Arbidol
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | N/A |
| Metabolism | N/A - |
| Elimination half-life | 17-21 hours |
| Excretion | renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
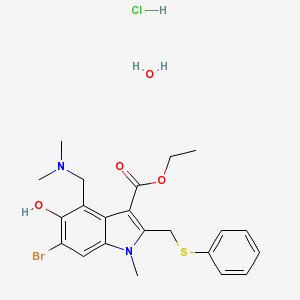
| Formula | C22H28BrClN2O4S |
| Molar mass | 531.891 g/mol |
|
WikiDoc Resources for Arbidol |
|
Articles |
|---|
|
Most recent articles on Arbidol |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Arbidol at Clinical Trials.gov Clinical Trials on Arbidol at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Arbidol
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Arbidol Risk calculators and risk factors for Arbidol
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Arbidol |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Arbidol) is an antiviral drug manufactured by Masterlek in Moscow, Russia.
Chemically, Arbidol features an indole core, functionalized at all positions but one with different substituents.
Uses
It is an alternative to Tamiflu (manufactured by Roche Pharmaceuticals) used in the fight against avian influenza. Its antiviral inhibitory effect is still being tested and the current results range from being well accepted in pharmaceutical industry to accepted with a dose of suspicion.
The drug has mainly been tested in Russia[1] and China,[2] and has been shown to be effective against avian flu,[3] suggesting it might be a more affordable and cost-effective drug than the widely used Tamiflu.
There is some evidence that it may be more effective at preventing infections from RNA viruses than from DNA viruses.[4]
It has also been investigated as a candidate drug against hepatitis C.[5]
Mechanism of action
The drug exhibits immunomodulation[6] as well as a specific anti-influenza action against the influenza A and influenza B viruses.
It prevents contact between the virus and host cells and penetration of virus particles into the cell by inhibiting the fusion of the virus lipid shell to the cell membranes. [7]
It possesses interferon inducing action, by stimulating the humoral reaction and the phagocytic function of macrophages.
Dosage forms
The drug is manufactured in the form of tablets and capsules, each tablet containing Arbidol® as its active ingredient (50 mg and 100 mg).
Pharmacokinetics and usage
Side effects in children include sensitization to the drug. No known overdose cases have been reported and allergic reactions are limited to people with hypersensitivity.
References
- ↑ Leneva IA, Fediakina IT, Gus'kova TA, Glushkov RG (2005). "[Sensitivity of various influenza virus strains to arbidol. Influence of arbidol combination with different antiviral drugs on reproduction of influenza virus A]". Ter. Arkh. (in Russian). 77 (8): 84–8. PMID 16206613.
- ↑ Wang MZ, Cai BQ, Li LY; et al. (2004). "[Efficacy and safety of arbidol in treatment of naturally acquired influenza]". Zhongguo Yi Xue Ke Xue Yuan Xaue Bao (in Chinese). 26 (3): 289–93. PMID 15266832.
- ↑ "Works as well as Tamiflu". Retrieved 2008-02-22.
- ↑ Shi L, Xiong H, He J; et al. (2007). "Antiviral activity of arbidol against influenza A virus, respiratory syncytial virus, rhinovirus, coxsackie virus and adenovirus in vitro and in vivo". Arch. Virol. 152 (8): 1447–55. doi:10.1007/s00705-007-0974-5. PMID 17497238.
- ↑ Pécheur EI, Lavillette D, Alcaras F; et al. (2007). "Biochemical mechanism of hepatitis C virus inhibition by the broad-spectrum antiviral arbidol". Biochemistry. 46 (20): 6050–9. doi:10.1021/bi700181j. PMID 17455911.
- ↑ Glushkov RG, Gus'kova TA, Krylova LIu, Nikolaeva IS (1999). "[Mechanisms of arbidole's immunomodulating action]". Vestn. Akad. Med. Nauk SSSR (in Russian) (3): 36–40. PMID 10222830.
- ↑ Boriskin YS, Pécheur EI, Polyak SJ (2006). "Arbidol: a broad-spectrum antiviral that inhibits acute and chronic HCV infection". Virol. J. 3: 56. doi:10.1186/1743-422X-3-56. PMID 16854226.
External links
- "Мастерлек" Pharmaceuticals, Moscow, Russia. Patent number № 2033157, Registry number № 003610/01.
- "Arbidol antiviral for influeza (flu), hepatitis, herpes, SARS..." Retrieved 2007-09-08.
- Arbidol®
- English Translations for Arbidol clinical studies 1973-2007
|
Influenza Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Arbidol On the Web |
|
American Roentgen Ray Society Images of Arbidol |
- Pages with script errors
- CS1 maint: Multiple names: authors list
- CS1 maint: Unrecognized language
- CS1 maint: Explicit use of et al.
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Anti-influenza agents
- Infectious disease