYP450 metabolism are not expected to alter the metabolism of rivastigmine.
- Population pharmacokinetic analysis with a database of 625 patients showed that the pharmacokinetics of rivastigmine taken orally were not influenced by commonly prescribed medications such as antacids (n=77), antihypertensives (n=72), ß-blockers (n=42), calcium channel blockers (n=75), antidiabetics (n=21), NSAIDs (n=79), estrogens (n=70), salicylate analgesics (n=177), antianginals (n=35) and antihistamines (n=15).
Nonclinical Toxicology
- Carcinogenesis
- In oral carcinogenicity studies conducted at doses up to 1.1 mg-base/kg/day in rats and 1.6 mg-base/kg/day in mice, rivastigmine was not carcinogenic. These doses are less than the maximum recommended human dose (MRHD) of 12 mg per day on a mg/m2 basis.
- Mutagenesis
- Rivastigmine was clastogenic in in vitro chromosomal aberration assays in mammalian cells in the presence, but not the absence, of metabolic activation. Rivastigmine was negative in an in vitro bacterial reverse mutation (Ames) assay, an in vitro HGPRT assay, and in an in vivo mouse micronucleus test.
- Impairment of Fertility
- Rivastigmine had no effect on fertility or reproductive performance in rats at oral doses up to 1.1 mg-base/kg/day, a dose less than the MRHD on a mg/m2 basis.
Clinical Studies
Mild to Moderate Alzheimer’s Disease
- The effectiveness of Exelon as a treatment for Alzheimer's disease is demonstrated by the results of 2 randomized, double-blind, placebo-controlled clinical investigations (Study 1 and Study 2) in patients with Alzheimer's disease [diagnosed by NINCDS-ADRDA and DSM-IV criteria, Mini-Mental State Examination (MMSE) ≥10 and ≤26, and the Global Deterioration Scale (GDS)]. The mean age of patients participating in Exelon trials was 73 years with a range of 41 to 95. Approximately 59% of patients were women and 41% were men. The racial distribution was Caucasian 87%, Black 4%, and other races 9%.
- In each study, the effectiveness of Exelon was evaluated using a dual outcome assessment strategy.
- The ability of Exelon to improve cognitive performance was assessed with the cognitive subscale of the Alzheimer's Disease Assessment Scale (ADAS-cog), a multi-item instrument that has been extensively validated in longitudinal cohorts of Alzheimer's disease patients. The ADAS-cog examines selected aspects of cognitive performance including elements of memory, orientation, attention, reasoning, language, and praxis. The ADAS-cog scoring range is from 0 to 70, with higher scores indicating greater cognitive impairment. Elderly normal adults may score as low as 0 or 1, but it is not unusual for non-demented adults to score slightly higher.
- The patients recruited as participants in each study had mean scores on ADAS-cog of approximately 23 units, with a range from 1 to 61. Experience gained in longitudinal studies of ambulatory patients with mild to moderate Alzheimer's disease suggests that they gain 6 to 12 units a year on the ADAS-cog. Lesser degrees of change, however, are seen in patients with very mild or very advanced disease because the ADAS-cog is not uniformly sensitive to change over the course of the disease. The annualized rate of decline in the placebo patients participating in Exelon trials was approximately 3 to 8 units per year.
- The ability of Exelon to produce an overall clinical effect was assessed using a Clinician's Interview-Based Impression of Change (CIBIC) that required the use of caregiver information, the CIBIC-Plus. The CIBIC-Plus is not a single instrument and is not a standardized instrument like the ADAS-cog. Clinical trials for investigational drugs have used a variety of CIBIC formats, each different in terms of depth and structure. As such, results from a CIBIC-Plus reflect clinical experience from the trial or trials in which it was used and cannot be compared directly with the results of CIBIC-Plus evaluations from other clinical trials. The CIBIC-Plus used in the Exelon trials was a structured instrument based on a comprehensive evaluation at baseline and subsequent time-points of 3 domains: patient cognition, behavior and functioning, including assessment of activities of daily living. It represents the assessment of a skilled clinician using validated scales based on his/her observation at interviews conducted separately with the patient and the caregiver familiar with the behavior of the patient over the interval rated. The CIBIC-Plus is scored as a 7-point categorical rating, ranging from a score of 1, indicating "markedly improved," to a score of 4, indicating "no change" to a score of 7, indicating "marked worsening." The CIBIC-Plus has not been systematically compared directly to assessments not using information from caregivers or other global methods.
- U.S. 26-Week Study of Exelon in Mild to Moderate Alzheimer’s Disease (Study 1)
- In a study of 26 weeks duration, 699 patients were randomized to either a dose range of 1 mg to 4 mg or 6 mg to 12 mg of Exelon per day or to placebo, each given in divided doses. The 26-week study was divided into a 12-week forced-dose titration phase and a 14-week maintenance phase. The patients in the active treatment arms of the study were maintained at their highest tolerated dose within the respective range.
- Figure 1 illustrates the time course for the change from baseline in ADAS-cog scores for all 3 dose groups over the 26 weeks of the study. At 26 weeks of treatment, the mean differences in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo were 1.9 and 4.9 units for the 1 mg to 4 mg and 6 mg to 12 mg treatments, respectively. Both treatments were statistically significantly superior to placebo and the 6 mg to 12 mg per day range was significantly superior to the 1 mg to 4 mg per day range.
- Figure 2 illustrates the cumulative percentages of patients from each of the 3 treatment groups who had attained at least the measure of improvement in ADAS-cog score shown on the X-axis. Three change scores, (7-point and 4-point reductions from baseline or no change in score) have been identified for illustrative purposes, and the percent of patients in each group achieving that result is shown in the inset table.
- The curves demonstrate that both patients assigned to Exelon and placebo have a wide range of responses, but that the Exelon groups are more likely to show the greater improvements. A curve for an effective treatment would be shifted to the left of the curve for placebo, while an ineffective or deleterious treatment would be superimposed upon, or shifted to the right of the curve for placebo, respectively.
- Figure 3 is a histogram of the frequency distribution of CIBIC-Plus scores attained by patients assigned to each of the 3 treatment groups who completed 26 weeks of treatment. The mean Exelon-placebo differences for these groups of patients in the mean rating of change from baseline were 0.32 units and 0.35 units for 1 mg to 4 mg and 6 mg to 12 mg of Exelon, respectively. The mean ratings for the 6 mg to 12 mg per day and 1 mg to 4 mg per day groups were statistically significantly superior to placebo. The differences between the 6 mg to 12 mg per day and the 1 mg to 4 mg per day groups were statistically significant.
- Global 26-Week Study in Mild to Moderate Alzheimer’s Disease (Study 2)
- In a second study of 26 weeks duration, 725 patients were randomized to either a dose range of 1 mg to 4 mg or 6 mg to 12 mg of Exelon per day or to placebo, each given in divided doses. The 26-week study was divided into a 12-week forced-dose titration phase and a 14-week maintenance phase. The patients in the active treatment arms of the study were maintained at their highest tolerated dose within the respective range.
- Figure 4 illustrates the time course for the change from baseline in ADAS-cog scores for all 3 dose groups over the 26 weeks of the study. At 26 weeks of treatment, the mean differences in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo were 0.2 and 2.6 units for the 1 mg to 4 mg and 6 mg to 12 mg treatments, respectively. The 6 mg to 12 mg per day group was statistically significantly superior to placebo, as well as to the 1 mg to 4 mg per day group. The difference between the 1 mg to 4 mg per day group and placebo was not statistically significant.
- Figure 5 illustrates the cumulative percentages of patients from each of the 3 treatment groups who had attained at least the measure of improvement in ADAS-cog score shown on the X-axis. Similar to the U.S. 26-week study, the curves demonstrate that both patients assigned to Exelon and placebo have a wide range of responses, but that the 6 mg to 12 mg per day Exelon group is more likely to show the greater improvements.
- Figure 6 is a histogram of the frequency distribution of CIBIC-Plus scores attained by patients assigned to each of the 3 treatment groups who completed 26 weeks of treatment. The mean Exelon-placebo differences for these groups of patients for the mean rating of change from baseline were 0.14 units and 0.41 units for 1 mg to 4 mg and 6 mg to 12 mg of Exelon, respectively. The mean ratings for the 6 mg to 12 mg per day group were statistically significantly superior to placebo. The comparison of the mean ratings for the 1 mg to 4 mg per day group and placebo group was not statistically significant.
- U.S. Fixed-Dose Study in Mild to Moderate Alzheimer’s Disease (Study 3)
- In a study of 26 weeks duration, 702 patients were randomized to doses of 3 mg, 6 mg, or 9 mg per day of Exelon or to placebo, each given in divided doses. The fixed-dose study design, which included a 12-week forced-dose titration phase and a 14-week maintenance phase, led to a high dropout rate in the 9 mg per day group because of poor tolerability. At 26 weeks of treatment, significant differences were observed for the ADAS-cog mean change from baseline for the 9 mg per day and 6 mg per day groups, compared to placebo. No significant differences were observed between any of the Exelon-dose groups and placebo for the analysis of the CIBIC-Plus mean rating of change. Although no significant differences were observed between Exelon treatment groups, there was a trend toward numerical superiority with higher doses.
Mild to Moderate Parkinson’s Disease Dementia
- International 24-Week Study (Study 4)
- The effectiveness of Exelon as a treatment for dementia associated with Parkinson’s disease is demonstrated by the results of 1 randomized, double-blind, placebo-controlled clinical investigation in patients with mild to moderate dementia, with onset at least 2 years after the initial diagnosis of idiopathic Parkinson’s disease. The diagnosis of idiopathic Parkinson’s disease was based on the United Kingdom Parkinson’s Disease Society Brain Bank clinical criteria. The diagnosis of dementia was based on the criteria stipulated under the DSM-IV category “Dementia Due To Other General Medical Condition” (code 294.1x), but patients were not required to have a distinctive pattern of cognitive deficits as part of the dementia. Alternate causes of dementia were excluded by clinical history, physical and neurological examination, brain imaging, and relevant blood tests. Patients enrolled in the study had a MMSE score ≥10 and ≤24 at entry. The mean age of patients participating in this trial was 72.7 years with a range of 50–91 years. Approximately, 35.1% of patients were women and 64.9% of patients were men. The racial distribution was 99.6% Caucasian and other races 0.4%.
- This study used a dual outcome assessment strategy to evaluate the effectiveness of Exelon.
- The ability of Exelon to improve cognitive performance was assessed with the ADAS-cog.
- The ability of Exelon to produce an overall clinical effect was assessed using the Alzheimer’s Disease Cooperative Study – Clinician’s Global Impression of Change (ADCS-CGIC). The ADCS-CGIC is a more standardized form of CIBIC-Plus and is also scored as a 7-point categorical rating, ranging from a score of 1, indicating "markedly improved," to a score of 4, indicating "no change" to a score of 7, indicating "marked worsening".
- In this study, 541 patients were randomized to a dose range of 3 mg to 12 mg of Exelon per day or to placebo in a ratio of 2:1, given in divided doses. The 24-week study was divided into a 16-week titration phase and an 8-week maintenance phase. The patients in the active treatment arm of the study were maintained at their highest tolerated dose within the specified dose range.
- Figure 7 illustrates the time course for the change from baseline in ADAS-cog scores for both treatment groups over the 24-week study. At 24 weeks of treatment, the mean difference in the ADAS-cog change scores for the Exelon-treated patients compared to the patients on placebo was 3.8 points. This treatment difference was statistically significant in favor of Exelon when compared to placebo.
- Figure 8 is a histogram of the distribution of patients’ scores on the ADCS-CGIC (Alzheimer’s Disease Cooperative Study - Clinician’s Global Impression of Change) at 24 weeks. The mean difference in change scores between the Exelon and placebo groups from baseline was 0.5 points. This difference was statistically significant in favor of Exelon treatment.
- Patients’ age, gender, or race did not predict clinical outcome of Exelon treatment.
How Supplied
- Exelon Capsules
- Exelon (rivastigmine tartrate) Capsules equivalent to 1.5 mg, 3 mg, 4.5 mg, or 6 mg of rivastigmine base are available as follows:
- 1.5 mg capsule – yellow, “Exelon 1,5 mg” is printed in red on the body of the capsule.
- Bottles of 60 NDC 0078-0323-44
- Bottles of 500 NDC 0078-0323-08
- Unit Dose (blister pack) Box of 100 (strips of 10) NDC 0078-0323-06
- 3 mg capsule – orange, “Exelon 3 mg” is printed in red on the body of the capsule.
- Bottles of 60 NDC 0078-0324-44
- Bottles of 500 NDC 0078-0324-08
- Unit Dose (blister pack) Box of 100 (strips of 10) NDC 0078-0324-06
- 4.5 mg capsule – red, “Exelon 4,5 mg” is printed in white on the body of the capsule.
- Bottles of 60 NDC 0078-0325-44
- Bottles of 500 NDC 0078-0325-08
- Unit Dose (blister pack) Box of 100 (strips of 10) NDC 0078-0325-06
- 6 mg capsule – orange and red, “Exelon 6 mg” is printed in red on the body of the capsule.
- Bottles of 60 NDC 0078-0326-44
- Bottles of 500 NDC 0078-0326-08
- Unit Dose (blister pack) Box of 100 (strips of 10) NDC 0078-0326-06
- Store at 25°C (77°F); excursions permitted to 15°C-30°C (59°F-86°F). Store in a tight container.
- Exelon Oral Solution
- Exelon (rivastigmine tartrate) Oral Solution is supplied as 120 mL of a clear, yellow solution (2 mg/mL base) in a 4-ounce USP Type III amber glass bottle with a child-resistant 19-mm linerless cap, dip tube and self-aligning plug. The oral solution is packaged with a dispenser set which consists of an assembled oral dosing syringe that allows dispensing a maximum volume of 3 mL corresponding to a 6-mg dose, with a plastic tube container.
- Bottles of 120 mL NDC 0078-0339-31
- Store at 25°C (77°F); excursions permitted to 15°C-30°C (59°F-86°F). Store in an upright position and protect from freezing.
- When Exelon Oral Solution is combined with cold fruit juice or soda, the mixture is stable at room temperature for up to 4 hours.
Storage
There is limited information regarding Rivastigmine (oral) Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Rivastigmine (oral) |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Rivastigmine (oral) |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Gastrointestinal Adverse Reactions
- Caregivers should be advised of the high incidence of nausea and vomiting associated with the use of the drug along with the possibility of anorexia and weight loss. Caregivers should be encouraged to monitor for these adverse events and inform the physician if they occur. It is critical to inform caregivers that if therapy has been interrupted for more than several days, the next dose should not be administered until they have discussed this with the physician.
- Skin Reactions
- Caregivers and patients should be advised that allergic skin reactions have been reported in association with Exelon regardless of formulation (capsules, oral solution or transdermal patch). In case of skin reaction while taking Exelon, patients should consult with their physician immediately.
- Importance of Correct Usage
- Caregivers should be instructed in the correct procedure for administering Exelon Oral Solution. In addition, they should be informed of the existence of an Instruction Sheet (included with the product) describing how the solution is to be administered. They should be urged to read this sheet prior to administering Exelon Oral Solution. Caregivers should direct questions about the administration of the solution to either their physician or pharmacist.
- Concomitant Use of Drugs with Cholinergic Action
- Caregivers and patients should be advised that cholinomimetics, including rivastigmine, may exacerbate or induce extrapyramidal symptoms. Worsening in patients with Parkinson’s disease, including an increased incidence or intensity of tremor, has been observed.
Precautions with Alcohol
- Alcohol-Rivastigmine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- EXELON®[1]
Look-Alike Drug Names
There is limited information regarding Rivastigmine (oral) Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Rivastigmine (oral) |Pill Name=No image.jpg |Drug Name= |Pill Ingred=|+sep=; |Pill Imprint= |Pill Dosage= |Pill Color=|+sep=; |Pill Shape= |Pill Size (mm)= |Pill Scoring= |Pill Image= |Drug Author= |NDC=
}}
{{#subobject:
|Label Page=Rivastigmine (oral) |Label Name=Rivastigmine13.png
}}
{{#subobject:
|Label Page=Rivastigmine (oral) |Label Name=Rivastigmine14.png
}}
{{#subobject:
|Label Page=Rivastigmine (oral) |Label Name=Rivastigmine15.png
}}
{{#subobject:
|Label Page=Rivastigmine (oral) |Label Name=Rivastigmine16.png
}}
{{#subobject:
|Label Page=Rivastigmine (oral) |Label Name=Rivastigmine17.png
}}
{{#subobject:
|Label Page=Rivastigmine (oral) |Label Name=Rivastigmine18.png
}}