Corneal transplantation
Template:Search infobox Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Corneal transplantation, also known as corneal grafting or penetrating keratoplasty, is a surgical procedure where a damaged or diseased cornea is replaced by donated corneal tissue which has been removed from a recently deceased individual having no known diseases which might affect the viability of the donated tissue. The cornea is the clear part of eye in front of the iris and pupil. The surgical procedure is performed by ophthalmologists, medical doctors who specialize in eyes, and are often done on an outpatient basis.
Indications
Indications for corneal transplantation include the following:
- Optical: To improve visual acuity by replacing the opaque or distorted host tissue by clear healthy donor tissue. The most common indication in this category is pseudophakic bullous keratopathy, followed by keratoconus, corneal degeneration, keratoglobus and dystrophy, as well as scarring due to keratitis and trauma.
- Tectonic/reconstructive: To preserve corneal anatomy and integrity in patients with stromal thinning and descemetoceles, or to reconstruct the anatomy of the eye, e.g. after corneal perforation.
- Therapeutic: To remove inflamed corneal tissue unresponsive to treatment by antibiotics or anti-virals.
- Cosmetic: To improve the appearance of patients with corneal scars that have given a whitish or opaque hue to the cornea. [2]
Pre-operative examination
In most instances, the patient will meet with their ophthalmologist for an examination in the weeks or months preceding the surgery. During the exam, the ophthalmologist will examine the eye and diagnose the condition. The doctor will then discuss the condition with the patient, including the different treatment options available. The doctor will also discuss the risks and benefits of the various options. If the patient elects to proceed with the surgery, the doctor will have the patient sign an informed consent form. The doctor might also perform a physical examination and order lab tests, such as blood work, X-rays, or an EKG.
The surgery date and time will also be set, and the patient will be told where the surgery will take place.
- The surgery only takes place when the best corresponding donor tissue is found. This can take weeks and months.
Procedure
On the day of the surgery, the patient arrives to either a hospital or an outpatient surgery center, where the procedure will be performed. The patient is given a brief physical examination by the surgical team and is taken to the operating room. In the OR, the patient lies down on an operating table and is either given general anesthesia, or local anesthesia and a sedative.
With anesthesia induced, the surgical team prepares the eye to be operated on and drapes the face around the eye. An eyelid speculum is placed to keep the lids open, and some lubrication is placed on the eye to prevent drying. In children, a metal ring is stitched to the sclera which will provide support of the sclera during the procedure.
A trephine (a circular cutting device) is then placed over the cornea and is used by the surgeon to cut the host cornea, which removes a circular disc of the patient cornea. The trephine is then removed and the surgeon cuts a circular graft (a "button") from the donor cornea. Once this is done, the surgeon returns to the patient's eye and removes the host cornea.
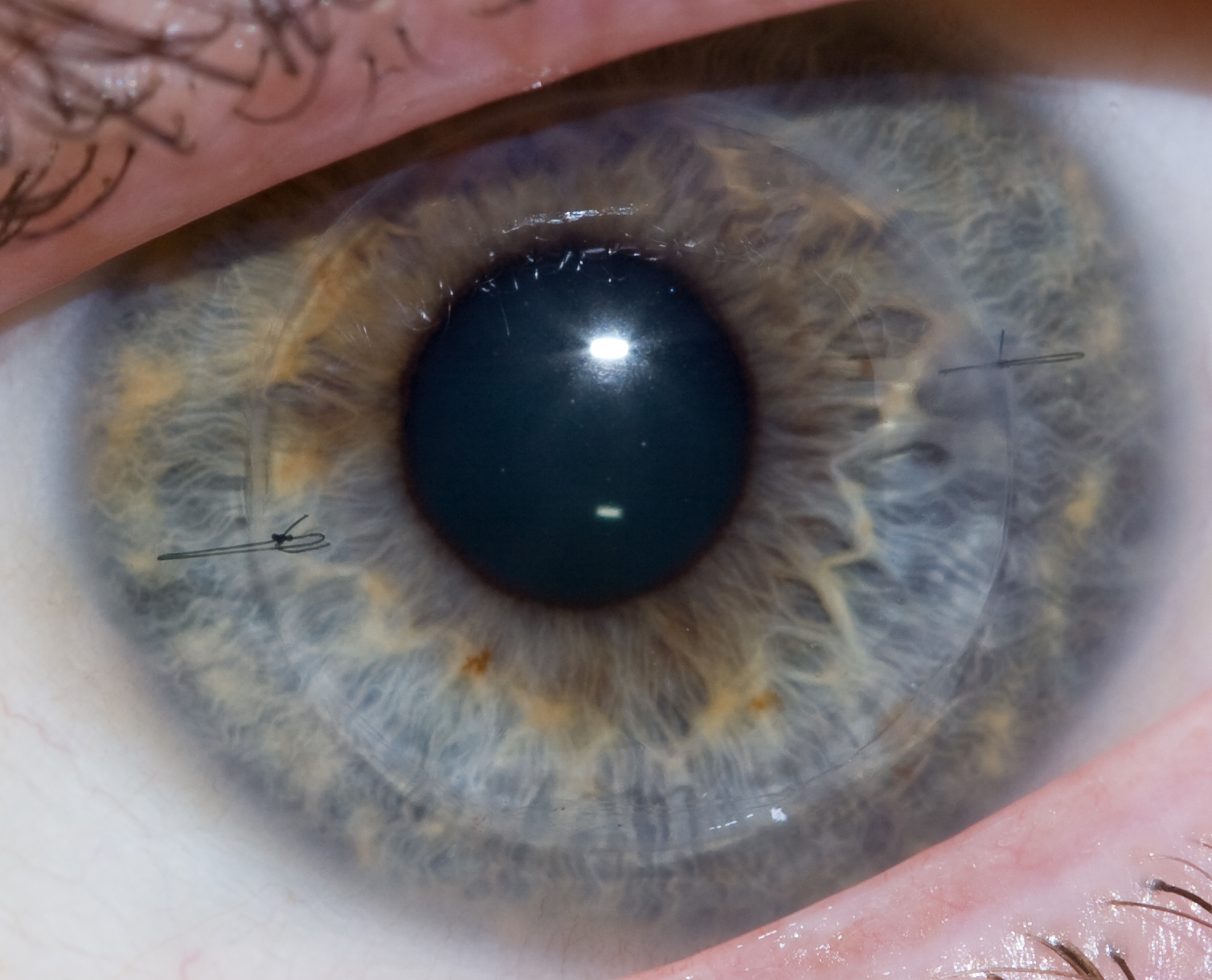
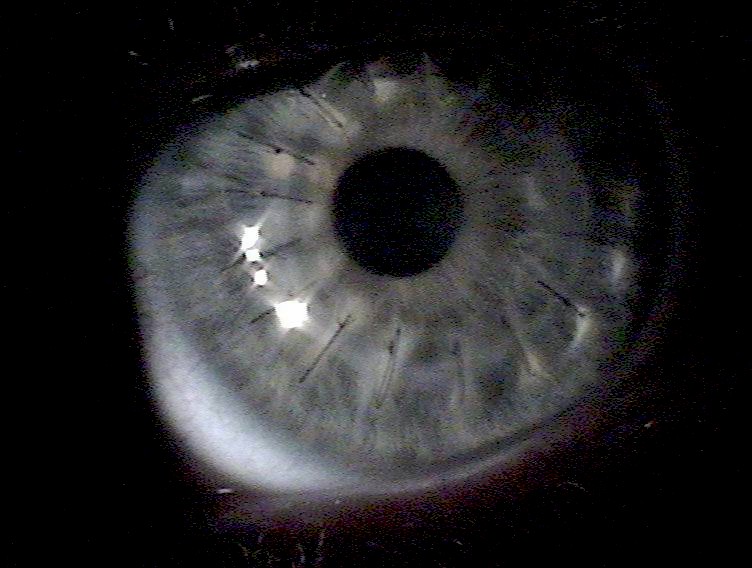
The donor cornea is then brought into the surgical field and maneuvered into place with forceps. Once in place, the surgeon will fasten the cornea to the eye with a running stitch (as used in the upper image above) or a multiple interrupted stitches (as in the lower image). The surgeon then reconstitutes the anterior chamber by injecting a sterile solution via cannula, then testing that it's watertight by placing a dye on the wound exterior.
Antibiotic eyedrops are placed, the eye is patched, and the patient is taken to a recovery area while the effects of the anesthesia wear off. The patient typically goes home following this and sees the doctor the following day for the first post operative appointment.
Risks
While the cornea is avascular, there is still a potential for some blood loss, usually from suturing the metal ring to the sclera. Any blood loss is typically less than 2ml.
There is also a risk of infection. Since the cornea has no blood vessels (it takes its nutrients from the aqueous humor) it heals much slower than a cut on the skin. While the wound is healing, it is possible that it might become infected by various microorganisms. This risk is minimized by antibiotic prophylaxis (using antibiotic eyedrops, even when no infection exists).
Graft failure can occur at any time after the cornea has been transplanted, even years or decades later. The causes can vary, though it is usually due to new injury or illness. Treatment can be either medical or surgical, depending on the individual case. An early, technical cause of failure, may be an overly tight stitch cheesewiring through the sclera.
Prognosis
When the primary purpose of a cornea transplant is to improve visual acuity, the prognosis is dependent upon whether the rest of the eye is healthy. If it is, then it should be possible to recover normal vision.
History
The first cornea transplant was performed in 1905, by Eduard Zirm, making it one of the first types of transplant surgery successfully performed. Another pioneer of the operation was Ramon Castroviejo. Soviet eye surgeon Vladimir Filatov's attempts at tranplanting cornea started with the first try in 1912 and were continued, gradually improving until at 6th of May, 1931 he successfully grafted a patient using corneal tissue from a deceased person.[1] He widely reported of another transplant in 1936, disclosing his technique in full detail.[2] In 1936, Castroviejo did a first transplantation in an advanced case of keratoconus, achieving significant improvement in patient's vision.[3][4]
Advances in microscopes enabled surgeons to get a better view of the surgical field, while advances in materials science enabled them to use sutures finer than a human hair.
Instrumental in the success of cornea transplants were the establishment of eye banks. These are organizations located throughout the world to coordinate the distribution of donated corneas to surgeons, as well as providing eyes for research. Some eye banks also distribute other anatomical gifts.
Synthetic corneas
AlphaCor
In cases where there have been several graft failures or the risk for keratoplasty is high, synthetic corneas can substitute successfully for donor corneas. Such a device contains a peripheral skirt and a transparent central region. These two parts are connected on a molecular level by an interpenetrating polymer network, made from poly-2-hydroxyethyl methacrylate (pHEMA). AlphaCor is an FDA-approved type of synthetic cornea measuring 7.0 mm in diameter and 0.5 mm in thickness. The main advantages of synthetic corneas are that they are biocompatible, and the network between the parts and the device prevents complications that could arise at their interface. The probability of retention in one large study was estimated at 62% at 2 years follow-up.[5] AlphaCor carry lesser risk of diseases that could be transmitted through donor tissue. However, they also cost $10,000.
AlphaCor surgery is reserved for patients who have had traditional cornea transplants either 1) fail repeatedly, 2) reject due to autoimmune process, or 3) have a high vascularized cornea that makes traditional cornea transplantation unsuitable.
Use of AlphaCor involved a two part procedure. First the AlphaCor disc is implanted under the tissue. Second, after healing many months later the membrane covering the lens of the disc is removed allowing that eye to see. Glasses will still be needed to optimize vision afterwards.
Osteo-Odonto-Keratoprosthesis
In a very rare and complicated multi-step surgical procedure, employed to help the most disabled patients, a lamina of the patient's tooth is grafted into the eye, with an artificial lens installed in the transplanted piece.
Alternatives
Phototherapeutic keratectomy (PTK)
Diseases that only affect the surface of the cornea can be treated with an operation called phototherapeutic keratectomy. With the precision of an excimer laser and a modulating agent coating the eye, irregularities on the surface can be removed. However, in most of the cases where corneal transplantation is recommended, PTK would not be effective.
Intrastromal corneal ring segments
The implants manufactured under the trade name Intacs are the only patented intrastromal corneal implant that has US FDA approval and European CE Mark for both Myopia and Keratoconus. There are over one-hundred clinical articles at [3] for clinical reference (search for Intacs and they all are arranged in chronological order) and are a well documented clinical solution for treating keratoconus. Another version of intrastromal cornea ring segments is manufactured under the trade name KeraRing and is available in South America and Europe.
With this procedure, the implants are placed in the stroma to reshape the cornea into a more natural shape. In mild myopia, this corrects a patients vision. In keratoconus, the goal is to reshape the cornea to where contact lens intolerant patients are able to achieve functional vision with contact lenses or glasses. Although surgical procedure don't carry a guarantee, one clinically proven benefit of Intacs is that they can be safely removed and the cornea returns to its pre-operative state. Future treatment options are not affected.
Contact lenses
In the early stages of some diseases (such as keratoconus), contact lenses can be used to improve vision. Contact lenses improve vision by tear fluid filling the gap between the irregular cornea surface and the regular inner surface of the lens. However, this is only for the early stages and corneal transplant is necessary for vision correction if eyesight worsens.
New technology
High speed lasers
Blades are being replaced by high speed lasers in order to make surgical incisions more precise. These improved incisions allow the cornea to heal more quickly and the sutures to be removed sooner. The cornea heals more strongly, and the risk of transplant failure is smaller than with standard blade operations. Not only does this dramatically improve visual recovery and healing, it also allows the possibility for improvement in visual outcomes.
Since 2004, Amnitrans Eyebank in Rotterdam, The Netherlands, provides donor corneas pre-cut for advanced keratoplasty procedures, such as DSEK, DSAEK, FS-DSEK and DMEK. In 2007, Seattle-based SightLife, one of the leading corneal tissue banks in the world, introduced a process for the preparation of donated corneal tissue using a Femtosecond Laser. This process is known as Custom Corneal Tissue.
Riboflavin
A treatment that involves a one-time application of riboflavin eye drops and thirty minutes of UV-A light can arrest the progression of Keratoconus. The riboflavin, when activated by UV-A light, augments the collagen cross-links in the stroma. With this treatment, the cornea regains some of its mechanical strength. When this treatment is combined with intrastromal cornea ring segments, it has been shown to reverse Keratoconus.
DSEK/DSAEK/DMEK
Endothelial keratoplasty (EK) has been introduced by Melles et al. in 1998. Today there are three forms of EK. Deep Lamellar Endothelial Keratoplasty (DLEK) in which the posterior part of the recipient cornea is replaced by donor tissue. Descemet's Stripping (Automated) Endothelial Keratoplasty (DSEK/DSAEK) in which the diseased Descemet's membrane is removed and replaced by a healthy donor posterior transplant. DSEK/DSAEK uses only a small incision that is either self-sealing or may be closed with a few sutures. The small incision offers several benefits over traditional methods of corneal transplant such as Penetrating Keratoplasty. Because the procedure is less invasive, DSAEK leaves the eye much stronger and less prone to injury than full-thickness transplants. Additionally, DSAEK has a more rapid rate of visual recovery. Vision is typically restored in three to six months rather than one to two years. Descemet Membrane Endothelial Keratoplasty (DMEK) is the most recent EK technique in which an isolated Descemet membrane is transplanted. The DMEK procedure combines the anatomical benefits of DSEK/DSAEK with visual rehabilitation to 20/40 or better in 90% of cases and 20/25 or better in 60% of cases within the first three months.
Stem cells
There is a bioengineering technique that uses stem cells to create corneas or part of corneas that can be transplanted into the eyes. Corneal stem cells are removed from a healthy cornea. They are collected and, through laboratory procedures, made into five to ten layers of cells that can be stitched into a patient’s eye. The stem cells are placed into the area where the damaged cornea tissue has been removed. This is a good alternative for those that cannot gain vision through regular cornea transplants. A new development, announced by the University of Cincinnati Medical School in May 2007, would use bone marrow stem cells to regrow the cornea and its cells. This technique, which proved successful in mouse trials, would be of use to those suffering from inherited genetic degenerative conditions of the cornea, especially if other means like a transplant aren't feasible. It works better than a transplant because these stem cells keep their ability to differentiate and replicate, and so keep the disease from recurring, longer and better.
Epidemiology and economics
Corneal transplant is one of the most common transplant procedures. Although approximately 100,000 procedures are performed worldwide each year, some estimates report that 10,000,000 people are affected by various disorders that would benefit from corneal transplantation.[4]
In Australia, approximately 1,500 grafts are performed each year. [5] According to the NHS Blood and Transplant, over 2,300 corneal transplant procedures are performed each year in the United Kingdom. [6] Between April 1, 2005 and March 31, 2006, 2,503 people received corneal transplants in the UK. [7]
In the United States, the cost is usually covered in part by Medicare and health insurers. Reimbursement depends on your personal healthcare provider. Usually 80% of the cost will be covered by your agency. Those on Medicare will be reimbursed up to $1,200 while the remainder is left up to the patient. The average cost of the procedure ranges from $7,500 to $11,000. However, there is a company that claims to be able to cover all costs for as little as $3,700. In 2005, there were about 32,840 corneal transplant recipients. The estimated first year billed charges per patient, including medications, was $19,100. There were a larger number of transplants for patients over 65 than under, 18,000 compared to 14,840. Milliman estimates that there will be 32,700 corneal transplant recipients in 2006 [8]. The predicted cost for the first year after transplantation in 2006 is $21,500[9].
In Sweden, corneal transplants are paid by taxes. Every year about six hundred of these operations are performed.
References
- ↑ Vladimir Filatov, peoples.ru
- ↑ Eye repair - TIME, Monday, Apr. 13, 1936
- ↑ Castroviejo R (1948). "Keratoplasty for the Treatment of Keratoconus". Trans Am Ophthalmol Soc. 46: 127–53. PMC 1312756. PMID 16693468.
- ↑ Castroviejo, R.: International Abstract of Surgery, 65:5, December, 1937.
- ↑ Hicks CR, Crawford GJ, Dart JK, Grabner G, Holland EJ, Stulting RD, Tan DT, Bulsara M (2006). "AlphaCor: Clinical outcomes". Cornea. 25 (9): 1034–42. doi:10.1097/01.ico.0000229982.23334.6b. PMID 17133049. Unknown parameter
|month=ignored (help)
Sources
- http://biomed.brown.edu/Courses/BI108/2006-108websites/group11corneal%20implants/index.html
- http://www.crstoday.com/PDF%20Articles/1104/crst1104_pr.pdf
- http://www.vision-institute.com/techniques/ptk.html
- http://dro.hs.columbia.edu/icrs.htm
- http://www.indystar.com/apps/pbcs.dll/article?AID=/20060419/CUSTOMPUB01/60413016/-1/ZONES04
- http://www.news.com.au/story/0,10117,18829980-29277,00.html
- http://www.cnyeyecare.com/newdetail.php?id=21
- http://science.howstuffworks.com/news-item94.htm
- http://www.milliman.com/expertise/healthcare/publications/rr/pdfs/US-Organ-Tissue-Transplant-2005-RR.pdf
- http://www.milliman.com/expertise/healthcare/publications/rr/pdfs/US-Organ-Tissue-Transplant-2006-RR.pdf
- http://www.niios.com/content.php?na=9#publications
External links
- Brent Stone's Cornea Transplant Blog
- Cornea Transplant Surgery Images
- Eye Bank Association of America
- EyeMD link cornea transplant page
- Larry Wall's cornea transplant diary
- Merck Manual: Corneal transplantation
- Fuchs' Dystrophy Home Page and support
- Fuchs' Dystrophy Support Group
- Lions Eye Donation Service, Melbourne Australia
- Cornea Transplant Surgery Procedure
- Facts About the Cornea and Corneal Disease The National Eye Institute (NEI)
- SightLife
- Heartland Lions Eye Banks
- Descemet Membrane Endothelial Keratoplasty
- PubMed peer-reviewed publications
Template:Organ transplantation Template:Eye surgery
de:Keratoplastik it:Cheratoplastica nl:Hoornvliestransplantatie no:Hornhinnetransplantasjon