Sandbox:Feham
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Feham Tariq, MD [2]
Obstructive lung disease
| Diseases | Clinical manifestations | Diagnosis | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Symptoms | Signs | Lab findings | PFT | Imaging | Gold standard | Other features | ||||||||||||||
| Cough | Dyspnea | Hemoptysis | Fever | Weight loss | Cyanosis | Clubbing | JVD | Peripheral edema | Auscultation | ABGs | FEV1/FVC | TLC | DLCO | |||||||
| CXR | CT scan | Other tests | ||||||||||||||||||
| Asthma[1] | + | + | ± | ± | − | − | − | − | − |
|
|
|
FEV1:FVC =<0.7 |
|
|
|
|
|
|
Associated with:
|
| Chronic bronchitis | + | + | ± | + | − | − | − | − | − |
|
|
|
|
|
|
|
|
Microbiological testing is done in cases of:
|
|
|
| Bronchiolitis | + | + | − | + | − | − | − | − | – |
|
|
|
|
|
|
|
|
|
Can be associated with:
| |
| Emphysema | + | + | – | + | + | + | + | – | – |
|
|
|
|
|
– |
|
|
|
|
|
| Bronchiectasis | + | + | + | + | – | + | + | – | – |
|
|
|
|
|
|
|
|
| ||
| Heart failure | + | + | – | – | – | + | – | + | + |
|
|
|
|
|
|
|
||||
| Tuberculosis | + | + | + | + | + | – | – | – | – |
|
|
|
|
|||||||
| Lymphangioleiomyomatosis | + | + | +(<5%) | - | - | - | +(rare) | - | + |
|
|
|
|
|
|
|
|
|
|
|
| Status Asthmaticus | + | + | - | ± | - | - | - | + | - |
|
|
|
|
|
|
|
- |
|
|
|
| Cystic fibrosis | + | + | + | + | - | + | + | - | - |
|
|
|
|
|
|
|||||
Pneumothorax pathophysiology
Anatomy and physiology of the thoracic cavity
- Thoracic cavity is defined as the space inside the chest that contains the heart,lungs, and several major blood vessels.
- On either side of the cavity, a pleural membrane covers the surface of lung (visceral pleura) and also lines the inside of the chest wall (parietal pleura).
- The two layers are separated by a small amount of lubricating serous fluid. The lungs are fully inflated within the cavity because the pressure inside the airways is higher than the pressure inside the pleural space. Despite the low pressure in the pleural space, air does not enter it because there are no natural connections to an air-containing passage, and the pressure of gases in the bloodstream is too low for them to be forced into the pleural space.[12] Therefore, a pneumothorax can only develop if air is allowed to enter, through damage to the chest wall or damage to the lung itself, or occasionally because microorganisms in the pleural space produce gas.[12]
Chest-wall defects are usually evident in cases of injury to the chest wall, such as stab or bullet wounds ("open pneumothorax"). In secondary spontaneous pneumothoraces, vulnerabilities in the lung tissue are caused by a variety of disease processes, particularly by rupturing of bullae (large air-containing lesions) in cases of severe emphysema. Areas of necrosis (tissue death) may precipitate episodes of pneumothorax, although the exact mechanism is unclear.[11] Primary spontaneous pneumothorax (PSP) has for many years been thought to be caused by "blebs" (small air-filled lesions just under the pleural surface), which were presumed to be more common in those classically at risk of pneumothorax (tall males) due to mechanical factors. In PSP, blebs can be found in 77% of cases, compared to 6% in the general population without a history of PSP.[23] As these healthy subjects do not all develop a pneumothorax later, the hypothesis may not be sufficient to explain all episodes; furthermore, pneumothorax may recur even after surgical treatment of blebs.[12] It has therefore been suggested that PSP may also be caused by areas of disruption (porosity) in the pleural layer, which are prone to rupture.[11][12][23] Smoking may additionally lead to inflammation and obstruction of small airways, which account for the markedly increased risk of PSPs in smokers.[13] Once air has stopped entering the pleural cavity, it is gradually reabsorbed.[13]
Tension pneumothorax occurs when the opening that allows air to enter the pleural space functions as a one-way valve, allowing more air to enter with every breath but none to escape. The body compensates by increasing the respiratory rate and tidal volume (size of each breath), worsening the problem. Unless corrected, hypoxia (decreased oxygen levels) and respiratory arrest eventually follow.[14] . The key issue is the spontaneous occurrence of a communication between the alveolar spaces and the pleura. Most authors believe that spontaneous rupture of a subpleural bleb, or of a bulla, is the cause of PSP
Although the majority of PSP patients, including children [14], present blebs or bullae (usually at the apices of the lungs) (fig. 1) [15,16,17,18], it is unclear how often these lesions are actually the site of air leakage [19,20,21]. Only a small number of blebs are ruptured at the time of thoracoscopy or surgery, whereas in the remaining cases other lesions are present, often referred to as ‘pleural porosity’ [19,20,21]: areas of disrupted mesothelial cells at the visceral pleura, replaced by an inflammatory elastofibrotic layer with increased porosity, allowing air leakage into the pleural space. The latter phenomenon may explain the high recurrence rates of up to 20% of bullectomy alone (without associated pleurodesis) as therapy [22,23,24,25]. The development of blebs, bullae and areas of pleural porosity may be linked to a variety of factors, including distal airway inflammation [21,22,23,24,25,26], hereditary predisposition [27], anatomical abnormalities of the bronchial tree [28], ectomorphic physiognomy with more negative intrapleural pressures [29] and apical ischemia [30] at the apices [31], low body mass index and caloric restriction [15, 32], and abnormal connective tissue [33, 34]. The role of increased plasma aluminium concentrations in the pathogenesis of PSP remains unresolved
Dysphagia screening
Early and systematic dysphagia screening by the Gugging Swallowing Screen method and intensified oral hygiene reduced the incidence of x-ray verified pneumonia. [5]
==Overview==
Mesenteric ischemia is a type of intestinal ischemia primarily affecting the small intestine. It is one of the life-threatening gastrointestinal vascular emergencies which requires prompt surgical/medical intervention depending upon the underlying cause.
It can be divided into occlusive/non-occlusive, arterial or venous, localized/generalized and superficial or transmural.[6] {{#ev:youtube|https://www.youtube.com/watch?v=lg2_ehm6I9Y%7C350}} new [7] V new [8] (A) Grade 1. Normal. No vascular congestion is present, and both the villous architecture and muscular layer are preserved. (B) Grade 2. Villous architecture is preserved, with some mucosal congestion and dilated capillaries (black arrow). (C) Grade 3. Mucosa is congested (black arrow) with loss of superficial glandular architecture, but deep villous architecture is preserved. (D) Grade 4. The mucosa is completely involved, with loss of all superficial and deep glandular architecture, but the muscular layer is preserved. (E) Grade 5. There is total loss of glandular architecture, and the muscularis propria shows degeneration, fragmentation, and myocyte death, all indicative of transmural infarction (black arrow).{| class="infobox" style="position: fixed; top: 65%; right: 10px; margin: 0 0 0 0; border: 0; float: right;" |- | {{#ev:youtube|https://www.youtube.com/watch?v=lg2_ehm6I9Y%7C350}} |- |}
Overview
The anatomy and physiology of the small intestine plays a vital role in the develpoment of mesenteric ischemia. Intestinal muscosa has a high metabolic rate and accordingly a high blood flow requirement. The majority of blood supply of the intestine comes from the superior mesenteric artery, with a collateral blood supply from superior and inferior pancreaticoduodenal arteries (branches of the celiac artery) as well as the inferior mesenteric artery. The splanchnic circulation (arteries supplying the viscera) receives 15-35% of the cardiac output, making it sensitive to the effects of decreased perfusion. Mesenteric ischemia occurs when intestinal blood supply is compromised by more than 50% of the original blood flow. This can lead to disrutpion of mucosal barrier, allowing the release of bacterial toxins (present in the intestinal lumen) and vasoactive mediators which ultimately lead to complete necrosis (cell death) of the intestinal mucosa. This can further progress to depression in myocardial activity, sepsis, multiorgan failure, and without prompt intervention, even death.[9]
Pathophysiology
schematically the pthophysiology can be presented as a tree diagram:
| Esophageal dysphagia | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solids & liquids (Neuromuscular) | Solids only (Mechanical obstruction) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Progressive | Intermittent | Intermittent | Progressive | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Scleroderma | Achalasia | Diffuse esophageal spasm | Lower esophageal ring | Cancer | Peptic stricture | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pathogenesis
Intestinal damage occurs in response to ischemic insult.[10]
Following factors play a role in the development of mesenteric ischemia.
(a) Mesenteric vascular anatomy (General circulation)
(b) Collateral circulation
(c) Response of mesenteric vasculature to autonomic stimuli
(d) Vasoactive and humoral factors
(a) Mesenteric vascular anatomy and physiology:
The arterial supply of the intestine originates from three major arteries:[11][12][13]
- Superior mesenteric artery (SMA)
- Supplies the small intestine, proximal and mid colon upto the splenic flexure.
- Inferior mesenteric artery (IMA)
- Supplies hind gut starting from the splenic flexure to the rectum.
- Celiac artery (CA)
- Supplies the foregut, hepatobiliary system and spleen.
- The venous system parallels the arterial branches and drains into the portal venous system.
- The mesenteric circulation recevies approximately 25% of the resting and 35% of the postprandial cardiac output.
- Mucosal and submucosal layers of the intestine receive 70% of the mesenteric blood flow, with the rest supplying the muscularis and serosal layers.
Commonly affected arteries:
- Embolus can typically lodge into points of normal anatomic narrowing.
- This makes superior mesenteric artery the most vulnerable site because of its relatively larger diameter (more blood flow) and low take off angle (more likely to from the aorta.
- The majority of emboli lodge 3-10cm distal to the origin of superior mesenteric artery, classically sparing the proximal jejunum and colon.
(b) Collateral circulation:
The role of collateral circulation in the development of mesenteric ischemia is as follows:[14][15][16]
- Intestines receive collateral blood supply at all levels from the superior and inferior pancreaticoduodenal arteries, branches of the celiac artery, which provides protection from ischemia.
- These arteries can compensate for 75% reduction in mesenteric blood flow for upto 12 hours, without substanial injury.
- An extensive collateral circulation protects the intestines from transient periods of inadequate perfusion. However, prolonged reduction in splanchnic blood flow leads to vasoconstriction in the affected vascular bed, and eventually reduces collateral blood flow.
- The SMA and IMA communicate via the marginal artery of Drummond and the meandering mesenteric artery.
- Collateralization between the IMA and systemic circulation occurs in the rectum as the superior rectal (hemorrhoidal) vessels merge with the middle rectal vessels from the internal iliac arteries.
- The areas lacking this collateralization are prone towards ischemia.
(c) Factors regulating the mesenteric blood flow:
Mechansims that control the regulation of vascular tone of mesenteric circulation in response to periods of stress such as systemic hypotension or postprandial state.
Physiologically mesenteric circulation is affected by:[17][18]
- Intrinsic regulatory system that includes metabolic and myogenic factors.
- Extrinsic regulatory system that includes neural and humoral factors.
Intrinsic regulation:
- Metabolic factors:
- Reduction in blood supply to the mesentery causes adaptive changes in the splanchnic circulation.
- A discrepancy between tissue oxyegn demand and supply raises the concentration of local metabolites such as hydrogen, potassium, carbon dioxide, and adenosine, resulting in vasodilation and hyperemia.
- Myogenic factors:
- Myogenic theory suggests that arteriolar tension receptors act to regulate vascular resistance in accordance with the transmural pressure.
- An acute decrease in perfusion pressure is compensated for by a reduction in arteriolar wall tension, thereby maintaining splanchnic blood flow.
Extrinsic regulation:
- Neural component:
- The extrinsic neural component of splanchnic circulatory regulation comprises the alpha-activated vasoconstrictor fibers.
- Intense activation of vasoconstrictor fibers through alpha-adrenergic stimulation results in vasoconstriction of small vessels and a decrease in mesenteric blood flow.
- After periods of prolonged alpha-adrenergic vasoconstriction, blood flow increases, presumably through β-adrenergic stimulation, which acts as a protective response.
- Although numerous types of neural stimulation (e.g. vagal, cholinergic, histaminergic, and sympathetic) can affect the blood supply of the gut, the adrenergic limb of the autonomic nervous system is the predominant neural influence on splanchnic circulation.
- Humoral component:
- Numerous endogenous and exogenous humoral factors affect the splanchnic circulation.
- Norepinephrine and high doses of epinephrine produce intense vasoconstriction by stimulating the adrenergic receptors.
- Other pharmacologic compounds that decrease splanchnic blood flow include:
- Vasopressin
- Phenylephrine
- Digoxin
- Low-dose dopamine causes splanchnic vasodilation, whereas higher doses lead to vasoconstriction by stimulating alpha adrenergic receptors.
- Exogenous agents that increase mesenteric blood flow include:
- Papaverine
- Adenosine
- Dobutamine
- Fenoldopam
- Sodium nitroprusside
- In addition, numerous natural neurotransmitters can serve as splanchnic vasodilators, including acetylcholine, histamine, nitric oxide, leukotrienes, thromboxane analogues, glucagon, and a couple of gastrointestinal hormones.
| Factors regulating mesenteric blood flow | |||
|---|---|---|---|
| Extrinsic reguatory system | |||
| Humoral (endogenous and exogenous) | Neural component | ||
| Decrease blood flow | Increase blood flow | Decrease blood flow | Increase blood flow |
|
|
|
|
| Intrinsic regulatory component | |||
| Decrease blood flow | Increase blood flow | ||
|
| ||
Areas prone to ischemia:
| Areas prone to ischemia | Blood supply | ||
|---|---|---|---|
|
End arteries of superior mesenteric artery | ||
|
End arteries of inferior mesenteric artery | ||
- The watershed areas that lack collateralization are as follows:
- Splenic flexure
- Supplied by the end arteries of SMA with no collateral circulation.
- Rectosigmoid junction
Supplied by the end arteries of IMA with no collateral circulation.
Mechanism of ischemia:
The sequence of events that take place in the small intestine subsequent to decreased blood flow:
| Ischemic insult | |||||||||||||||||||
| Decreased delivery of oxygen and nutrients | |||||||||||||||||||
| Disruption in cellular metabolism | |||||||||||||||||||
| Tissue injury due to hypoxia and reperfusion | |||||||||||||||||||
| Full thickness necrosis of the bowel | |||||||||||||||||||
| Perforation of the bowel wall | |||||||||||||||||||
- Mesenteric ischemia occurs when the blood supply to mesentery is reduced leading to disruption of cellular metabolism owing to oxygen and nutrient deficiency.
- In the first 4 hours following ischemia, necrosis of the mucosal villi occurs.
- Persistent ischemia for more than 6 hours results in transmural, mural or mucosal infarction, ultimately leading to bowel perforation.
- Prolonged ischemia leads to progressive vasoconstriction of the mesenetric vessels which raises the pressure in them resulting in lowering the collateral flow.
- This is followed by vasodilation, trying to restore blood flow to the area of ischemic insult which causes reperfusion injury.
- Reperfusion injury causes release of oxygen free radicals, toxic byproducts of ischemic injury and neutrophil activation.
The pathophysiology of mesenteric ischemia can be explained on the basis of etiology:[19][20]
- Acute mesenteric arterial embolism: Attributes to 50% cases of mesenteric ischemia.
- Mesenteric embolus can oringinate from the left atrium, associated with cardiac arrythmias such as atrial fibrillation.
- Recent myocardial infarction resulting in segmental wall motion abnormality leading to poor ejaction fraction and embolus formation.
- Infective endocarditis: vegetations on the cardiac valves resulting in turbulence in blood flow predisposing to formation of emboli into the blood stream.
- Acute mesenteric arterial thrombosis:
- 25% cases of mesenteric ischemia result from mesenteric arterial thrombosis.
- Most likely due to underlying atherosclerosis(plaque formation) leading to stenosis.
- An underlying plaque(fatty streak) in the superior mesenteric artery leads to critical stenosis over the years forming collaterals.
- It is thought that [disease name] is the result of / is mediated by / is produced by / is caused by either [hypothesis 1], [hypothesis 2], or [hypothesis 3].
- [Pathogen name] is usually transmitted via the [transmission route] route to the human host.
- Following transmission/ingestion, the [pathogen] uses the [entry site] to invade the [cell name] cell.
- [Disease or malignancy name] arises from [cell name]s, which are [cell type] cells that are normally involved in [function of cells].
- The progression to [disease name] usually involves the [molecular pathway].
- The pathophysiology of [disease/malignancy] depends on the histological subtype.
Genetics
- [Disease name] is transmitted in [mode of genetic transmission] pattern.
- Genes involved in the pathogenesis of [disease name] include [gene1], [gene2], and [gene3].
- The development of [disease name] is the result of multiple genetic mutations.
Associated Conditions
Gross Pathology
After gaining access, the SMA is cannulated and catheterized. To directly aspirate the thrombus, a series of wires and catheters are used to place a relatively stiff wire into the ileocolic branch of the SMA over which an introducer with a removable hub is placed proximal to the embolus in the SMA (typically a 7-Fr, 45-cm introducer [eg, Destination, Terumo]) [9]. Inside the introducer, a 6-Fr guiding catheter is introduced into the clot. The clot is then aspirated into the guiding catheter with a 20-mL syringe as the catheter is withdrawn over the wire. The hub of the introducer can be removed to clear any residual clots. Repeat arteriography is performed, and, if needed, repeated aspirations can be performed. An alternative to this method is an over-the-wire double lumen aspiration catheter (eg, Export), which may allow removal of smaller, more peripheral clots (image 4).
Catheter-directed thrombolysis is an alternative for cases of incomplete aspiration embolectomy or distal mesenteric embolization. In cases of incomplete aspiration embolectomy or distal embolization, local thrombolysis is a reasonable alternative in patients without peritonitis [52]. With the introducer placed in the proximal SMA, a 4-Fr end-hole catheter can be advanced up to the clot or a multiple sidehole catheter (holes over 10 cm) advanced through the clot. The catheter is secured at its exit site and a dressing applied to the access site. Low-dose heparin (500 units/hour) administered through the sheath prevents its thrombosis. Papaverine infusion should not be used concomitantly, as it can precipitate in the presence of heparin. Termination of the infusion and abdominal exploration are indicated for any patient who develops progressive symptoms or signs of ischemia. Provided the patient remains clinically stable, arteriography should be repeated within four hours, and if clot lysis is not demonstrated, the patient should be taken to the operating room for abdominal exploration. (See 'Surgery' below.)
A review of 20 case reports and seven small series using thrombolytic therapy for acute SMA occlusion reported angiographic resolution of the SMA occlusion in 43 out of 48 patients (90 percent) [53]. Most were treated with infusions of urokinase. The overall 30-day survival rate was 43 out of 48 patients (90 percent).
Mesenteric angioplasty/stenting — Mesenteric artery angioplasty/stenting can be performed in an antegrade fashion (device introduced via the aorta into the SMA) or retrograde (device introduced via the SMA distal to the obstruction) [38,54]. Open mesenteric stenting for acute mesenteric ischemia is discussed below. Antegrade stenting, which is more typically performed in the setting of chronic ischemia (or acute-on-chronic ischemia), is discussed separately. (See "Chronic mesenteric ischemia", section on 'Management'.)
Retrograde open mesenteric stenting — In selected patients, retrograde catheter-based mesenteric revascularization may be appropriate [36,55-58]. In settings where thrombectomy is unsuccessful in restoring arterial inflow and autogenous bypass conduit (saphenous vein, femoral [deep] vein) to perform surgical bypass is not available or cannot be expeditiously harvested, it may be possible to establish arterial inflow using retrograde catheterization of the superior mesenteric artery. If successful, this approach avoids the risk of contaminating a prosthetic vascular graft. With appropriate endovascular equipment and fluoroscopic imaging, the procedure is relatively straightforward, with reported results competitive with those of open surgical bypass.
SURGERY — Immediate surgery is indicated for patients with acute mesenteric ischemia with clinical symptoms or signs of advanced ischemia (eg, peritonitis, sepsis, pneumatosis intestinalis) [59].
Abdominal exploration/damage control — Laparotomy is indicated for patients with acute abdominal findings on exam indicating peritonitis. Laparotomy, rather than laparoscopy, may be safer and more expedient for evaluating the viscera in the face of grossly dilated bowel. The intestinal tract should be evaluated first for areas of impending or gross perforation; these areas should be immediately resected using a stapler to contain gross spillage. A general abdominal exploration should be performed, looking for obvious pathology or other signs of visceral embolization.
The extent and severity of intestinal ischemia, including the appearance of the abdominal contents (color, distention), peristalsis, arterial pulsations in the mesenteric arcades, and bleeding from cut surfaces, should be assessed. Although mesenteric arterial revascularization is preferably performed before bowel resection, areas of the small or large intestine that are clearly nonviable (ie, full-thickness ischemia with dilated, dark, paralyzed bowel (picture 4)) can be quickly resected using a damage control approach. Bowel of questionable viability that peristalses even a little should be left intact until after perfusion is restored, after which bowel viability should be reassessed. (See "Overview of damage control surgery and resuscitation in patients sustaining severe injury".)
Revascularization
Embolectomy — Open superior mesenteric artery (SMA) embolectomy remains a good option and should be performed in those with indications for open surgical intervention. Mesenteric embolectomy is performed through a midline abdominal incision that allows full inspection of the bowel. The proximal superior mesenteric artery can be exposed at the root of the mesentery by retracting the transverse colon cephalad, mobilizing the distal duodenum, and palpating the SMA in the root of the mesentery (picture 5).
A transverse arteriotomy is made, through which 3-Fr or 4-Fr Fogarty embolectomy catheters can be introduced to extract the clot (picture 6). The catheters should be advanced distally as well as proximally. Absence of additional thrombus with multiple passes and restoration of pulsatile inflow confirms clearance of the artery. The vessel is flushed with heparin and the arteriotomy repaired primarily. If arterial inflow cannot be obtained, repeat mesenteric arteriography may be necessary. If inflow cannot be restored, bypass from the aorta or other location can be necessary. (See 'Mesenteric bypass' below.)
Mesenteric bypass — Revascularization of the SMA can be achieved in several ways [60-63]. Superior mesenteric artery bypass is the most practical method in the setting of acute mesenteric arterial occlusion. Other options include thromboendarterectomy and translocation; however, these techniques are less commonly used in the setting of acute mesenteric ischemia. Translocation (transection of the SMA distal to the occlusive lesion with reimplantation into the infrarenal aorta) is unlikely to be feasible in acute circumstances given the typically distal level of obstruction. For patients with chronic mesenteric ischemia (or possibly acute-on-chronic disease), thromboendarterectomy may be an option if disease is confined to the origin of the visceral vessels. (See "Chronic mesenteric ischemia", section on 'Management'.)
Mesenteric bypass constructs a graft from the chosen inflow vessel (eg, aorta, iliac artery) to a site distal to the occlusive lesion. Autologous reversed saphenous vein may be the preferred conduit, but polytetrafluoroethylene (PTFE) grafts reinforced with rings are a reasonable option for retrograde revascularizations to prevent kinking. However, in general, prosthetic reconstruction is discouraged in the acute setting, particularly in the face of abdominal contamination because of an increased high risk for graft infection. If native conduit is not available, angioplasty and stenting (antegrade or retrograde) may be a better option rather than placing a prosthetic graft within a contaminated field. (See 'Mesenteric angioplasty/stenting' above.)
●Antegrade bypass – The inflow is from the supraceliac aorta.
●Retrograde bypass – The inflow is from the infrarenal aorta or iliac arteries.
Mesenteric artery bypass has good long-term patency rates and high rates of symptom-free survival; however, perioperative mortality in the face of acute intestinal ischemia remains high [64]. (See 'Morbidity and mortality' below.)
Bowel and abdominal closure — Following open revascularization, the small bowel should be carefully reexamined for areas of irreversible ischemic injury, which may require resection. In one review of 83 patients requiring revascularization for acute mesenteric ischemia, 24 percent required resection of a median length of 43 cm of bowel [60]. The presence of Doppler signals over the serosal surface may be helpful in identifying potentially salvageable ischemic segments to be left in place for reevaluation at second-look operation; however, surgeon experience and visual inspection have been shown to be as accurate as other adjunctive diagnostic techniques in the intraoperative assessment of bowel viability. At the time of definitive abdominal closure, intravenous injection of fluorescein dye with inspection of the intestine illuminated via a Wood's lamp can assist in determining remaining bowel viability (picture 7).
Restoration of bowel continuity can be performed during the index surgery for well-demarcated, clearly viable bowel segments. If bowel viability is in question or the patient is hemodynamically unstable, a damage control approach can be undertaken by resecting the nonviable segments and stapling the small bowel closed awaiting a second-look procedure to restore bowel continuity.
The abdominal wall is left open when repeat laparotomy is planned, which is particularly likely if there has been a significant interval of ischemia that leads to bowel edema with reperfusion. If closure is elected (eg, no necrotic bowel, minimal ischemic time), abdominal compartment pressures should be monitored. A planned "second-look" laparotomy is frequently needed to reassess and resect irreversibly ischemic bowel. (See 'Second-look laparotomy and abdominal wall closure' below.)
POSTPROCEDURE CARE AND FOLLOW-UP — Patients with acute mesenteric ischemia are often very ill post-intervention, requiring intensive care management and nutritional support. In one study, the mean length of hospital stay was 23 days [35].
Second-look laparotomy and abdominal wall closure — A second-look laparotomy is needed for most patients after mesenteric revascularization for acute mesenteric arterial occlusion to reevaluate the bowel 24 to 48 hours after the initial operation. In a review of 93 patients undergoing arterial revascularization for mesenteric ischemia, 80 percent of patients underwent a second-look laparotomy, and among those patients, 28 percent had necrotic bowel requiring resection at the second operation [35]. (See "Management of the open abdomen in adults".)
If primary abdominal closure was elected (eg, no necrotic bowel, minimal ischemic time), abdominal compartment pressures should be monitored. (See "Abdominal compartment syndrome in adults", section on 'Measurement of intra-abdominal pressure'.)
Microscopic Pathology
- On microscopic histopathological analysis, [feature1], [feature2], and [feature3] are characteristic findings of [disease name].
The goal of surgical intervention for AMI consists of:
Re-establishment blood supply to the ischemic bowel.
Resection of all non-viable areas.
preservation of all possible bowel.
Intestinal viability is the maximum vital element influencing outcome in sufferers with AMI. Non-feasible gut, if unrecognized, effects in multi-machine organ dysfunction and in the end demise. activate laparotomy lets in for direct assessment of bowel viability.
After preliminary resuscitation, midline laparotomy should be done observed by means of assessment of all areas of the gut with choices for resection of all surely necrotic areas. In instances of uncertainty, intraoperative Doppler can be beneficial, as the presence of Doppler indicators over distal branches of SMA allows bowel conservation, heading off lengthy-time period disability. The SMA is without problems palpated with the aid of setting palms at the back of the basis of the mesentery. The SMA is recognized as a firm tubular shape, which may additionally or might not have a palpable pulse. in any other case, the SMA can also be reached via following the middle colic artery where it enters the SMA at the mesentery. Direct sharp dissection, exposing the artery from its surrounding mesenteric tissue, is needed for correct exposure to carry out revascularization. In instances of diagnostic uncertainties, arteriogram is the study of preference. it is able to be executed intraoperatively mainly in hybrid suites. ===Surgery===Mesenteric angioplasty/stenting — Mesenteric artery angioplasty/stenting can be performed in an antegrade fashion (device introduced via the aorta into the SMA) or retrograde (device introduced via the SMA distal to the obstruction) [38,54]. Open mesenteric stenting for acute mesenteric ischemia is discussed below. Antegrade stenting, which is more typically performed in the setting of chronic ischemia (or acute-on-chronic ischemia), is discussed separately. (See "Chronic mesenteric ischemia", section on 'Management'.)
Retrograde open mesenteric stenting — In selected patients, retrograde catheter-based mesenteric revascularization may be appropriate [36,55-58]. In settings where thrombectomy is unsuccessful in restoring arterial inflow and autogenous bypass conduit (saphenous vein, femoral [deep] vein) to perform surgical bypass is not available or cannot be expeditiously harvested, it may be possible to establish arterial inflow using retrograde catheterization of the superior mesenteric artery. If successful, this approach avoids the risk of contaminating a prosthetic vascular graft. With appropriate endovascular equipment and fluoroscopic imaging, the procedure is relatively straightforward, with reported results competitive with those of open surgical bypass.
SURGERY — Immediate surgery is indicated for patients with acute mesenteric ischemia with clinical symptoms or signs of advanced ischemia (eg, peritonitis, sepsis, pneumatosis intestinalis) [59].
Abdominal exploration/damage control — Laparotomy is indicated for patients with acute abdominal findings on exam indicating peritonitis. Laparotomy, rather than laparoscopy, may be safer and more expedient for evaluating the viscera in the face of grossly dilated bowel. The intestinal tract should be evaluated first for areas of impending or gross perforation; these areas should be immediately resected using a stapler to contain gross spillage. A general abdominal exploration should be performed, looking for obvious pathology or other signs of visceral embolization.
The extent and severity of intestinal ischemia, including the appearance of the abdominal contents (color, distention), peristalsis, arterial pulsations in the mesenteric arcades, and bleeding from cut surfaces, should be assessed. Although mesenteric arterial revascularization is preferably performed before bowel resection, areas of the small or large intestine that are clearly nonviable (ie, full-thickness ischemia with dilated, dark, paralyzed bowel (picture 4)) can be quickly resected using a damage control approach. Bowel of questionable viability that peristalses even a little should be left intact until after perfusion is restored, after which bowel viability should be reassessed. (See "Overview of damage control surgery and resuscitation in patients sustaining severe injury".)
Revascularization
Embolectomy — Open superior mesenteric artery (SMA) embolectomy remains a good option and should be performed in those with indications for open surgical intervention. Mesenteric embolectomy is performed through a midline abdominal incision that allows full inspection of the bowel. The proximal superior mesenteric artery can be exposed at the root of the mesentery by retracting the transverse colon cephalad, mobilizing the distal duodenum, and palpating the SMA in the root of the mesentery (picture 5).
A transverse arteriotomy is made, through which 3-Fr or 4-Fr Fogarty embolectomy catheters can be introduced to extract the clot (picture 6). The catheters should be advanced distally as well as proximally. Absence of additional thrombus with multiple passes and restoration of pulsatile inflow confirms clearance of the artery. The vessel is flushed with heparin and the arteriotomy repaired primarily. If arterial inflow cannot be obtained, repeat mesenteric arteriography may be necessary. If inflow cannot be restored, bypass from the aorta or other location can be necessary. (See 'Mesenteric bypass' below.)
Mesenteric bypass — Revascularization of the SMA can be achieved in several ways [60-63]. Superior mesenteric artery bypass is the most practical method in the setting of acute mesenteric arterial occlusion. Other options include thromboendarterectomy and translocation; however, these techniques are less commonly used in the setting of acute mesenteric ischemia. Translocation (transection of the SMA distal to the occlusive lesion with reimplantation into the infrarenal aorta) is unlikely to be feasible in acute circumstances given the typically distal level of obstruction. For patients with chronic mesenteric ischemia (or possibly acute-on-chronic disease), thromboendarterectomy may be an option if disease is confined to the origin of the visceral vessels. (See "Chronic mesenteric ischemia", section on 'Management'.)
Mesenteric bypass constructs a graft from the chosen inflow vessel (eg, aorta, iliac artery) to a site distal to the occlusive lesion. Autologous reversed saphenous vein may be the preferred conduit, but polytetrafluoroethylene (PTFE) grafts reinforced with rings are a reasonable option for retrograde revascularizations to prevent kinking. However, in general, prosthetic reconstruction is discouraged in the acute setting, particularly in the face of abdominal contamination because of an increased high risk for graft infection. If native conduit is not available, angioplasty and stenting (antegrade or retrograde) may be a better option rather than placing a prosthetic graft within a contaminated field. (See 'Mesenteric angioplasty/stenting' above.)
●Antegrade bypass – The inflow is from the supraceliac aorta.
●Retrograde bypass – The inflow is from the infrarenal aorta or iliac arteries.
Mesenteric artery bypass has good long-term patency rates and high rates of symptom-free survival; however, perioperative mortality in the face of acute intestinal ischemia remains high [64]. (See 'Morbidity and mortality' below.)
Bowel and abdominal closure — Following open revascularization, the small bowel should be carefully reexamined for areas of irreversible ischemic injury, which may require resection. In one review of 83 patients requiring revascularization for acute mesenteric ischemia, 24 percent required resection of a median length of 43 cm of bowel [60]. The presence of Doppler signals over the serosal surface may be helpful in identifying potentially salvageable ischemic segments to be left in place for reevaluation at second-look operation; however, surgeon experience and visual inspection have been shown to be as accurate as other adjunctive diagnostic techniques in the intraoperative assessment of bowel viability. At the time of definitive abdominal closure, intravenous injection of fluorescein dye with inspection of the intestine illuminated via a Wood's lamp can assist in determining remaining bowel viability (picture 7).
Restoration of bowel continuity can be performed during the index surgery for well-demarcated, clearly viable bowel segments. If bowel viability is in question or the patient is hemodynamically unstable, a damage control approach can be undertaken by resecting the nonviable segments and stapling the small bowel closed awaiting a second-look procedure to restore bowel continuity.
The abdominal wall is left open when repeat laparotomy is planned, which is particularly likely if there has been a significant interval of ischemia that leads to bowel edema with reperfusion. If closure is elected (eg, no necrotic bowel, minimal ischemic time), abdominal compartment pressures should be monitored. A planned "second-look" laparotomy is frequently needed to reassess and resect irreversibly ischemic bowel. (See 'Second-look laparotomy and abdominal wall closure' below.)
POSTPROCEDURE CARE AND FOLLOW-UP — Patients with acute mesenteric ischemia are often very ill post-intervention, requiring intensive care management and nutritional support. In one study, the mean length of hospital stay was 23 days [35].
Second-look laparotomy and abdominal wall closure — A second-look laparotomy is needed for most patients after mesenteric revascularization for acute mesenteric arterial occlusion to reevaluate the bowel 24 to 48 hours after the initial operation. In a review of 93 patients undergoing arterial revascularization for mesenteric ischemia, 80 percent of patients underwent a second-look laparotomy, and among those patients, 28 percent had necrotic bowel requiring resection at the second operation [35]. (See "Management of the open abdomen in adults".)
If primary abdominal closure was elected (eg, no necrotic bowel, minimal ischemic time), abdominal compartment pressures should be monitored. (See "Abdominal compartment syndrome in adults", section on 'Measurement of intra-abdominal pressure'.)
References
- ↑ Elbehairy AF, Raghavan N, Cheng S, Yang L, Webb KA, Neder JA; et al. (2015). "Physiologic characterization of the chronic bronchitis phenotype in GOLD grade IB COPD". Chest. 147 (5): 1235–1245. doi:10.1378/chest.14-1491. PMID 25393126.
- ↑ Gaeta M, Minutoli F, Girbino G, Murabito A, Benedetto C, Contiguglia R, Ruggeri P, Privitera S (2013). "Expiratory CT scan in patients with normal inspiratory CT scan: a finding of obliterative bronchiolitis and other causes of bronchiolar obstruction". Multidiscip Respir Med. 8 (1): 44. doi:10.1186/2049-6958-8-44. PMC 3710098. PMID 23835554.
- ↑ Park JE, Kim Y, Lee SW, Shim SS, Lee JK, Lee JH (2016). "The usefulness of low-dose CT scan in elderly patients with suspected acute lower respiratory infection in the emergency room". Br J Radiol. 89 (1060): 20150654. doi:10.1259/bjr.20150654. PMC 4846199. PMID 26861744.
- ↑ Espiritu JD, Ruppel G, Shrestha Y, Kleinhenz ME (June 2003). "The diffusing capacity in adult cystic fibrosis". Respir Med. 97 (6): 606–11. PMID 12814143.
- ↑ Sørensen RT, Rasmussen RS, Overgaard K, Lerche A, Johansen AM, Lindhardt T (2013). "Dysphagia screening and intensified oral hygiene reduce pneumonia after stroke". J Neurosci Nurs. 45 (3): 139–46. doi:10.1097/JNN.0b013e31828a412c. PMID 23636069.
- ↑ Corcos O, Nuzzo A (2013). "Gastro-intestinal vascular emergencies". Best Pract Res Clin Gastroenterol. 27 (5): 709–25. doi:10.1016/j.bpg.2013.08.006. PMID 24160929.
- ↑ Dunphy, J. E. (1936). Abdominal pain of vascular origin. The American Journal of the Medical Sciences, 192(1), 109-113.
- ↑ Oderich, Gustavo (2014). Mesenteric vascular disease : current therapy. New York: Springer. ISBN 9781493918461.
- ↑ Rosenblum JD, Boyle CM, Schwartz LB (1997). "The mesenteric circulation. Anatomy and physiology". Surg Clin North Am. 77 (2): 289–306. PMID 9146713.
- ↑ Sánchez-Fernández P, Mier y Díaz J, Blanco-Benavides R (2000). "[Acute mesenteric ischemia. Profile of an aggressive disease]". Rev Gastroenterol Mex. 65 (3): 134–40. PMID 11464607.
- ↑ Kumar S, Sarr MG, Kamath PS (2001). "Mesenteric venous thrombosis". N Engl J Med. 345 (23): 1683–8. doi:10.1056/NEJMra010076. PMID 11759648.
- ↑ Ha C, Magowan S, Accortt NA, Chen J, Stone CD (2009). "Risk of arterial thrombotic events in inflammatory bowel disease". Am J Gastroenterol. 104 (6): 1445–51. doi:10.1038/ajg.2009.81. PMID 19491858.
- ↑ Granger DN, Richardson PD, Kvietys PR, Mortillaro NA (1980). "Intestinal blood flow". Gastroenterology. 78 (4): 837–63. PMID 6101568.
- ↑ McKinsey JF, Gewertz BL (1997). "Acute mesenteric ischemia". Surg Clin North Am. 77 (2): 307–18. PMID 9146714.
- ↑ Walker TG (2009). "Mesenteric vasculature and collateral pathways". Semin Intervent Radiol. 26 (3): 167–74. doi:10.1055/s-0029-1225663. PMC 3036491. PMID 21326561.
- ↑ Fisher DF, Fry WJ (1987). "Collateral mesenteric circulation". Surg Gynecol Obstet. 164 (5): 487–92. PMID 3554567.
- ↑ Hansen MB, Dresner LS, Wait RB (1998). "Profile of neurohumoral agents on mesenteric and intestinal blood flow in health and disease". Physiol Res. 47 (5): 307–27. PMID 10052599.
- ↑ Schoenberg MH, Beger HG (1993). "Reperfusion injury after intestinal ischemia". Crit Care Med. 21 (9): 1376–86. PMID 8370303.
- ↑ Acosta S (2015). "Mesenteric ischemia". Curr Opin Crit Care. 21 (2): 171–8. doi:10.1097/MCC.0000000000000189. PMID 25689121.
- ↑ Acosta S, Ogren M, Sternby NH, Bergqvist D, Björck M (2005). "Clinical implications for the management of acute thromboembolic occlusion of the superior mesenteric artery: autopsy findings in 213 patients". Ann Surg. 241 (3): 516–22. PMC 1356992. PMID 15729076.
Risk Factors
Common Risk Factors
The following conditions pose a signifiacnt risk towards the development of mesenteric ischemia either by interrupting the blood flow through the artery or vein supplying the small intestine (e.g.thromboemboli) or by reducing the blood supply (e.g. vasoconstriction). Also, there are certain life-style related risk factors which predominantly cause mesenteric ischemia in the older age group. [1][2][3]
| Risk factors | ||
|---|---|---|
| Occlusive | Embolic | Atrial fibrillation |
| Cardiac arrhythmia | ||
| Valvular heart disease | ||
| Infective endocarditis | ||
| Recent myocardial infarction | ||
| Ventricular aneurysm | ||
| Aortic atherosclerosis | ||
| Thrombotic | Advanced age | |
| Low cardiac output states | ||
| Peripheral arterial disease | ||
| Traumatic injury | ||
Inherited thrombophilia-
| ||
| Acquired thrombophilia- malignancy, oral contraceptives intake. | ||
| Non-occlusive | Heart failure | |
| Aortic insufficiency | ||
| Septic shock | ||
| Vasoconstrictive drugs: | ||
| Cocaine abuse or ergot poisoning | ||
| Hemodialysis | ||
| Other causes | Lifestyle related risk factors:
Less common risk factors:
| |
- Common risk factors in the development of mesenteric ischemia include:
- Occlusive causes
- Embolic causes:[1]
- Atrial fibrillation
- Cardiac arrhythmias
- Valvular heart diseases
- Infective endocarditis
- Recent myocardial infarction
- Ventricular aneurysm
- Aortic atherosclerosis
- Aortic aneurysm
- Thrombotic causes:[2]
- Advanced age
- Low cardiac output states
- Traumatic injury
- Peripheral artery disease
- Occlusive causes
- Non-occlusive causes:[3]
- Heart failure
- Aortic insufficiency
- Septic shock
- Vasoconstrictive drugs(e.g. Digoxin, alpha-adrenergic agonists)
- Cocaine abuse or ergot poisoning
- Hemodialysis
Less Common Risk Factors
- Less common risk factors in the development of mesenteric ischemia include:
- Fibromuscular dysplasia
- Hepatitis
- Beta recpetor blocking agents
- Polyarteritis nodosa
Causes
Narrowing of the arteries that supply blood to the intestine causes mesenteric ischemia. The arteries that supply blood to the intestines run directly from the aorta. Mesenteric ischemia is often seen in people who have hardening of the arteries in other parts of the body (for example, those with coronary artery disease or peripheral vascular disease). The condition is more common in smokers and in patients with high blood pressure or blood cholesterol. Mesenteric ischemia may also be caused by an embolus that suddenly blocks one of the mesenteric arteries. The emboli usually come from the heart or aorta. These clots are more commonly seen in patients with arrhythmias, such as atrial fibrillation. They can be broadly classified into four categories:[4]
| Classification based on etiology | |||
|---|---|---|---|
| Etiology | Cause | Incidence | Examples |
| Occlusive causes | Aterial embolism | 50-70% |
|
| Arterial thrombosis | 15-25% |
| |
| Venous thrombosis | 5% |
| |
| Non-Occlusive causes | Non-occlusive ischemia | 20-30% |
|
History and symptoms
Overview
Mesenteric ischemia is not an isloated entity but a group of diseases, including mesenteric artery thromboembolic occlusion, meseenteric vein thrombosis and non occlusive, that impair the blood supply to the small intestine, leading to development of same clinical features. However, they can differentiate based on preexisting comorbidities.[5]
Symptoms
In order to diagnose the condition promptly, a special attention towards the history and symptoms should be given.[6][7]
Symptoms of mesenteric ischemia vary and can be acute (especially if embolic)[8], subacute, or chronic[9].
Case series report prevalence of clinical findings and provide the best available, yet biased, estimate of the sensitivity of clinical findings[10][11]. In a series of 58 patients with mesenteric ischemia due to mixed causes[11]:
- Abdominal pain was present in 95% (median of 24 hours duration). The other three patients presented with shock and metabolic acidosis.
- Nausea in 44%
- Vomiting in 35%
- Diarrhea in 35%
- Heart rate > 100 in 33%
- 'Blood per rectum' in 16% (not stated if this number also included occult blood - presumably not)
- Constipation 7%
History
- The majority of patients with [disease name] are asymptomatic.
OR
- The hallmark of [disease name] is [finding]. A positive history of [finding 1] and [finding 2] is suggestive of [disease name]. The most common symptoms of [disease name] include [symptom 1], [symptom 2], and [symptom 3].
- Symptoms of [disease name] include [symptom 1], [symptom 2], and [symptom 3].
History
Patients with [disease name]] may have a positive history of:
- [History finding 1]
- [History finding 2]
- [History finding 3]
Common Symptoms
Common symptoms of [disease] include:
- [Symptom 1]
- [Symptom 2]
- [Symptom 3]
Less Common Symptoms
Less common symptoms of [disease name] include
- [Symptom 1]
- [Symptom 2]
- [Symptom 3]
References
- ↑ 1.0 1.1 Fitzgerald T, Kim D, Karakozis S, Alam H, Provido H, Kirkpatrick J (2000). "Visceral ischemia after cardiopulmonary bypass". Am Surg. 66 (7): 623–6. PMID 10917470.
- ↑ 2.0 2.1 Martinelli I, Mannucci PM, De Stefano V, Taioli E, Rossi V, Crosti F; et al. (1998). "Different risks of thrombosis in four coagulation defects associated with inherited thrombophilia: a study of 150 families". Blood. 92 (7): 2353–8. PMID 9746774.
- ↑ 3.0 3.1 Acosta S, Ogren M, Sternby NH, Bergqvist D, Björck M (2006). "Fatal nonocclusive mesenteric ischaemia: population-based incidence and risk factors". J Intern Med. 259 (3): 305–13. doi:10.1111/j.1365-2796.2006.01613.x. PMID 16476108.
- ↑ Reinus JF, Brandt LJ, Boley SJ (1990). "Ischemic diseases of the bowel". Gastroenterol Clin North Am. 19 (2): 319–43. PMID 2194948.
- ↑ Yasuhara H (2005). "Acute mesenteric ischemia: the challenge of gastroenterology". Surg Today. 35 (3): 185–95. doi:10.1007/s00595-004-2924-0. PMID 15772787.
- ↑ Meyer T, Klein P, Schweiger H, Lang W (1998). "[How can the prognosis of acute mesenteric artery ischemia be improved? Results of a retrospective analysis]". Zentralbl Chir. 123 (3): 230–4. PMID 9586181.
- ↑ Stone JR, Wilkins LR (2015). "Acute mesenteric ischemia". Tech Vasc Interv Radiol. 18 (1): 24–30. doi:10.1053/j.tvir.2014.12.004. PMID 25814200.
- ↑ Oldenburg WA, Lau LL, Rodenberg TJ, Edmonds HJ, Burger CD (2004). "Acute mesenteric ischemia: a clinical review". Arch. Intern. Med. 164 (10): 1054–62. doi:10.1001/archinte.164.10.1054. PMID 15159262.
- ↑ Font VE, Hermann RE, Longworth DL (1989). "Chronic mesenteric venous thrombosis: difficult diagnosis and therapy". Cleveland Clinic journal of medicine. 56 (8): 823–8. PMID 2691119.
- ↑ Levy PJ, Krausz MM, Manny J (1990). "Acute mesenteric ischemia: improved results--a retrospective analysis of ninety-two patients". Surgery. 107 (4): 372–80. PMID 2321134.
- ↑ 11.0 11.1 Park WM, Gloviczki P, Cherry KJ, Hallett JW, Bower TC, Panneton JM, Schleck C, Ilstrup D, Harmsen WS, Noel AA (2002). "Contemporary management of acute mesenteric ischemia: Factors associated with survival". J. Vasc. Surg. 35 (3): 445–52. doi:10.1067/mva.2002.120373. PMID 11877691.
Template:WH Template:WS The above studies ultimately led to the aggressive roentgenologic and surgical approach to acute mesenteric ischemia we proposed in 1973.15 The cornerstones of that protocol are the earlier and more liberal use of angiography in patients at risk and the infusion of papaverine through the angiographic catheter as part of the treatment of both occlusive and nonocclusive mesenteric arterial insufficiency (Figs. 1 and 2). Utilizing the protocol based on these principles, we and Clark and Gallant20 were able to lower the mortality rate to 50% or less while preserving all or most of the intestines in the majority of patients. In a subsequent study of SMA embolus,9 we observed that in patients in whom there was a “doctor delay” of less than 12 hours before starting the protocol and when the management followed the protocol, there was a 67% survival—a realistic therapeutic goal.
Mesenteric Venous Thrombosis
Before the recognition of nonocclusive mesenteric ischemia, mesenteric venous thrombosis was thought to be the principal cause of acute mesenteric ischemia. Cokkinis, in 1926,21 reported that 60% of his patients with acute mesenteric infarction had venous thrombosis. By 1963, Jackson40 found that only 25% of his series had venous thrombosis, and in our institution the incidence had fallen to 10% by 1973 and to less than 5% by 1983.85
Although Klein46 and others had earlier described the varying clinical course that mesenteric venous thrombosis could take and differentiated the entities of arterial and venous occlusion, it was Donaldson and Stout25 and Warren and Eberhard84 who focused attention on mesenteric venous thrombosis as a distinct entity in 1935.
In the 1950s, 25% to 55% of cases of mesenteric venous thrombosis were thought to be of the “agnogenic” or “primary” type, developing de novo in the absence of any precipitating factors. Since then, the identification of the hypercoagulable states seen with oral contraceptives61 and antithrombin III,35 protein S, or protein C34 deficiencies has reduced the number of cases with no identifiable contributing disorders to less than 20%.
During the past 25 years, advances in imaging techniques have greatly increased our ability to diagnose mesenteric venous thrombosis before operation. Angiographic diagnosis in 1980,77 ultrasonographic identification in 1979,81 and diagnosis by CT in 198463 and by MRI in 19903 confirmed the early observations by Cokkinis21 that superior mesenteric venous thrombosis can follow a very benign course, and made possible added therapeutic options in the management of this condition.
By 1950, resection followed by immediate postoperative heparin administration57 had been established as the standard treatment. The first successful thrombectomy of the SMV was performed by Fontaine et al30 in 1953, but few of these operations have been reported since.38,53 Papaverine infusion into the SMA has more recently been employed in selected cases. An individualized approach to patients with mesenteric venous thrombosis incorporating newer diagnostic and therapeutic techniques was proposed by Boley et al in 1992.10 During the past 25 years, our ability to diagnose has paralleled advances in imaging techniques.
Chronic Mesenteric Ischemia
The earliest report of chronic intestinal ischemia was by Councilman in 1894.24 He reported three cases of chronic occlusion of the SMA associated with abdominal pain. In 1901, Schnitzler67 described a patient with long-standing postprandial pain who went on to develop an acute thrombosis and intestinal infarction. Despite these reports, the concept of chronic mesenteric ischemia remained controversial because at autopsy some patients had evidence of stenosis or occlusion of all three splanchnic arteries but had had no abdominal symptoms. In 1936, Dunphy26 reconfirmed the entity when he described premonitory symptoms of postprandial pain, weight loss, and altered intestinal motility in 7 of 12 patients dying of intestinal infarction. In 1957, Mikkelson55 coined the term “intestinal angina” to describe this symptom complex. Chronic mesenteric ischemia has always been a difficult diagnosis to prove. Recently, functional testing of the body's ability to increase intestinal blood flow after a test meal has helped refine our diagnostic accuracy.8
In 1958 Shaw and Maynard70 performed the first successful endarterectomy for chronic mesenteric ischemia. Technically more successful procedures, such as Dacron bypass grafting from the infrarenal aorta to the SMA, were described in 1962 by Morris et al,56 and antegrade aortovisceral bypass and transaortic visceral thromboendarterectomy were described in 1966 by Stoney and Wylie.75 More recently, balloon dilatation of stenoses of both the celiac artery and SMA has been successfully performed.31
References
| A01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C01 | C02 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| D01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| E01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| F01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| G01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| H01 | H02 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I01 | I02 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| J01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Overview
Surgery may be needed to treat mesenteric ischemia. Surgery for chronic mesenteric artery ischemia involves removing the blockage and reconnecting the arteries to the aorta. A bypass around the blockage is another procedure. It is usually done with a plastic tube graft. An alternative to surgery is a stent. It may be inserted to enlarge the blockage in the mesenteric artery or deliver medicine directly to the affected area. This is a new technique and it should only be done by experienced health care providers. The outcome is usually better with surgery. Surgical revascularisation remains the treatment of choice for mesenteric ischemia, but thrombolytic medical treatment and vascular interventional radiological techniques have a growing role [1].
Management of Patients With Peripheral Artery Disease (Compilation of 2005 and 2011 ACCF/AHA Guideline Recommendations) : A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines[2]
Nonocclusive Intestinal Ischemia Treatment (DO NOT EDIT)[3]
| Class I |
| "1. Treatment of the underlying shock state is the most important initial step in treatment of nonocclusive intestinal ischemia. (Level of Evidence: C)" |
| "2. Laparotomy and resection of nonviable bowel is indicated in patients with nonocclusive intestinal ischemia who have persistent symptoms despite treatment. (Level of Evidence: B) " |
| Class IIa |
| "1. Transcatheter administration of vasodilator medications into the area of vasospasm is indicated in patients with nonocclusive intestinal ischemia who do not respond to systemic supportive treatment and in patients with intestinal ischemia due to cocaine or ergot poisoning. (Level of Evidence: B)" |
Acute Obstructive Intestinal Ischemia (DO NOT EDIT)[3]
| Class I |
| "1. Surgical treatment of acute obstructive intestinal ischemia includes revascularization, resection of necrotic bowel, and, when appropriate, a “second look” operation 24 to 48 hours after the revascularization. (Level of Evidence: B)" |
Endovascular Treatment (DO NOT EDIT)[3]
| Class IIb |
| "1. Percutaneous interventions (including transcatheter lytic therapy, balloon angioplasty, and stenting) are appropriate in selected patients with acute intestinal ischemia caused by arterial obstructions. Patients so treated may still require laparotomy. (Level of Evidence: C)" |
Chronic Intestinal Ischemia Surgical Treatment (DO NOT EDIT)[3]
| Class I |
| "1. Surgical treatment of chronic intestinal ischemia is indicated in patients with chronic intestinal ischemia. (Level of Evidence: B)" |
| Class IIb |
| "1. Revascularization of asymptomatic intestinal arterial obstructions may be considered for patients undergoing aortic/renal artery surgery for other indications. (Level of Evidence: B)" |
| Class III (No Benefit) |
| "1. Surgical revascularization is not indicated for patients with asymptomatic intestinal arterial obstructions, except in patients undergoing aortic/renal artery surgery for other indications. (Level of Evidence: B)" |
Chronic Intestinal Ischemia Interventional Treatment (DO NOT EDIT)[3]
| Class I |
| "1. Percutaneous endovascular treatment of intestinal arterial stenosis is indicated in patients with chronic intestinal ischemia. (Level of Evidence: B) " |
References
- ↑ Sreenarasimhaiah J (2003). "Diagnosis and management of intestinal ischaemic disorders". BMJ. 326 (7403): 1372–6. doi:10.1136/bmj.326.7403.1372. PMID 12816826.
- ↑ Rooke TW, Hirsch AT, Misra S, Sidawy AN, Beckman JA, Findeiss L; et al. (2013). "Management of patients with peripheral artery disease (compilation of 2005 and 2011 ACCF/AHA Guideline Recommendations): a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines". J Am Coll Cardiol. 61 (14): 1555–70. doi:10.1016/j.jacc.2013.01.004. PMC 4492473. PMID 23473760.
- ↑ 3.0 3.1 3.2 3.3 3.4 Hirsch AT, Haskal ZJ, Hertzer NR, Bakal CW, Creager MA, Halperin JL, Hiratzka LF, Murphy WR, Olin JW, Puschett JB, Rosenfield KA, Sacks D, Stanley JC, Taylor LM, White CJ, White J, White RA, Antman EM, Smith SC, Adams CD, Anderson JL, Faxon DP, Fuster V, Gibbons RJ, Hunt SA, Jacobs AK, Nishimura R, Ornato JP, Page RL, Riegel B (2006). "ACC/AHA 2005 Practice Guidelines for the management of patients with peripheral arterial disease (lower extremity, renal, mesenteric, and abdominal aortic): a collaborative report from the American Association for Vascular Surgery/Society for Vascular Surgery, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, Society of Interventional Radiology, and the ACC/AHA Task Force on Practice Guidelines (Writing Committee to Develop Guidelines for the Management of Patients With Peripheral Arterial Disease): endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation; National Heart, Lung, and Blood Institute; Society for Vascular Nursing; TransAtlantic Inter-Society Consensus; and Vascular Disease Foundation". Circulation. 113 (11): e463–654. doi:10.1161/CIRCULATIONAHA.106.174526. PMID 16549646. Retrieved 2012-10-09. Unknown parameter
|month=ignored (help)
Surgery Latest Feham is annoyed
- The goals of surgical therapy are as follows:
- Re-establishment blood supply to the ischemic bowel
- Resection of all non-viable areas of the bowel
- Preservation of the viable bowel
- Intestinal viability is defined as the maximum vital element influencing outcome in patients with AMI.
- Non-viable bowel, if unrecognized, can cause multi-organ failure and lead to the death eventually.
- Laparotomy allows to determine the viability of the bowel.
- After preliminary resuscitation, midline laparotomy should be done observed by means of assessment of all areas of the gut with choices for resection of all surely necrotic areas. In instances of uncertainty, intraoperative Doppler can be beneficial.
Medical therapy:
The important factors in the treatment of acute mesenteric ischemia are summarized in Fig. . The for open surgical an endovascular of key . First and is the and severity of bowel ischemic, of the occlusive lesion, and is of the availability and the of an
of of acute mesenteric ischemia are summarized in Fig. . The for open surgical an endovascular of key . First and is the and severity of bowel ischemic, of the occlusive lesion, and is of the availability and the of an
Flow chart
| A01 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B01 | B02 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C01 | C02 | C03 | C04 | C05 | C06 | C07 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C01 | C02 | C03 | C04 | C05 | C06 | C07 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| B01 | B02 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The following table summarizes the mechanism, genetic association, gross pathology features and microscopic findings of common causes of dysphagia.
| Cause of dysphagia | Type of food | Type of progression | Pathophysiological changes | Genetic association | Gross pathology features | Microscopic findings | |
|---|---|---|---|---|---|---|---|
| Oropharyngeal dysphagia | Soilds | Liquids | Intermittent/Progressive | ||||
| •Zenker's diverticulum
•Webs |
Yes | No | Progressive | •Zenker's diverticulum(ZD):
|
Diverticulum or a sac is seen in the esophagus |
| |
| •Webs | |||||||
| •Neoplasm | Yes | Yes | Progressive | •Neoplasm | |||
| Myogenic causes
•Myasthenia gravis •Connective tissue disorder •Myotonic dystrophy Neurogenic causes •ALS •Parkinsonism •Stroke |
Yes | Yes | Progressive | ||||
| Esophageal dysphagia | |||||||
| •Pill esophagitis
•Caustic injury •Chemotherapy |
Yes | No | |||||
| •Strictures
•Esophageal Cancer |
Yes | No | Progressive | Esophageal stricture is the result of:[1][2]
|
The following genes can be involved:
|
On gross pathology, circumferential thickening of the lower esophageal wall are characteristic finding of esophageal stricture due to gastroesophageal reflux disease.[3]
|
On microscopic histopathological analysis, characteristic findings of esophageal stricture due to gastroesophageal reflux disease are:[7]
|
| •Esophageal Cancer: |
Mutations in the following genes can cause esophageal cancer: |
Squamous cell carcinoma or adenocarcinoma of the esophagus may appear as:[9]
|
Nuclear atypia of malignancy:
| ||||
| •Rings
•Webs |
Yes | No | Intermittent | Rings:
Webs: Multiple theories have been found:
|
|||
| •Achalasia
•Diffuse esophageal spasm(DES) |
Yes | Yes | Intermittent | •Achalasia:
|
|
| |
•Diffuse esophageal spasm(DES):
|
There is a genetic association between DES and achalasia[14] | Gross thickening of muscularis propria layer and lower esophageal sphincter (LES) due to hyperplasia are characteristic findings of DES | There is degeneration of vagal fibres, inflammatory infiltration of myenteric plexus, and hyperplasia of smooth muscles are characteristic findings of DES | ||||
| •Scleroderma | Yes | Yes | Progressive | ||||
Gallery
{{#ev:youtube|X4ryV6wGK1Y}}
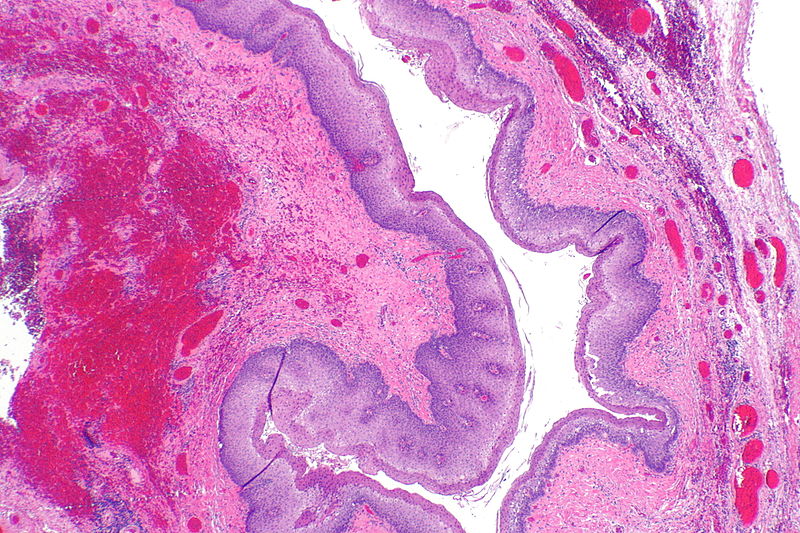
Zenkers diverticulum
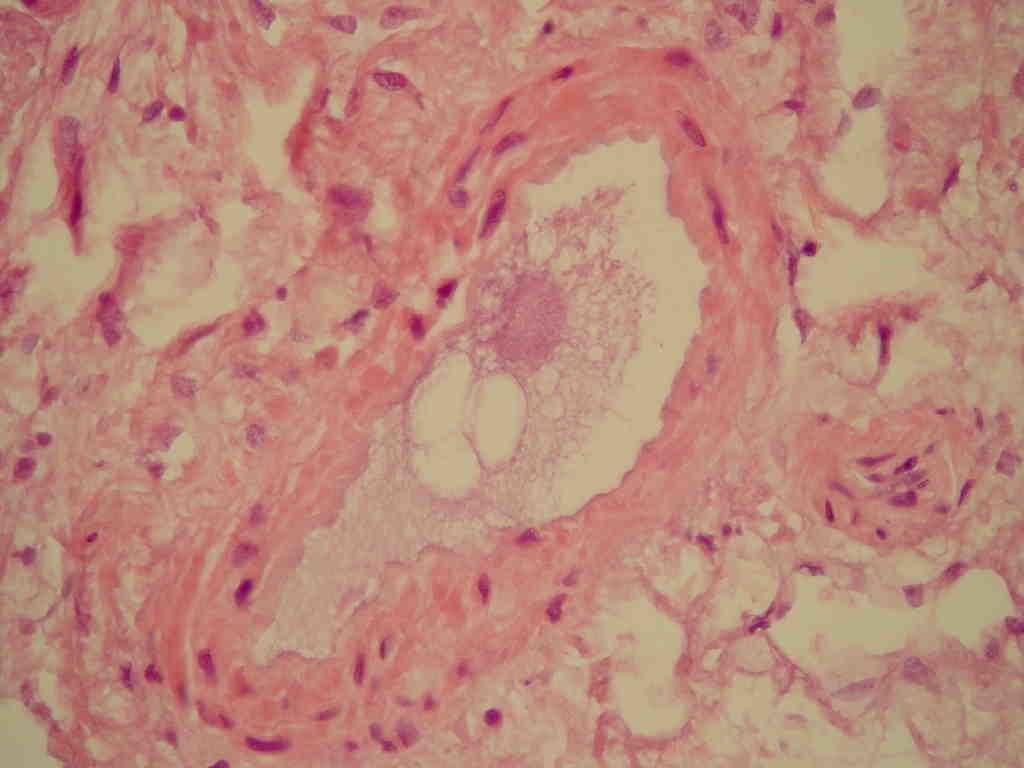
Fat embolism
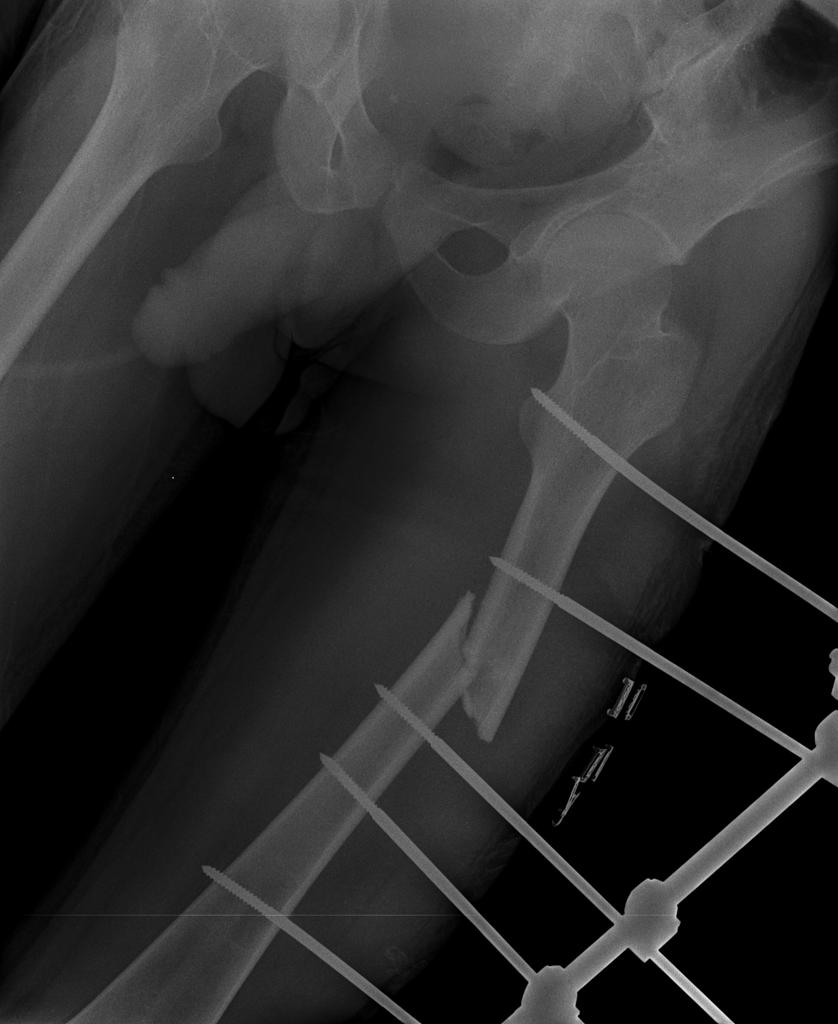
Esophageal cancer
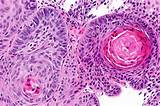 |
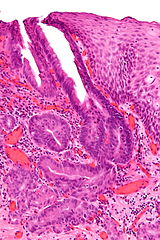 |
- ↑ Holzheimer, R (2001). Surgical treatment : evidence-based and problem-oriented. München New York: Zuckschwerdt. ISBN 3-88603-714-2.
- ↑ Belevich VL, Ovchinnikov DV (2013). "[Treatment of benign esophageal stricture]". Vestn. Khir. Im. I. I. Grek. (in Russian). 172 (5): 111–4. PMID 24640761.
- ↑ Yamasaki, Yasushi; Ozawa, Soji; Oguma, Junya; Kazuno, Akihito; Ninomiya, Yamato (2016). "Long peptic strictures of the esophagus due to reflux esophagitis: a case report". Surgical Case Reports. 2 (1). doi:10.1186/s40792-016-0190-1. ISSN 2198-7793.
- ↑ Maejima, Ryuhei; Uno, Kaname; Iijima, Katsunori; Fujishima, Fumiyoshi; Noguchi, Tetsuya; Ara, Nobuyuki; Asano, Naoki; Koike, Tomoyuki; Imatani, Akira; Shimosegawa, Tooru (2016). "A Japanese case of lymphocytic esophagitis". Digestive Endoscopy. 28 (4): 476–480. doi:10.1111/den.12578. ISSN 0915-5635.
- ↑ Contini, Sandro (2013). "Caustic injury of the upper gastrointestinal tract: A comprehensive review". World Journal of Gastroenterology. 19 (25): 3918. doi:10.3748/wjg.v19.i25.3918. ISSN 1007-9327.
- ↑ Wilcox CM (2013). "Overview of infectious esophagitis". Gastroenterol Hepatol (N Y). 9 (8): 517–9. PMC 3980995. PMID 24719600.
- ↑ "Esophageal stricture - Libre Pathology".
- ↑
- ↑ Sugarbaker, David (2015). Adult chest surgery. New York: McGraw-Hill Education. ISBN 0071781897.
- ↑ "Squamous cell carcinoma of the esophagus".
- ↑ Paladini F, Cocco E, Cascino I, Belfiore F, Badiali D, Piretta L; et al. (2009). "Age-dependent association of idiopathic achalasia with vasoactive intestinal peptide receptor 1 gene". Neurogastroenterol Motil. 21 (6): 597–602. doi:10.1111/j.1365-2982.2009.01284.x. PMID 19309439.
- ↑ Alahdab YO, Eren F, Giral A, Gunduz F, Kedrah AE, Atug O; et al. (2012). "Preliminary evidence of an association between the functional c-kit rs6554199 polymorphism and achalasia in a Turkish population". Neurogastroenterol Motil. 24 (1): 27–30. doi:10.1111/j.1365-2982.2011.01793.x. PMID 21951831.
- ↑ de León AR, de la Serna JP, Santiago JL, Sevilla C, Fernández-Arquero M, de la Concha EG; et al. (2010). "Association between idiopathic achalasia and IL23R gene". Neurogastroenterol Motil. 22 (7): 734–8, e218. doi:10.1111/j.1365-2982.2010.01497.x. PMID 20367798.
- ↑ Frieling T, Berges W, Borchard F, Lübke HJ, Enck P, Wienbeck M (1988). "Family occurrence of achalasia and diffuse spasm of the oesophagus". Gut. 29 (11): 1595–602. PMC 1433819. PMID 3061886.