Solifenacin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sophia Saad, Associate Editor - WikiDoc [2]; Adeel Jamil, M.D. [3]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Solifenacin is a muscarinic receptor antagonist that is FDA approved for the treatment of overactive bladder with symptoms of urge urinary incontinence, urgency, and urinary frequency. Common adverse reactions include constipation, Xerostomia.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Dosing information
- Recommended dosage: 5 mg PO qd.
- If the 5 mg dose is well tolerated, the dose may be increased to 10 mg once daily.
- Solifenacin should be taken with water and swallowed whole. Solifenacin can be administered with or without food.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Solifenacin in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Solifenacin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
The safety and effectiveness of Solifenacin in pediatric patients have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Solifenacin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Solifenacin in pediatric patients.
Contraindications
Solifenacin is contraindicated in patients with:
- in patients who have demonstrated hypersensitivity to the drug
Warnings
Angioedema and Anaphylactic Reactions=
Angioedema of the face, lips, tongue, and/or larynx have been reported with solifenacin. In some cases angioedema occurred after the first dose. Cases of angioedema have been reported to occur hours after the first dose or after multiple doses. Angioedema associated with upper airway swelling may be life threatening. If involvement of the tongue, hypopharynx, or larynx occurs, solifenacin should be promptly discontinued and appropriate therapy and/or measures necessary to ensure a patent airway should be promptly provided. Anaphylactic reactions have been reported rarely in patients treated with solifenacin succinate. Solifenacin succinate should not be used in patients with a known or suspected hypersensitivity to solifenacin succinate. In patients who develop anaphylactic reactions, solifenacin succinate should be discontinued and appropriate therapy and/or measures should be taken.
Urinary Retention
Solifenacin, like other anticholinergic drugs, should be administered with caution to patients with clinically significant bladder outflow obstruction because of the risk of urinary retention.
Gastrointestinal Disorders
Solifenacin, like other anticholinergics, should be used with caution in patients with decreased gastrointestinal motility.
Central Nervous System Effects
Solifenacin is associated with anticholinergic central nervous system (CNS) effects. A variety of CNS anticholinergic effects have been reported, including headache, confusion, hallucinations and somnolence. Patients should be monitored for signs of anticholinergic CNS effects, particularly after beginning treatment or increasing the dose. Advise patients not to drive or operate heavy machinery until they know how Solifenacin affects them. If a patient experiences anticholinergic CNS effects, dose reduction or drug discontinuation should be considered.
Controlled Narrow-Angle Glaucoma
Solifenacin should be used with caution in patients being treated for narrow-angle glaucoma.
Hepatic Impairment
Solifenacin should be used with caution in patients with hepatic impairment. Doses of Solifenacin greater than 5 mg are not recommended in patients with moderate hepatic impairment (Child-Pugh
Adverse Reactions
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. Solifenacin has been evaluated for safety in 1811 patients in randomized, placebo-controlled trials. Expected adverse reactions of antimuscarinic agents are dry mouth, constipation, blurred vision (accommodation abnormalities), urinary retention, and dry eyes. The incidence of dry mouth and constipation in patients treated with Solifenacin was higher in the 10 mg compared to the 5 mg dose group. In the four 12-week double-blind clinical trials, severe fecal impaction, colonic obstruction, and intestinal obstruction were reported in one patient each, all in the Solifenacin 10 mg group. Angioneurotic edema has been reported in one patient taking Solifenacin 5 mg. Compared to 12 weeks of treatment with Solifenacin, the incidence and severity of adverse reactions were similar in patients who remained on drug for up to 12 months. The most frequent adverse reaction leading to study discontinuation was dry mouth (1.5%). Table 1 lists the rates of identified adverse reactions, derived from all reported adverse events, in randomized, placebo-controlled trials at an incidence greater than placebo and in 1% or more of patients treated with Solifenacin 5 or 10 mg once daily for up to 12 weeks.
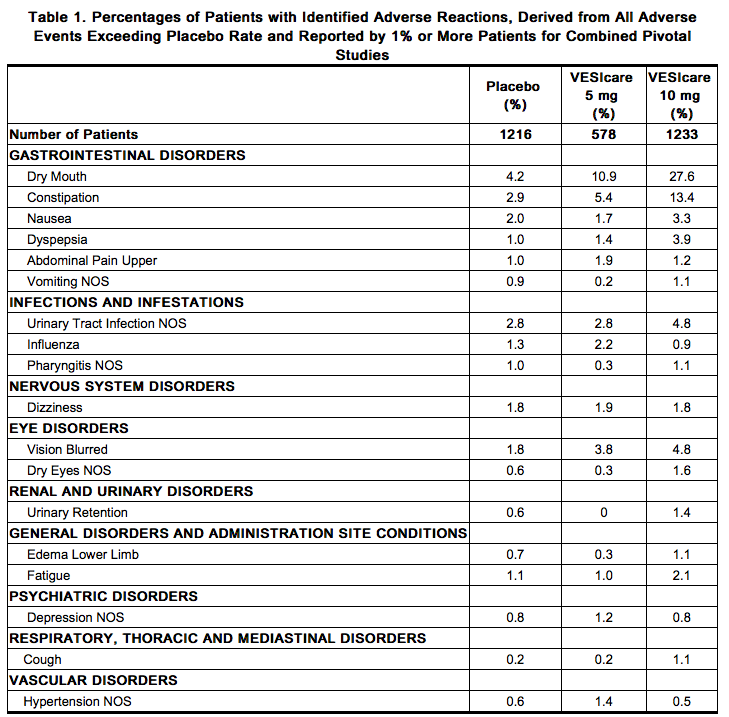
Postmarketing Experience
Because these spontaneously reported events are from the worldwide postmarketing experience, the frequency of events and the role of solifenacin in their causation cannot be reliably determined. The following events have been reported in association with solifenacin use in worldwide postmarketing experience:
General: peripheral edema, hypersensitivity reactions, including angioedema with airway obstruction, rash, pruritus, urticaria, and anaphylactic reaction; Central Nervous: headache, confusion, hallucinations, delirium and somnolence; Cardiovascular: QT prolongation; Torsade de Pointes, atrial fibrillation, tachycardia, palpitations; Hepatic: liver disorders mostly characterized by abnormal liver function tests, AST (aspartate aminotransferase), ALT (alanine aminotransferase), GGT (gamma-glutamyl transferase); Renal: renal impairment; Metabolism and nutrition disorders: decreased appetite, hyperkalemia; Dermatologic: exfoliative dermatitis and erythema multiforme; Eye disorders:glaucoma; Gastrointestinal disorders:gastroesophageal reflux disease and ileus; Respiratory, thoracic and mediastinal disorders: dysphonia; Musculoskeletal and connective tissue disorders: muscular weakness;
Drug Interactions
Potent CYP3A4 Inhibitors
Following the administration of 10 mg of Solifenacin in the presence of 400 mg of ketoconazole, a potent inhibitor of CYP3A4, the mean Cmax and AUC of solifenacin increased by 1.5 and 2.7-fold, respectively. Therefore, it is recommended not to exceed a 5 mg daily dose of Solifenacin when administered with therapeutic doses of ketoconazole or other potent CYP3A4 inhibitors . The effects of weak or moderate CYP3A4 inhibitors were not examined.
CYP3A4 Inducers
There were no in vivo studies conducted to evaluate the effect of CYP3A4 inducers on Solifenacin. In vitro drug metabolism studies have shown that solifenacin is a substrate of CYP3A4. Therefore, inducers of CYP3A4 may decrease the concentration of solifenacin.
Drugs Metabolized by Cytochrome P450
At therapeutic concentrations, solifenacin does not inhibit CYP1A1/2, 2C9, 2C19, 2D6, or 3A4 derived from human liver microsomes.
Warfarin
Solifenacin has no significant effect on the pharmacokinetics of R-warfarin or S-warfarin.
Oral Contraceptives
In the presence of solifenacin there are no significant changes in the plasma concentrations of combined oral contraceptives (ethinyl estradiol/levonorgestrel).
Digoxin
Solifenacin had no significant effect on the pharmacokinetics of digoxin (0.125 mg/day) in healthy subjects
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C
There are no adequate and well-controlled studies in pregnant women.
Reproduction studies have been performed in mice, rats and rabbits. After oral administration of 14C-solifenacin succinate to pregnant mice, drug-related material was shown to cross the placental barrier. No embryotoxicity or teratogenicity was observed in mice treated with 1.2 times (30 mg/kg/day) the expected exposure at the maximum recommended human dose [MRHD] of 10 mg. Administration of solifenacin succinate to pregnant mice at 3.6 times and greater (100 mg/kg/day and greater) the exposure at the MRHD, during the major period of organ development resulted in reduced fetal body weights. Administration of 7.9 times (250 mg/kg/day) the MRHD to pregnant mice resulted in an increased incidence of cleft palate. In utero and lactational exposures to maternal doses of solifenacin succinate of 3.6 times (100 mg/kg/day) the MRHD resulted in reduced peripartum and postnatal survival, reductions in body weight gain, and delayed physical development (eye opening and vaginal patency). An increase in the percentage of male offspring was also observed in litters from offspring exposed to maternal doses of 250 mg/kg/day. No embryotoxic effects were observed in rats at up to 50 mg/kg/day (<1 times the exposure at the MRHD) or in rabbits at up to 1.8 times (50 mg/kg/day) the exposure at the MRHD. Because animal reproduction studies are not always predictive of human response, Solifenacin should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Solifenacin in women who are pregnant.
Labor and Delivery
The effect of Solifenacin on labor and delivery in humans has not been studied. There were no effects on natural delivery in mice treated with 1.2 times (30 mg/kg/day) the expected exposure at the maximum recommended human dose [MRHD] of 10 mg. Administration of solifenacin succinate at 3.6 times (100 mg/kg/day) the exposure at the MRHD or greater increased peripartum pup mortality.
Nursing Mothers
After oral administration of 14C-solifenacin succinate to lactating mice, radioactivity was detected in maternal milk. There were no adverse observations in mice treated with 1.2 times (30 mg/kg/day) the expected exposure at the maximum recommended human dose [MRHD]. Pups of female mice treated with 3.6 times (100 mg/kg/day) the exposure at the MRHD or greater revealed reduced body weights, postpartum pup mortality or delays in the onset of reflex and physical development during the lactation period. It is not known whether solifenacin is excreted in human milk. Because many drugs are excreted in human milk, Solifenacin should not be administered during nursing. A decision should be made whether to discontinue nursing or to discontinue Solifenacin in nursing mothers.
Pediatric Use
The safety and effectiveness of Solifenacin in pediatric patients have not been established.
Geriatic Use
In placebo-controlled clinical studies, similar safety and effectiveness were observed between older (623 patients ≥ 65 years and 189 patients ≥ 75 years) and younger patients (1188 patients < 65 years) treated with Solifenacin. Multiple dose studies of Solifenacin in elderly volunteers (65 to 80 years) showed that Cmax, AUC and t1/2 values were 20-25% higher as compared to the younger volunteers (18 to 55 years).
Gender
The pharmacokinetics of solifenacin is not significantly influenced by gender.
Race
There is no FDA guidance on the use of Solifenacin with respect to specific racial populations.
Renal Impairment
Solifenacin should be used with caution in patients with renal impairment. There is a 2.1-fold increase in AUC and 1.6-fold increase in t1/2 of solifenacin in patients with severe renal impairment. Doses of Solifenacin greater than 5 mg are not recommended in patients with severe renal impairment (CLcr < 30 mL/min)
Hepatic Impairment
Solifenacin should be used with caution in patients with reduced hepatic function. There is a 2-fold increase in the t1/2 and 35% increase in AUC of solifenacin in patients with moderate hepatic impairment. Doses of Solifenacin greater than 5 mg are not recommended in patients with moderate hepatic impairment (Child-Pugh B). Solifenacin is not recommended for patients with severe hepatic impairment (Child-Pugh C)
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Solifenacin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Solifenacin in patients who are immunocompromised.
Administration and Monitoring
Administration
Oral
Monitoring
There is limited information regarding Solifenacin Monitoring in the drug label.
IV Compatibility
There is limited information about the IV Compatibility.
Overdosage
Overdosage with Solifenacin can potentially result in severe anticholinergic effects and should be treated accordingly. The highest dose ingested in an accidental overdose of solifenacin succinate was 280 mg in a 5-hour period. This case was associated with mental status changes. Some cases reported a decrease in the level of consciousness. Intolerable anticholinergic side effects (fixed and dilated pupils, blurred vision, failure of heel-to-toe exam, tremors and dry skin) occurred on day 3 in normal volunteers taking 50 mg daily (5 times the maximum recommended therapeutic dose) and resolved within 7 days following discontinuation of drug. In the event of overdose with Solifenacin, treat with gastric lavage and appropriate supportive measures. ECG monitoring is also recommended.
Pharmacology
| |
Solifenacin
| |
| Systematic (IUPAC) name | |
| 1-azabicyclo[2.2.2]oct-3-yl (1S)-1-phenyl-3,4-dihydro-1H-isoquinoline-2-carboxylate | |
| Identifiers | |
| CAS number | |
| ATC code | G04 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 362.465 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | 90% |
| Protein binding | 98% |
| Metabolism | ? |
| Half life | 45 to 68 hours |
| Excretion | Renal (69.2%) and fecal (22.5%) |
| Therapeutic considerations | |
| Licence data |
|
| Pregnancy cat. | |
| Legal status |
POM(UK) [[Prescription drug|Template:Unicode-only]](US) |
| Routes | Oral |
Mechanism of Action
Solifenacin is a competitive muscarinic receptor antagonist. Muscarinic receptors play an important role in several major cholinergically mediated functions, including contractions of urinary bladder smooth muscle and stimulation of salivary secretion.
Structure
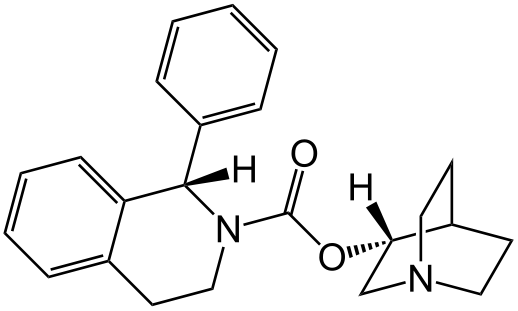
Solifenacin® (solifenacin succinate) is a competitive muscarinic receptor antagonist. Chemically, solifenacin succinate is butanedioic acid, compounded with (1S)-(3R)-1-azabicyclo[2.2.2]oct-3-yl 3,4-dihydro-1-phenyl-2(1H)-iso-quinolinecarboxylate (1:1) having an empirical formula of C23H26N2O2C4H6O4, and a molecular weight of 480.55. The structural formula of solifenacin succinate is:
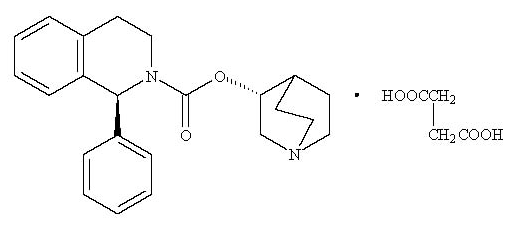
Solifenacin succinate is a white to pale-yellowish-white crystal or crystalline powder. It is freely soluble at room temperature in water, glacial acetic acid, dimethyl sulfoxide, and methanol. Each Solifenacin tablet contains 5 or 10 mg of solifenacin succinate and is formulated for oral administration. In addition to the active ingredient solifenacin succinate, each Solifenacin tablet also contains the following inert ingredients: lactose monohydrate, corn starch, hypromellose 2910, magnesium stearate, talc, polyethylene glycol 8000 and titanium dioxide with yellow ferric oxide (5 mg Solifenacin tablet) or red ferric oxide (10 mg Solifenacin tablet).
Pharmacodynamics
Cardiac Electrophysiology
The effect of 10 mg and 30 mg solifenacin succinate on the QT interval was evaluated at the time of peak plasma concentration of solifenacin in a multi-dose, randomized, double-blind, placebo and positive-controlled (moxifloxacin 400 mg) trial. Subjects were randomized to one of two treatment groups after receiving placebo and moxifloxacin sequentially. One group (n=51) went on to complete 3 additional sequential periods of dosing with solifenacin 10, 20, and 30 mg while the second group (n=25) in parallel completed a sequence of placebo and moxifloxacin. Study subjects were female volunteers aged 19 to 79 years. The 30 mg dose of solifenacin succinate (three times the highest recommended dose) was chosen for use in this study because this dose results in a solifenacin exposure that covers those observed upon co-administration of 10 mg Solifenacin with potent CYP3A4 inhibitors (e.g. ketoconazole, 400 mg). Due to the sequential dose escalating nature of the study, baseline EKG measurements were separated from the final QT assessment (of the 30 mg dose level) by 33 days. The median difference from baseline in heart rate associated with the 10 and 30 mg doses of solifenacin succinate compared to placebo was -2 and 0 beats/minute, respectively. Because a significant period effect on QTc was observed, the QTc effects were analyzed utilizing the parallel placebo control arm rather than the pre-specified intra-patient analysis. Representative results are shown in Table 2.

Moxifloxacin was included as a positive control in this study and, given the length of the study, its effect on the QT interval was evaluated in 3 different sessions. The placebo subtracted mean changes (90% CI) in QTcF for moxifloxacin in the three sessions were 11 (7, 14), 12 (8, 17), and 16 (12, 21), respectively. The QT interval prolonging effect appeared greater for the 30 mg compared to the 10 mg dose of solifenacin. Although the effect of the highest solifenacin dose (three times the maximum therapeutic dose) studied did not appear as large as that of the positive control moxifloxacin at its therapeutic dose, the confidence intervals overlapped. This study was not designed to draw direct statistical conclusions between the drugs or the dose levels.
Pharmacokinetics
Absorption
After oral administration of Solifenacin to healthy volunteers, peak plasma levels (Cmax) of solifenacin are reached within 3 to 8 hours after administration, and at steady state ranged from 32.3 to 62.9 ng/mL for the 5 and 10 mg Solifenacin tablets, respectively. The absolute bioavailability of solifenacin is approximately 90%, and plasma concentrations of solifenacin are proportional to the dose administered.
Effect of food
Solifenacin may be administered without regard to meals. A single 10 mg dose administration of Solifenacin with food increased Cmax and AUC by 4% and 3%, respectively.
Distribution
Solifenacin is approximately 98% (in vivo) bound to human plasma proteins, principally to ∝1-acid glycoprotein. Solifenacin is highly distributed to non-CNS tissues, having a mean steady-state volume of distribution of 600L.
Metabolism
Solifenacin is extensively metabolized in the liver. The primary pathway for elimination is by way of CYP3A4; however, alternate metabolic pathways exist. The primary metabolic routes of solifenacin are through N-oxidation of the quinuclidin ring and 4R-hydroxylation of tetrahydroisoquinoline ring. One pharmacologically active metabolite (4R-hydroxy solifenacin), occurring at low concentrations and unlikely to contribute significantly to clinical activity, and three pharmacologically inactive metabolites (N-glucuronide and the N-oxide and 4R-hydroxy-N-oxide of solifenacin) have been found in human plasma after oral dosing.
Excretion
Following the administration of 10 mg of 14C-solifenacin succinate to healthy volunteers, 69.2% of the radioactivity was recovered in the urine and 22.5% in the feces over 26 days. Less than 15% (as mean value) of the dose was recovered in the urine as intact solifenacin. The major metabolites identified in urine were N-oxide of solifenacin, 4R-hydroxy solifenacin and 4R-hydroxy-N-oxide of solifenacin and in feces 4R-hydroxy solifenacin. The elimination half-life of solifenacin following chronic dosing is approximately 45-68 hours.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
No increase in tumors was found following the administration of solifenacin succinate to male and female mice for 104 weeks at doses up to 200 mg/kg/day (5 and 9 times, respectively, of the exposure at the maximum recommended human dose [MRHD] of 10 mg), and male and female rats for 104 weeks at doses up to 20 and 15 mg/kg/day, respectively (<1 times the exposure at the MRHD). Solifenacin succinate was not mutagenic in the in vitro Salmonella typhimurium or Escherichia coli microbial mutagenicity test or chromosomal aberration test in human peripheral blood lymphocytes with or without metabolic activation, or in the in vivo micronucleus test in rats. Solifenacin succinate had no effect on reproductive function, fertility or early embryonic development of the fetus in male and female mice treated with 250 mg/kg/day (13 times the exposure at the MRHD) of solifenacin succinate, and in male rats treated with 50 mg/kg/day (<1 times the exposure at the MRHD) and female rats treated with 100 mg/kg/day (1.7 times the exposure at the MRHD) of solifenacin succinate.
Clinical Studies
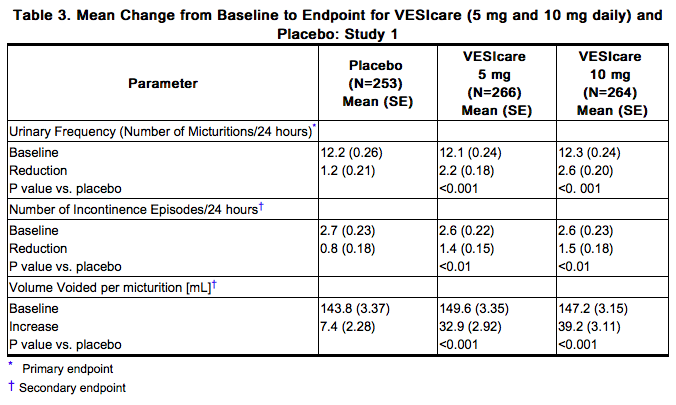
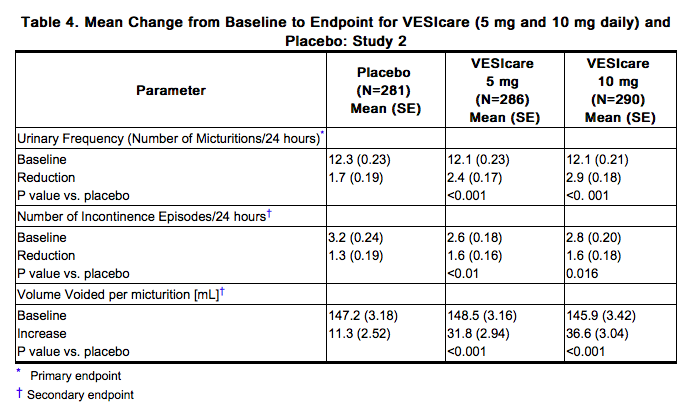
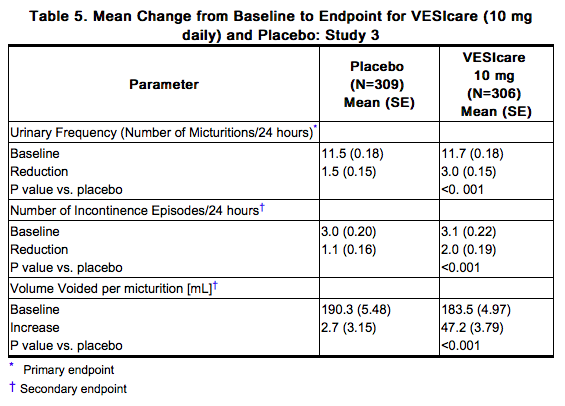
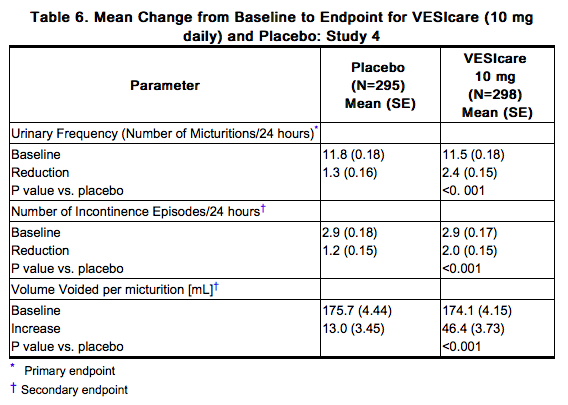
Solifenacin was evaluated in four twelve-week, double-blind, randomized, placebo-controlled, parallel group, multicenter clinical trials for the treatment of overactive bladder in patients having symptoms of urinary frequency, urgency, and/or urge or mixed incontinence (with a predominance of urge). Entry criteria required that patients have symptoms of overactive bladder for ≥ 3 months duration. These studies involved 3027 patients (1811 on Solifenacin and 1216 on placebo), and approximately 90% of these patients completed the 12-week studies. Two of the four studies evaluated the 5 and 10 mg Solifenacin doses and the other two evaluated only the 10 mg dose. All patients completing the 12-week studies were eligible to enter an open label, long term extension study and 81% of patients enrolling completed the additional 40-week treatment period. The majority of patients were Caucasian (93%) and female (80%) with a mean age of 58 years. The primary endpoint in all four trials was the mean change from baseline to 12 weeks in number of micturitions/24 hours. Secondary endpoints included mean change from baseline to 12 weeks in number of incontinence episodes/24 hours, and mean volume voided per micturition. The efficacy of Solifenacin was similar across patient age and gender. The mean reduction in the number of micturitions per 24 hours was significantly greater with Solifenacin 5 mg (2.3; p<0.001) and Solifenacin 10 mg (2.7; p<0.001) compared to placebo, (1.4). The mean reduction in the number of incontinence episodes per 24 hours was significantly greater with Solifenacin 5 mg (1.5; p<0.001) and Solifenacin 10 mg (1.8; p<0.001) treatment groups compared to placebo (1.1). The mean increase in the volume voided per micturition was significantly greater with Solifenacin 5 mg (32.3 mL; p<0.001) and Solifenacin 10 mg (42.5 mL; p<0.001) compared with placebo (8.5 mL). The results for the primary and secondary endpoints in the four individual 12-week clinical studies of Solifenacin are reported in Table 3 through 6.
How Supplied
Solifenacin is supplied as round, film-coated tablets, available in bottles and unit dose blister packages as follows: Each 5 mg tablet is light yellow and debossed with a logo and “150” and is available as follows:

Each 10 mg tablet is light pink and debossed with a logo and “151” and is available as follows:
Storage
Store at 25C (77F) with excursions permitted from 15C to 30C (59°F-86F)
Images
Drug Images
{{#ask: Page Name::Solifenacin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Solifenacin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Patients should be informed that antimuscarinic agents such as Solifenacin have been associated with constipation and blurred vision. Patients should be advised to contact their physician if they experience severe abdominal pain or become constipated for 3 or more days. Because Solifenacin may cause blurred vision, patients should be advised to exercise caution in decisions to engage in potentially dangerous activities until the drug’s effect on the patient’s vision has been determined. Heat prostration (due to decreased sweating) can occur when anticholinergic drugs, such as Solifenacin, are used in a hot environment. Patients should read the patient leaflet entitled “Patient Information Solifenacin” before starting therapy with Solifenacin. Patients should be informed that solifenacin may produce angioedema, which could result in life-threatening airway obstruction. Patients should be advised to promptly discontinue solifenacin therapy and seek immediate attention if they experience edema of the tongue or laryngopharynx, or difficulty breathing. FDA Approved Patient Labeling Solifenacin® (VES-ih-care) (solifenacin succinate) Tablet
Read the Patient Information that comes with Solifenacin before you start taking it and each time you get a refill. There may be new information. This leaflet does not take the place of talking with your doctor about your medical condition or treatment.
What is Solifenacin?
Solifenacin is a prescription medicine for adults used to treat the following symptoms due to a condition called overactive bladder:
- Urge urinary incontinence: a strong need to urinate with leaking or wetting accidents
- Urgency: a strong need to urinate right away
- Frequency: urinating often
It is not known if Solifenacin is safe and effective in children.
Who should NOT take Solifenacin? Do not take Solifenacin if you:
- are not able to empty your bladder (urinary retention)
- have delayed or slow emptying of your stomach (gastric retention)
- have an eye problem called “uncontrolled narrow-angle glaucoma”
- are allergic to solifenacin succinate or any of the ingredients in Solifenacin. See the end of this leaflet for a complete list of ingredients.
What should I tell my doctor before taking Solifenacin? Before you take Solifenacin, tell your doctor if you:
- have any stomach or intestinal problems or problems with constipation
- have trouble emptying your bladder or you have a weak urine stream
- have an eye problem called "narrow angle glaucoma"
- have liver problems
- have kidney problems
- have a rare heart problem called "QT prolongation"
- are pregnant or plan to become pregnant. It is not known if Solifenacin will harm your unborn baby. Talk to your doctor if you are pregnant or plan to become pregnant.
- are breastfeeding or plan to breastfeed. It is not known if Solifenacin passes into your breast milk. You and your doctor should decide if you will take Solifenacin or breastfeed. You should not do both.
Tell your doctor about all the medicines you take, including prescription and nonprescription medicines, vitamins, and herbal supplements. Solifenacin may affect the way other medicines work, and other medicines may affect how Solifenacin works.
How should I take Solifenacin?
- Take Solifenacin exactly as your doctor tells you to take it.
- You should take 1 Solifenacin tablet 1 time a day.
- You should take Solifenacin with water and swallow the tablet whole.
- You can take Solifenacin with or without food.
- If you miss a dose of Solifenacin, begin taking Solifenacin again the next day. Do not take 2 doses of Solifenacin the same day.
- If you take too much Solifenacin, call your doctor or go to the nearest hospital emergency room right away.
What should I avoid while taking Solifenacin? Solifenacin can cause blurred vision or drowsiness. Do not drive or operate heavy machinery until you know how Solifenacin affects you.
What are the possible side effects of Solifenacin? Solifenacin may cause serious side effects including:
- Serious allergic reaction. Stop taking Solifenacin and get medical help right away if you have:
- severe itching
- trouble breathing
The most common side effects of Solifenacin include:
- dry mouth
- constipation. Call your doctor if you get severe stomach area (abdominal) pain or become constipated for 3 or more days.
- heat exhaustion or heat-stroke. This can happen when Solifenacin is used in hot environments. Symptoms may include:
- decreased sweating
- dizziness
- tiredness
- nausea
- increase in body temperature
Tell your doctor if you have any side effect that bothers you or that does not go away. These are not all the possible side effects of Solifenacin. For more information, ask your doctor or pharmacist. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store Solifenacin?
- Store Solifenacin at 59°F to 86°F (15°C to 30°C). Keep the bottle closed.
- Safely throw away medicine that is out of date or that you no longer need.
Keep Solifenacin and all medicines out of the reach of children.
General information about Solifenacin. Medicines are sometimes prescribed for purposes other than those listed in Patient Information leaflets. Do not use Solifenacin for a condition for which it was not prescribed. Do not give Solifenacin to other people, even if they have the same symptoms you have. It may harm them. This leaflet summarizes the most important information about Solifenacin. If you would like more information, talk with your doctor. You can ask your doctor or pharmacist for information about Solifenacin that is written for health professionals. For more information, go to www.Solifenacin.com or call (800) 727-7003.
What are the ingredients in Solifenacin?
Active ingredient: solifenacin succinate Inactive ingredients: lactose monohydrate, corn starch, hypromellose 2910, magnesium stearate, talc, polyethylene glycol 8000 and titanium dioxide with yellow ferric oxide (5 mg Solifenacin tablet) or red ferric oxide (10 mg Solifenacin tablet)
What is overactive bladder?
Overactive bladder occurs when you cannot control your bladder contractions. When these muscle contractions happen too often or cannot be controlled you can get symptoms of overactive bladder, which are urinary frequency, urinary urgency, and urinary incontinence (leakage).
Precautions with Alcohol
Alcohol-Solifenacin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
VESICARE
Look-Alike Drug Names
Solifenacin - Vesanoid[1]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "https://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Solifenacin |Pill Name=Solifenacin_5 mg_NDC 51248-150.jpg |Drug Name=solifenacin succinate tablet, film coated |Pill Ingred=N/A|+sep=; |Pill Imprint=150 |Pill Dosage=5 mg |Pill Color=Yellow|+sep=; |Pill Shape=Round |Pill Size (mm)=8.00 |Pill Scoring=1 |Pill Image= |Drug Author=Astellas Pharma Technologies, Inc. |NDC=51248-150
}}
{{#subobject:
|Page Name=Solifenacin |Pill Name=Solifenacin_10 mg_NDC 51248-151.jpg |Drug Name=solifenacin succinate tablet, film coated |Pill Ingred=N/A|+sep=; |Pill Imprint=151 |Pill Dosage=10 mg |Pill Color=Pink|+sep=; |Pill Shape=Round |Pill Size (mm)=8.00 |Pill Scoring=1 |Pill Image= |Drug Author=Astellas Pharma Technologies, Inc. |NDC=51248-151
}}
{{#subobject:
|Label Page=Solifenacin |Label Name=Solifenacin_label_01.jpg
}}
{{#subobject:
|Label Page=Solifenacin |Label Name=Solifenacin_label_02.jpg
}}
{{#subobject:
|Label Page=Solifenacin |Label Name=Solifenacin_label_03.jpg
}}
{{#subobject:
|Label Page=Solifenacin |Label Name=Solifenacin_label_04.jpg
}}
{{#subobject:
|Label Page=Solifenacin |Label Name=Solifenacin_panel_01.png
}}
{{#subobject:
|Label Page=Solifenacin |Label Name=Solifenacin_panel_02.png
}}