PDE3 inhibitor
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
A PDE3 inhibitor is a drug which inhibits the action of the PDE3 phosphodiesterase enzyme.
First generation PDE inhibitors
Recognition that the knowledge about PDE could be used to develop drugs that were PDE inhibitors led to extensive research. Most studies used analogues of the nucleotide substrates or derivatives of natural product inhibitors such as xanthine (e.g. theophylline) and papaverine.[1][2]
The cAMP PDE active site can be considered as a summary of ideas about receptor topography resulting from the first generation inhibitors. The model of the Wells et al. version as cited in Erhardt and Chou (1991) includes the following:
- A phosphate binding area
- A lipophilic area that accommodates the non-polar side of the ribose moiety
- A pyrimidine binding site
- An imidazole binding site portion of the pyrimidine binding site
- A sterically hindered site
- An area with bulk tolerance[1]
Second generation PDE inhibitors
Since selective PDE3 inhibitors were recognised to be cardiotonic drugs there has been great interest in developing new drugs in this category. A large number of heterocyclic compounds have been synthesized during related research. These compounds constitute a second generation of PDE inhibitors. Although they have been directed mostly at PDE3, they present significant SAR for the PDE's in general.[1]
A “heterocycle-phenyl-imidazole” (H-P-I) pattern (figure 3) has been considered to be necessary for positive inotropic activity in cardiac muscle and many second generation inhibitors fit this pattern.[1]
The heterocycle region: Within each heterocycle there is the presence of a dipole and an adjacent acid proton (an amide function). These atoms are believed to mimic the electrophilic center in the phosphate group in cAMP and are confirmed as the primary site of binding. The heterocycle is a transition state analogue inhibitor of PDE. Alkyl groups, limited to either methyl or ethyl, on the heterocyclic ring usually enhance potency, with occasional exceptions.[1][2]
The phenyl region: It seems that an electron rich, centre such as phenyl, needs to be present. The beneficial effects of small alkyl groups on the heterocycle could be to twist the central ring away from exact coplanarity with the heterocyclic ring. There is a similar twist in cAMP and there is general agreement that high affinity PDE3 inhibitors should adopt an energetically favoured planar conformation that mimics the anti conformation of cAMP.[1][2]
The imidazole region: Various substituents have been placed at the para-position of the central phenyl ring. They are electron rich moieties and apparently a positively charged moiety cannot be tolerated in this region of the PDE receptor. There is general agreement about this inhibitor potency: Lactam ≥ Alkyl-CONH- ≥ Imidazoyl = Pyridine in place of the central phenyl with its nitrogen in the analogous 4 position ≥ Alkyl-S- > Simple [ether|ethers] > Halide = Amines > Imidazolium (which is totally inactive).[1]
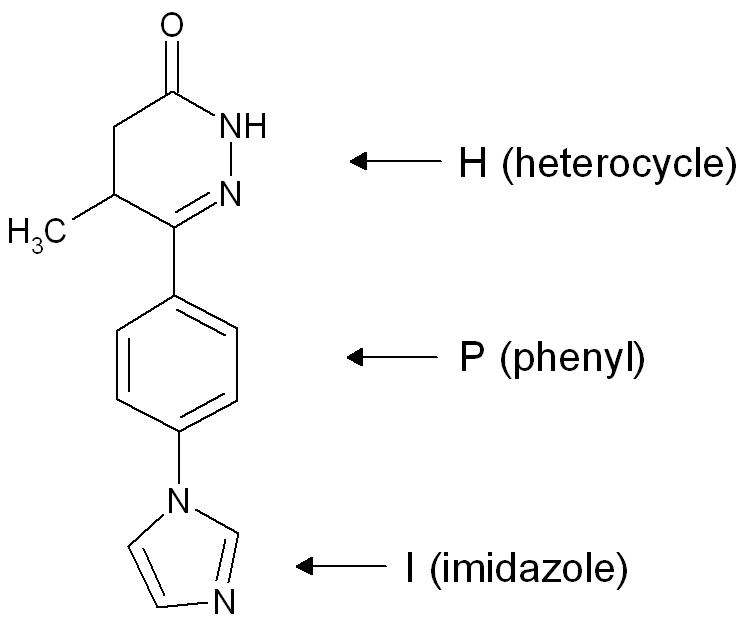
Identification of features common to the most selective inhibitors has led to a “five-point model” with:
- Presence of a strong dipole (carbonyl moiety) at one end of the molecule.
- An adjacent acid proton.
- A small sized alkyl substituent on the heterocyclic ring.
- A relatively flat overall topography.
- An electron rich centre and/or a hydrogen bond acceptor site opposite to the dipole.[2]
Examples of a selective PDE3 inhibitors:
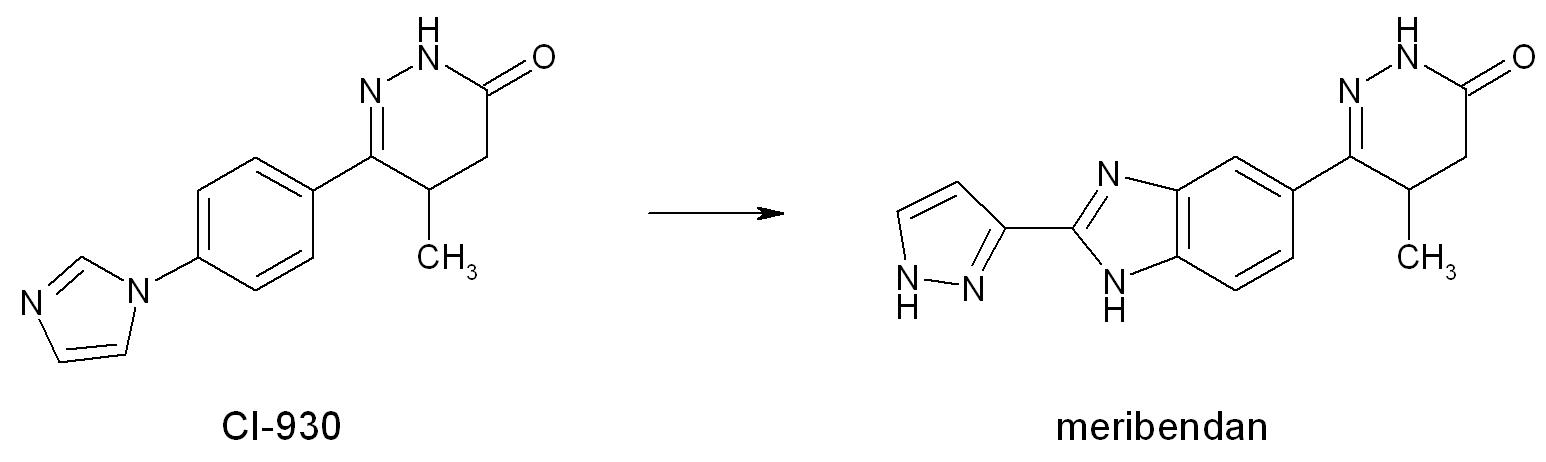
Theophylline is a non-selective agent. In contrast, meribendan is a very selective inhibitor (figure 4).[2]

Also, meribendan, has a higher level of selectivity in comparison with the parent compound CI-930 because, beside the basic nitrogen adjacent to the lactam moiety it possesses another basic nitrogen (benzimidazole ring), opposite to the primary binding site (figure 5).[2]
Many PDE3 inhibitor drugs have been developed, e.g. cilostazol, milrinone, vesnarionone, enoximone and pimobendan. PDE3 inhibitors have been studied for their effectiveness as inotropic agents in severe heart failure but unfortunately have shown increased mortality in well controlled studies. These drugs are therefore only used if the benefit exceeds the risk.[3]
To date no isoform selective inhibitors have been discovered but research in that field is of great interest.[4]
References
- ↑ 1.0 1.1 1.2 1.3 1.4 1.5 1.6 Erhardt P.W., Chou Y. (1991). "A topographical model for the c-AMP phosphodiesterase III active site". Life Sciences, 49(8): 553-568.
- ↑ 2.0 2.1 2.2 2.3 2.4 2.5 Fossa P., Boggia R., Mosti L. (1998). "Toward the identification of the cardiac cGMP inhibited-phosphodiesterase catalytic site". Journal of Computer-Aided Molecular Design, 12(4): 361-372.
- ↑ "Approval of Cilostazol". http://www.fda.gov/cder/news/cilostazol/approval.htm. Retrieved 25 September 2006. Check date values in:
|date=(help); External link in|publisher=(help) - ↑ Matthew M. (2003). "Isoform-Selective Inhibitors and Activators of PDE3 Cyclic Nucleotide Phosphodiesterases". WO 2003/012030: International patent application (PCT), World Intellectual Property Organization.
Template:Phosphodiesterase inhibitors Template:Cardiac stimulants excluding cardiac glycosides