Sulfonic acid
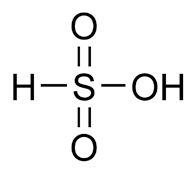
Sulfonic acid is a hypothetical acid with formula H-S(=O)2-OH. This compound is a less stable tautomer of sulfurous acid HO-S(=O)-OH, so sulfonic acid converts rapidly when it is formed. Derived compounds which replace the sulfur-bonded hydrogen with organic groups are stable. These may then form salts or esters, called sulfonates.
Sulfonic acids

Sulfonic acids are a class of organic acids with the general formula R-S(=O)2-OH, where R is usually a hydrocarbon side chain. Sulfonic acids are typically much stronger acids than their carboxylic equivalents, and have the unique tendency to bind to proteins and carbohydrates tightly; most "washable" dyes are sulfonic acids (or have the functional sulfonyl group in them) for this reason. They are also used as catalysts and intermediates for a number of different products. Sulfonic acid salts (sulfonates) are important as detergents, and the antibacterial sulfa drugs are also sulfonic acid derivatives. The simplest example is methanesulfonic acid, CH3SO2OH, which is a reagent regularly used in organic chemistry. p-Toluenesulfonic acid is also an important reagent.
Note that the sulfonic acids and sulfonates are analogous to carboxylic acids and carboxylates; in both cases, -C(=O)- is replaced by -S(=O)2-. Chemical properties are similar as well, although sulfonic acids are often even stronger acids than carboxylic acids, the hydrogen being easier to leave than in most compounds, and they readily form esters.
The sulfonic acid and sulfonate functional groups, -SO2OH and -SO2O-, are found in many chemical compounds, e.g. certain detergents and dyes as well as in strongly acidic cation exchange resins.
Sulfonic acid chlorides
Sulfonic acid chlorides are a class of organic compounds with the general formula R-SO2-Cl. These compounds react readily with alcohols to sulfonic esters. Important acid chlorides are tosyl chloride, brosyl chloride, nosyl chloride and mesyl chloride. One synthetic procedure to synthesize sulfonic acid chlorides is the Reed reaction.
Sulfonic esters
Sulfonic esters are a class of organic compounds with the general formula R-SO2-OR. Sulfonic esters are considered good leaving groups in nucleophilic aliphatic substitution.
See also
- Sulfuric acid H2SO4
- Sulfonium S+, S+R3