Tautomer
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Tautomers are organic compounds that are interconvertible by a chemical reaction called tautomerization. As most commonly encountered, this reaction results in the formal migration of a hydrogen atom or proton, accompanied by a switch of a single bond and adjacent double bond. In solutions where tautomerization is possible, a chemical equilibrium of the tautomers will be reached. The exact ratio of the tautomers depends on several factors, including temperature, solvent, and pH. The concept of tautomers that are interconvertible by tautomerizations is called tautomerism. Tautomerism is a special case of structural isomerism and can play an important role in non-canonical base pairing in DNA and especially RNA molecules.
Tautomerizations are catalyzed by:
- base (1. deprotonation; 2. formation of a delocalized anion (e.g. an enolate); 3. protonation at a different position of the anion).
- acids (1. protonation; 2. formation of a delocalized cation; 3. deprotonation at a different position adjacent to the cation).
Common tautomeric pairs are:
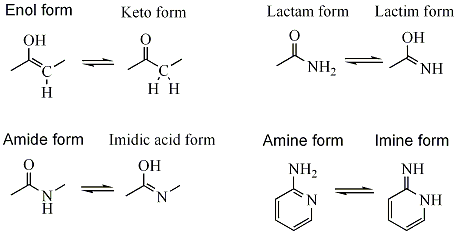
- ketone - enol, e.g. for acetone (see: keto-enol tautomerism).
- amide - imidic acid, e.g. during nitrile hydrolysis reactions.
- lactam - lactim, an amide - imidic acid tautomerism in heterocyclic rings, e.g. in the nucleobases guanine, thymine, and cytosine.
- enamine - imine
- enamine - enamine, e.g. during pyridoxalphosphate catalyzed enzymatic reactions.
Prototropic tautomerism refers to the relocation of a proton, as in the above examples, and may be considered a subset of acid-base behavior. Prototropic tautomers are sets of isomeric protonation states with the same empirical formula and total charge.
Annular tautomerism is a type of prototropic tautomerism where a proton can occupy two or more positions of a heterocyclic system. for example, 1H- and 3H-imidazole; 1H-, 2H- and 4H- 1,2,4-triazole; 1H- and 2H- isoindole.
Ring-chain tautomerism occurs when the movement of the proton is accompanied by a change from an open structure to a ring, such as the aldehyde and pyran forms of glucose.
Valence tautomerism is distinct from prototropic tautomerism, and involves processes with rapid reorganisation of bonding electrons. An example of this type of tautomerism can be found in bullvalene. Another example is open and closed forms of certain heterocycles, such as azide - tetrazole. Valence tautomerism requires a change in molecular geometry and should not be confused with canonical resonance structures or mesomers.
References
- Smith, M. B.; March, J. Advanced Organic Chemistry, 5th ed., Wiley Interscience, New York, 2001; pp 1218-1223. ISBN 0471585890
- Katritzky, A.R.; Elguero, J. et al. The Tautomerism of heterocycles, Academic Press, New York, 1976, ISBN 012020651X
de:Tautomerie el:Ταυτομερή it:Tautomeria nl:Tautomeer sv:Tautomeri