Breast cancer chemotherapy
|
Breast Cancer Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Breast cancer chemotherapy On the Web |
|
American Roentgen Ray Society Images of Breast cancer chemotherapy |
|
Risk calculators and risk factors for Breast cancer chemotherapy |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1],Assistant Editor(s)-In-Chief: Jack Khouri, Associate Editor(s)-in-Chief: Soroush Seifirad, M.D.[2]
Overview
Breast cancer chemotherapy is a combination of cytotoxic drugs (chemotherapy), hormonal therapy, hormonal receptor modulators, aromatase inhibitors, monoclonal antibodies, and growth factor receptor tyrosine kinase inhibitors.
Principles of therapy
- The main aims of therapy are prolonging survival, improving quality of life and avoiding treatment-induced toxicity. Given that treatment is palliative, patients should be given treatment holidays in order to reduce drug-induced toxicity.
- HER2 overexpression and hormone receptor status are very important factors that guide therapy and influence prognosis.
- Multiagent chemotherapy regimens don't show significant survival benefit compared to single-drug regimens and augment toxicity.
- Chemotherapy is recommended for patients with ER-negative metastatic breast cancer and those with ER-positive breast cancer resistant to endocrine therapy or associated with visceral disease.
- Trastuzumab (Her2 inhibitor) increases the clinical benefit of first-line chemotherapy in metastatic breast cancer that overexpresses HER2.[1]
- Single-agent trastuzumab is active and well tolerated as first-line treatment of women with metastatic breast cancer with HER2 overexpression.[2]
Chemotherapy
- The most active single agents are Anthracyclines (Doxorubicin 60-75 mg/m2 IV every 21 days), Taxanes (paclitaxel 175 mg/m2 every 21 days or 80 mg/m2 IV days 1,8,15 every 21 days; docetaxel 60-100 mg/m2 IV every 21 days), Capecitabine (1000-1250 mg/m2 PO twice a day on days 1-14 every 21 days), Gemcitabine (800-1200 mg/m2 IV days 1,8,15 every 28 days), and Vinorelbine (25 mg/m2 weekly or days 1,8,15 every 28 days)
- Bevacizumab (the monoclonal antibody that inhibits VEGF, thus preventing angiogenesis) has been shown to improve the efficacy of taxanes in frontline treatment of patients with metastatic breast cancer.[3] A randomized phase III study showed that the combination of bevacizumab with docetaxel did not significantly impact on the safety profile of docetaxel. Bevacizumab 15 mg/kg every 3 weeks significantly increased progression free survival when combined with docetaxel as first-line therapy for netastatic breast cancer compared with docetaxel plus placebo.[4]
- Consider using combination chemotherapy to treat patients with advanced breast cancer for whom a greater probability of response is important and who understand and are likely to tolerate the additional toxicity.[5]
- Combination chemotherapy include Gemcitabine combined with paclitaxel and docetaxel combined with capecitabine.
- There is little evidence from trials reported from 2000 to 2007 that major survival differences exist between many commonly employed chemotherapy regimens.[6]
Chemotherapy types
Chemotherapy can be given both before and after surgery. Neoadjuvant chemotherapy is used to shrink the size of a tumor prior to surgery. Adjuvant chemotherapy is given after surgery to reduce the risk of recurrence. Palliative chemotherapy is used to control (but not cure) cancer in settings in which cancer has spread beyond the breast and localized lymph nodes.
Common adjuvant chemotherapy for breast cancer
Several different chemotherapy regimens may be used.[7] Determining the appropriate regimen depends on many factors, including the character of the tumor, lymph node status, and the age and health of the patient. In general, chemotherapy has increasing side effects as the patient's age passes 65. The following is a list of commonly used adjuvant chemotherapy for breast cancer:
- CMF: cyclophosphamide, methotrexate, and 5-fluorouracil given 4-weekly for 6 cycles
- FAC (or CAF): 5-fluorouracil, doxorubicin, cyclophosphamide given 3-weekly for 6 cycles
- AC (or CA): Adriamycin (doxorubicin) and cyclophosphamide given 3-weekly for 4 cycles
- AC-Taxol: AC given 3-weekly for 4 cycles followed by paclitaxel given either 3-weekly for 4 cycles or weekly (at a smaller dose) for 12 weeks
- TAC: Taxotere (docetaxel), Adriamycin (doxorubicin), and cyclophosphamide given 3-weekly for 6 cycles
- FEC: 5-fluorouracil, epirubicin and cyclophosphamide given 3-weekly for 6 cycles
- FECD: FEC given 3-weekly for 3 cycles followed by docetaxel given 3-weekly for 3 cycles
- TC: Taxotere (docetaxel) and cyclophosphamide given 3-weekly for 4 cycles
- Dose dense regimen: Some of the regimens above (e.g. AC followed by paclitaxel) may be given in a shorter period (i.e. every 2 weeks instead of every 3 weeks).
- In addition to chemotherapy, trastuzumab may also be added to the regimen depending on the tumor characteristics (i.e. HER2/neu status) and risk of relapse. It is usually given either 3 weekly or weekly for a total duration of 1 year.
Since chemotherapy affects the production of white blood cells, granulocyte colony-stimulating factor (G-CSF) is sometimes administered along with chemotherapy. This has been shown to reduce, though not completely prevent, the rate of infection and low white cell count. Most adjuvant breast cancer chemotherapy regimens do not routinely require growth factor support except for those associated with a high incidence of bone marrow suppression and infection. These may include chemotherapy given in the dose-dense fashion i.e. 2-weekly instead of 3-weekly or TAC chemotherapy (see above).
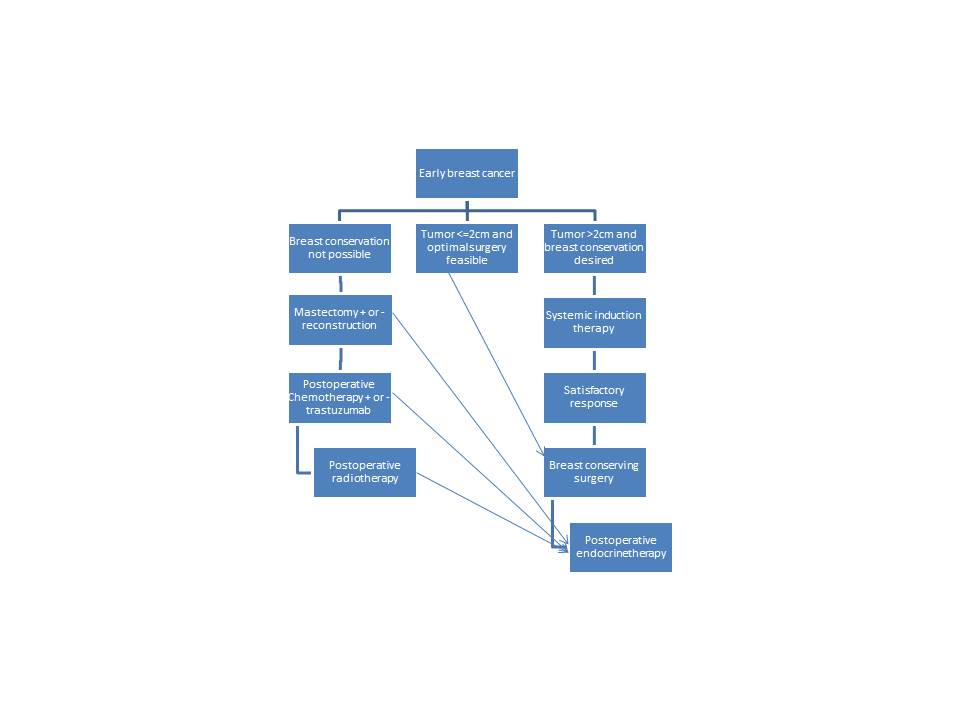
Adjuvant Chemotherapy for Early-Stage Breast Cancer
Risk Stratification
The decision of whether a patient should or not receive adjuvant chemotherapy is generally made by estimating the individual's risk for recurrence and the expected benefit of therapy. Risk stratification is based on tumor size, nodal status, histologic grade, hormone receptor, and Her-2 statuses.
Hormone Receptor Status
Hormone receptor status is a definite prognostic and predictive factor. Most breast cancers are Estrogen receptor- positive. ER-negative tumors have a higher risk of recurrence from 1 to 2 years after surgery that declines rapidly thereafter; however, ER-positive tumors preserve the ability to recur many years after surgery. The role of ER status as a predictive factor was assessed in a retrospective subset analysis of three cooperative group adjuvant chemotherapy trials in women who had node-positive breast cancer results showed that absolute benefits due to chemotherapy were greater for patients with ER-negative compared with ER-positive tumors: 22.8% more ER-negative patients survived to 5 years disease-free if receiving chemotherapy vs 7.0% for ER-positive patients; corresponding improvements for overall survival were 16.7% vs 4.0%.[9]
HER2 Status
The HER2 receptor is overexpressed in approximately 20% to 25% of breast cancers.[10] HER2 status is an important predictive factor. There is evidence that Trastuzumab exhibits synergy when administered in combination with other cytotoxic therapies, such as taxanes and vinorelbine, among HER2-positive patients. [11]
Oncotype DX
It is a multigene assay used to predict the recurrence risk among women with ER-positive, node-negative, early-stage breast cancer. The 21-gene expression profile categorizes a woman's risk for recurrence as low, intermediate, or high, with associated 10-year distant recurrence rates of 6.8%, 14.3%, and 30.5%, respectively. This assay may be a useful tool in the risk-benefit calculus, ensuring that truly low-risk women are not overtreated and exposed to the deleterious effects of treatment and that women at a higher risk for recurrence may be considered for a more aggressive therapeutic strategy. The ideal management strategy for women at intermediate risk for recurrence is uncertain, however.[10] Chemotherapy is not indicated for patients with low recurrence score (ie, <18) who only benefit from endocrine therapy with tamoxifen or an aromatase inhibitor. Chemotherapy is indicated for women who have a high recurrence score (>31).
Indications for systemic chemotherapy in lymph node-negative, estrogen receptor–negative, early-stage breast cancer
There is a modest risk for recurrence for women who have node-negative breast cancer that may be reduced with adjuvant systemic therapy.[10] Three clinical trials conducted by the National Surgical Adjuvant Breast and Bowel Project (NSABP) concluded that a significant risk for recurrence and death persisted among this “low-risk” population after surgery, which could be reduced with adjuvant chemotherapy. [12] In the NSABP trials, CMF (Cyclophosphamide, Methotrexate, and 5-FU) administration every 4 weeks or AC (Doxorubicin and Cyclophosphamide) administration every 3 weeks for four cycles showed same Outcomes in all age groups. Because of the long-term toxicity associated with anthracyclines, CMF is a reasonable option for patients who have lower-risk tumors. Adjuvant trastuzumab (Trastuzumab) is commonly administered for patients who have HER2-positive node-positive and high-risk, node-negative tumors, frequently in combination with a anthracycline and a taxane (AC-T).
Indications for systemic chemotherapy in lymph node-negative, estrogen receptor–positive, early-stage breast cancer
A recent study was conducted to look at the effect of Oncotype DX recurrence score on treatment recommendations for patients with estrogen receptor-positive early stage breast cancer and the correlation with the estimation of recurrence risk by breast cancer specialists.[13] The study had two primary endpoints:
- To evaluate the impact of Oncotype DX recurrence scores (RS) on chemotherapy recommendations
- To compare the estimated recurrence risk predicted by breast oncology specialists to RS.
154 patients with ER-positive early-stage breast cancer and available RS results were selected. Clinicopathologic data were provided to four surgeons, four medical oncologists, and four pathologists. Participants were asked to estimate the recurrence risk category and offer their chemotherapy recommendations initially without and later with knowledge of RS results. The three most important clinicopathologic features guiding their recommendations were requested. 95 (61.7%), 45 (29.2%), and 14 (9.1%) tumors were low, intermediate, and high risk by RS, respectively.
RS significantly correlated with tumor grade, mitotic activity, lymphovascular invasion, hormone receptor, and HER2/neu status. Estimated recurrence risk by participants agreed with RS in 54.2% ± 2.3% of cases. Without and with knowledge of RS, 82.3% ± 1.3% and 69.0% ± 6.9% of patients may be overtreated, respectively (p = 0.0322). Inclusion of RS data resulted in a 24.9% change in treatment recommendations. There was no significant difference in recommendations between groups of participants. Breast oncology specialists tended to overestimate the risk of tumor recurrence compared with RS. The study concluded that the Oncotype DX recurrence score provides useful information that improves patient selection for chemotherapy and changes treatment recommendations in approximately 25% of cases.
Adjuvant chemotherapy regimens for early stage breast cancer
- Despite the controversy regarding the superiority of anthracycline-containing regimens to CMF combination chemotherapy, anthracycline-based regimens are believed to be significantly superior and are the most commonly used regimens in the US unless a contraindication to anthracyclines (heart disease) precludes their use.
- The CMF regimen is reserved for older patients with early-stage breast cancer and patients with low-risk tumors.
- Standard adjuvant anthracycline-containing regimens include adriamycin and cyclophosphamide AC; cyclophosphamide, doxorubicin, and 5-fluorouracil (CAF); 5-fluorouracil, doxorubicin, and cyclophosphamide (FAC); 5-fluorouracil, epirubicin, and cyclophosphamide (FEC or CEF); docetaxel, doxorubicin, and cyclophosphamide (TAC); and AC followed by paclitaxel (AC-T) or docetaxel (AC-D).[10]
- Several studies have demonstrated the superiority of taxane-containing regimens with paclitaxel or docetaxel in the adjuvant setting, where the addition of 4 cycles of paclitaxel after the completion of a standard course of AC improved the disease-free and overall survival of patients with early breast cancer.[14]
- The American Society of Clinical Oncology guidelines suggest the use of anthracycline-containing regimens, mainly in patients with node-positive tumors or HER2 overexpression.[15]
- Taxanes should be added in case of node-positive tumors and high risk (large or ER-negative) node-negative breast cancer.
- Four 3-week cycles of AC (threshold doses for adriamycin and cyclophosphamide 60 mg/m2 and 600 mg/m2, respectively) followed by four 3-week cycles of paclitaxel is a very popular anthracycline-containing regimen. Currently, it has been largely replaced with the dose-dense (ie, every 2 weeks) AC followed by dose-dense paclitaxel, where dose density was found to improve clinical outcomes significantly.[16]
- Concomitant administration of chemotherapy and radiotherapy is not recommended due to increased toxicity
- In clinical practice, patients usually receive postoperative radiation therapy after chemotherapy. This practice does not adversely affect outcome for local-regional recurrence.[17]
Endocrine Therapy
- Endocrine therapy is based on the fact that estrogen receptor-positive tumors are highly estrogen-dependent for growth.
- Endocrine therapy agents for breast cancer are meant to block the effect the estrogen growth effect on breast cancer cells via several mechanisms:
- Blocking the estrogen receptor (eg, Selective Estrogen Receptor Modulators (SERMs) like Tamoxifen)
- Down-regulating and degrading the estrogen receptor (eg, Fulvestrant which is a pure estrogen antagonist)[18]
- Decreasing estrogen synthesis by blocking the enzyme called Aromatase, which converts androgens to estrogens. Aromatase inhibitors include many agents like Anastrozole, Letrozole, and Exemestane
- Decreasing estrogen level by:
- Shutting down the hypothalamus-pituitary-ovarian axis by means of GnRH agonists, which block the hypothalamic signal that normally promotes estrogen synthesis by the ovaries
- Ablating the ovaries (oophorectomy)
- Endocrine therapy should be the first-line treatment for ER-positive metastatic breast cancer unless there is significant visceral involvement (the so-called visceral crisis (ie, liver or lung disease)) where first-line chemotherapy should be offered first.
- There is no survival advantage for the combined administration of chemotherapy and endocrine therapy over either single therapy.[19] [20]
The factors that need to be taken into account when considering what endocrine therapy is appropriate for a particular patient include:[5]
- Whether or not they have had previous endocrine therapy (including as an adjuvant)
- If so, which agent
- The extent and duration of any previous response to endocrine therapy
- Menopausal status
Tamoxifen
- As mentioned above, Tamoxifen (TAM) is a SERM meaning that its effect on estrogen receptors depends on the target tissue. In fact, TAM inhibits breast cancer growth by competitive blockade of the estrogen receptor in breast tissue. However, it has agonistic activity in other tissues such as bone, uterus and blood vessels which is responsible for the side effects observed in patients on TAM, such as venous thromboembolism and uterine cancer.
- TAM is the first-line recommended endocrine agent for premenopausal metastatic breast cancer patients and the second-line agent for postmenopausal patients relapsing on/after aromatase inhibitors.
- Small, randomized trials in premenopausal women have found that oophorectomy is not superior to TAM.[21]
- Tamoxifen is a first-line treatment to men with ER-positive advanced breast cancer.[5]
- An oral dose of 20 mg daily is the standard of care.
Aromatase Inhibitors
Aromatase Inhibitors (AIs) are drugs that suppress the enzyme aromatase present in many tissues such as the gonads, the adipose tissue, the placenta, and the endometrium. By blocking this enzyme, estrogen synthesis is suppressed and thus the trophic effects of estrogen on the breast tissue are rendered minimal. Aromatase inhibitor therapy is ONLY effective in suppressing estrogen levels in postmenopausal women.[5] Because of that, measurement of serum follicle-stimulating hormone (FSH) levels can help selecting the proper patient population.
- A Phase III study of letrozole versus tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women concluded that letrozole was superior to tamoxifen in first-line endocrine therapy in postmenopausal women with advanced breast cancer.[22]
- In a randomized, double-blind, multicenter study designed to evaluate anastrozole (an AI), it was found that there was a significant increase in time to progression and a lower incidence of thromboembolic events and vaginal bleeding with anastrozole. These findings indicate that anastrozole should be considered as first-line therapy for postmenopausal women with advanced breast cancer.[23]
Offer an AI to: [5]
- postmenopausal women with ER-positive breast cancer and no prior history of endocrine therapy
- postmenopausal women with ER-positive breast cancer previously treated with tamoxifen
All aromatase inhibitors appear to be equally effective in terms of the primary outcome (overall survival).
The most commonly used AIs are the following:
- Letrozole (Femara): 2.5 mg PO daily
- Anastrozole (Arimidex): 1 mg PO daily
- Exemestane (Aromasin) 25 mg PO daily
Ovarian suppression with GnRH agonists
Ovarian suppression is indicated for premenopausal and perimenopausal women who have previously been treated with tamoxifen and then experience disease progression.[5] The combination of a GnRH agonist and tamoxifen is superior to GnRH agonist alone in premenopausal women with advanced breast cancer. Therefore, if a premenopausal woman with advanced breast cancer is thought to be suitable for endocrine treatment, the combination of a GnRH agonist plus tamoxifen be considered as the new standard treatment.[24]
Fulvestrant: The antiestrogen agent
Fulvestrant (Faslodex) is an estrogen receptor (ER) antagonist that downregulates the ER and has no agonist effects.[25] Fulvestrant is an effective, and well-tolerated treatment for advanced breast cancer in postmenopausal women whose disease progressed on prior endocrine therapy including Tamoxifen.[26][27]
Biologic Therapy: Trastuzumab and Lapatinib
- Monotherapy with trastuzumab is active and well tolerated as first-line treatment of women with metastatic breast cancer with HER2 overexpression.
- Trastuzumab combination therapy with a taxane, vinorelbine, or platinum (carboplatin or cisplatin) has higher response rates than monotherapy. For instance, paclitaxel and trastuzumab have shown higher response rates (41% vs. 17%), time to progression (6.9 vs 3.0 months) and a trend toward overall survival (22.1 vs 18.4 months) compared to paclitaxel alone.[1]
- Concurrent administration of trastuzumab and anthracyclines is not recommended due to enhanced cardiotoxicity.[1]
- Trastuzumab lacks CNS penetration. Consequently, brain metastases tend to progress when a patient is on trastuzumab though the disease is controlled systemically.
- For patients who are receiving treatment with trastuzumab for advanced breast cancer, it can be discontinued at the time of disease progression outside the central nervous system. Do not discontinue trastuzumab if disease progression is within the central nervous system alone.[5]
- Lapatinib is a HER2 and EGFR receptor tyrosine kinase inhibitor and is given orally (vs trastuzumab which is given IV).
- Lapatinib in combination with trastuzumab significantly improved progression-free survival versus lapatinib alone in trastuzumab-refractory metastatic breast cancer, thus offering a chemotherapy-free option with an acceptable safety profile to patients with Her2-positive metastatic breast cancer.[28]
- Lapatinib and capecitabine have shown to be superior to capecitabine alone in women with HER2-positive advanced breast cancer that has progressed after treatment with regimens that included an anthracycline, a taxane, and trastuzumab.[29] Consequently, if progression of disease occurs, Lapatinib and capecitabine are recommended concomitantly.
References
- ↑ 1.0 1.1 1.2 Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A et al. (2001) Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med 344 (11):783-92. DOI:10.1056/NEJM200103153441101 PMID: 11248153
- ↑ Vogel CL, Cobleigh MA, Tripathy D, Gutheil JC, Harris LN, Fehrenbacher L et al. (2002) Efficacy and safety of trastuzumab as a single agent in first-line treatment of HER2-overexpressing metastatic breast cancer. J Clin Oncol 20 (3):719-26. PMID: 11821453
- ↑ Alvarez RH, Guarneri V, Icli F, Johnston S, Khayat D, Loibl S et al. (2011) Bevacizumab Treatment for Advanced Breast Cancer. Oncologist ():. DOI:10.1634/theoncologist.2011-0113 PMID: 21976315
- ↑ Miles DW, Chan A, Dirix LY, Cortés J, Pivot X, Tomczak P et al. (2010) Phase III study of bevacizumab plus docetaxel compared with placebo plus docetaxel for the first-line treatment of human epidermal growth factor receptor 2-negative metastatic breast cancer. J Clin Oncol 28 (20):3239-47. DOI:10.1200/JCO.2008.21.6457 PMID: 20498403
- ↑ 5.0 5.1 5.2 5.3 5.4 5.5 5.6 [[]]. PMID 21901868. Missing or empty
|title=(help);|access-date=requires|url=(help) - ↑ Wilcken N, Dear R (2008) Chemotherapy in metastatic breast cancer: A summary of all randomised trials reported 2000-2007. Eur J Cancer 44 (15):2218-25. DOI:10.1016/j.ejca.2008.07.019 PMID: 18722111
- ↑ von Minckwitz, G (Mar 2007). "Docetaxel/anthracycline combinations for breast cancer treatment". Expert Opinion on Pharmacotherapy. 8 (4): 485–495.
- ↑ "Primary-Breast-Cancer".
- ↑ Berry DA, Cirrincione C, Henderson IC, Citron ML, Budman DR, Goldstein LJ et al. (2006) Estrogen-receptor status and outcomes of modern chemotherapy for patients with node-positive breast cancer. JAMA 295 (14):1658-67. DOI:10.1001/jama.295.14.1658 PMID: 16609087
- ↑ 10.0 10.1 10.2 10.3 McArthur, H.L. Hudis, C.A. (2007). Adjuvant Chemotherapy for Early-Stage Breast Cancer. Hematology/Oncology Clinics of North America, 21, 207-222. http://dx.doi.org/10.1016/j.hoc.2007.03.008.
- ↑ Marty M, Cognetti F, Maraninchi D, Snyder R, Mauriac L, Tubiana-Hulin M et al. (2005) Randomized phase II trial of the efficacy and safety of trastuzumab combined with docetaxel in patients with human epidermal growth factor receptor 2-positive metastatic breast cancer administered as first-line treatment: the M77001 study group. J Clin Oncol 23 (19):4265-74. DOI:10.1200/JCO.2005.04.173 PMID: 15911866
- ↑ Fisher B, Jeong JH, Anderson S, Wolmark N (2004) Treatment of axillary lymph node-negative, estrogen receptor-negative breast cancer: updated findings from National Surgical Adjuvant Breast and Bowel Project clinical trials. J Natl Cancer Inst 96 (24):1823-31. DOI:10.1093/jnci/djh338 PMID: 15601638
- ↑ Joh JE, Esposito NN, Kiluk JV, Laronga C, Lee MC, Loftus L et al. (2011) The Effect of Oncotype DX Recurrence Score on Treatment Recommendations for Patients with Estrogen Receptor-Positive Early Stage Breast Cancer and Correlation with Estimation of Recurrence Risk by Breast Cancer Specialists. Oncologist ():. DOI:10.1634/theoncologist.2011-0045 PMID: 22016474
- ↑ Henderson IC, Berry DA, Demetri GD, Cirrincione CT, Goldstein LJ, Martino S et al. (2003) Improved outcomes from adding sequential Paclitaxel but not from escalating Doxorubicin dose in an adjuvant chemotherapy regimen for patients with node-positive primary breast cancer. J Clin Oncol 21 (6):976-83. PMID: 12637460
- ↑ Harris L, Fritsche H, Mennel R, Norton L, Ravdin P, Taube S et al. (2007) American Society of Clinical Oncology 2007 update of recommendations for the use of tumor markers in breast cancer. J Clin Oncol 25 (33):5287-312. DOI:10.1200/JCO.2007.14.2364 PMID: 17954709
- ↑ Citron ML, Berry DA, Cirrincione C, Hudis C, Winer EP, Gradishar WJ et al. (2003) Randomized trial of dose-dense versus conventionally scheduled and sequential versus concurrent combination chemotherapy as postoperative adjuvant treatment of node-positive primary breast cancer: first report of Intergroup Trial C9741/Cancer and Leukemia Group B Trial 9741. J Clin Oncol 21 (8):1431-9. DOI:10.1200/JCO.2003.09.081 PMID: 12668651
- ↑ Metz JM, Schultz DJ, Fox K, Mathews A, Glick J, Solin LJ (2000) Analysis of outcomes for high-risk breast cancer based on interval from surgery to postmastectomy radiation therapy. Cancer J 6 (5):324-30. PMID: 11079172
- ↑ Kansra S, Yamagata S, Sneade L, Foster L, Ben-Jonathan N (2005) Differential effects of estrogen receptor antagonists on pituitary lactotroph proliferation and prolactin release. Mol Cell Endocrinol 239 (1-2):27-36. DOI:10.1016/j.mce.2005.04.008 PMID: 15950373
- ↑ Stockler M, Wilcken NR, Ghersi D, Simes RJ (2000) Systematic reviews of chemotherapy and endocrine therapy in metastatic breast cancer. Cancer Treat Rev 26 (3):151-68. DOI:10.1053/ctrv.1999.0161 PMID: 10814559
- ↑ Fossati R, Confalonieri C, Torri V, Ghislandi E, Penna A, Pistotti V et al. (1998) Cytotoxic and hormonal treatment for metastatic breast cancer: a systematic review of published randomized trials involving 31,510 women. J Clin Oncol 16 (10):3439-60. PMID: 9779724
- ↑ Buchanan RB, Blamey RW, Durrant KR, Howell A, Paterson AG, Preece PE et al. (1986) A randomized comparison of tamoxifen with surgical oophorectomy in premenopausal patients with advanced breast cancer. J Clin Oncol 4 (9):1326-30. PMID: 3528402
- ↑ Mouridsen H, Gershanovich M, Sun Y, Perez-Carrion R, Boni C, Monnier A, Apffelstaedt J, Smith R, Sleeboom HP, Jaenicke F, Pluzanska A, Dank M, Becquart D, Bapsy PP, Salminen E, Snyder R, Chaudri-Ross H, Lang R, Wyld P, Bhatnagar A (2003). "Phase III study of letrozole versus tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women: analysis of survival and update of efficacy from the International Letrozole Breast Cancer Group". Journal of Clinical Oncology : Official Journal of the American Society of Clinical Oncology. 21 (11): 2101–9. doi:10.1200/JCO.2003.04.194. PMID 12775735. Retrieved 2011-12-02. Unknown parameter
|month=ignored (help) - ↑ Nabholtz JM, Buzdar A, Pollak M, Harwin W, Burton G, Mangalik A et al. (2000) Anastrozole is superior to tamoxifen as first-line therapy for advanced breast cancer in postmenopausal women: results of a North American multicenter randomized trial. Arimidex Study Group. J Clin Oncol 18 (22):3758-67. PMID: 11078488
- ↑ Klijn JG, Blamey RW, Boccardo F, Tominaga T, Duchateau L, Sylvester R et al. (2001) Combined tamoxifen and luteinizing hormone-releasing hormone (LHRH) agonist versus LHRH agonist alone in premenopausal advanced breast cancer: a meta-analysis of four randomized trials. J Clin Oncol 19 (2):343-53. PMID: 11208825
- ↑ Osborne CK, Wakeling A, Nicholson RI (2004) Fulvestrant: an oestrogen receptor antagonist with a novel mechanism of action. Br J Cancer 90 Suppl 1 ():S2-6. DOI:10.1038/sj.bjc.6601629 PMID: 15094757
- ↑ Howell A, Robertson JF, Quaresma Albano J, Aschermannova A, Mauriac L, Kleeberg UR et al. (2002) Fulvestrant, formerly ICI 182,780, is as effective as anastrozole in postmenopausal women with advanced breast cancer progressing after prior endocrine treatment. J Clin Oncol 20 (16):3396-403. PMID: 12177099
- ↑ Osborne CK, Pippen J, Jones SE, Parker LM, Ellis M, Come S et al. (2002) Double-blind, randomized trial comparing the efficacy and tolerability of fulvestrant versus anastrozole in postmenopausal women with advanced breast cancer progressing on prior endocrine therapy: results of a North American trial. J Clin Oncol 20 (16):3386-95. PMID: 12177098
- ↑ Blackwell KL, Burstein HJ, Storniolo AM, Rugo H, Sledge G, Koehler M et al. (2010) Randomized study of Lapatinib alone or in combination with trastuzumab in women with ErbB2-positive, trastuzumab-refractory metastatic breast cancer. J Clin Oncol 28 (7):1124-30. DOI:10.1200/JCO.2008.21.4437 PMID: 20124187
- ↑ Geyer CE, Forster J, Lindquist D, Chan S, Romieu CG, Pienkowski T et al. (2006) Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N Engl J Med 355 (26):2733-43. DOI:10.1056/NEJMoa064320 PMID: 17192538