Ebola pathophysiology
|
Ebola Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Postmortem Care |
|
Case Studies |
|
Ebola pathophysiology On the Web |
|
American Roentgen Ray Society Images of Ebola pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Michael Maddaleni, B.S.; Guillermo Rodriguez Nava, M.D. [2]
Overview
The Ebola virus affects the mononuclear phagocyte system, but also hepatocytes, spongiocytes, fibroblasts and endothelial cells, and makes severe changes in the hematological, and coagulation systems. Since the Ebola Hemorrhagic Fever is endemic among inaccessible areas, it is difficult to study the pathophysiology of the disease using a human model, almost all the information of the pathogenesis of the disease has been obtained from animal models, such as rodents and nonhuman primates[1][2][3][4] .
Pathophysiology
Transmission
Among humans, the virus is transmitted by direct contact with infected body fluids, or to a lesser extent, skin or mucous membrane contact. The incubation period can be anywhere from 2 to 21 days, but is generally between 5 and 10 days.
Although airborne transmission between monkeys has been demonstrated by an accidental outbreak in a laboratory located in Virginia, USA, there is very limited evidence for human-to-human airborne transmission in any reported epidemics. Nurse Mayinga might represent the only possible case. The means by which she contracted the virus remains uncertain.
The infection of human cases with Ebola virus has been documented through the handling of infected chimpanzees, gorillas, and forest antelopes--both dead and alive--as was documented in Côte d'Ivoire, the Republic of Congo and Gabon. The transmission of the Ebola Reston strain through the handling of cynomolgus monkeys has also been reported.
So far, all epidemics of Ebola have occurred in sub-optimal hospital conditions, where practices of basic hygiene and sanitation are often either luxuries or unknown to caretakers and where disposable needles and autoclaves are unavailable or too expensive. In modern hospitals with disposable needles and knowledge of basic hygiene and barrier nursing techniques, Ebola has never spread on such a large scale.
In the early stages, Ebola may not be highly contagious. Contact with someone in early stages may not even transmit the disease. As the illness progresses, bodily fluids from diarrhea, vomiting, and bleeding represent an extreme biohazard. Due to lack of proper equipment and hygienic practices, large scale epidemics occur mostly in poor, isolated areas without modern hospitals or well-educated medical staff. Many areas where the infectious reservoir exists have just these characteristics. In such environments, all that can be done is to immediately cease all needle-sharing or use without adequate sterilization procedures, to isolate patients, and to observe strict barrier nursing procedures with the use of a medical rated disposable face mask, gloves, goggles, and a gown at all times. This should be strictly enforced for all medical personnel and visitors.
Ebola is unlikely to develop(sometimes) into a pandemic, or world-wide infection, due to its difficulty in spreading by airborne transmission and the period of time that the virus can use a living and contagious victim to spread compared to other infectious diseases. In isolated settings such as a quarantined hospital or a remote village, most victims are infected shortly after the first case of infection is present. In addition, the quick onset of symptoms from the time the disease becomes contagious in an individual makes it easy to identify sick individuals and limits an individual's ability to spread the disease by traveling. Because bodies of the deceased are still infectious, many doctors implemented measures to properly dispose of dead bodies in spite of some traditional local burial rituals.[5]
Life Cycle
- Virus attaches to host receptors through the GP (glycoprotein) surface peplomer and is endocytosed into vesicles in the host cell.
- Fusion of virus membrane with the vesicle membrane occurs; nucleocapsid is released into the cytoplasm.
- The encapsidated, negative-sense genomic ssRNA is used as a template for the synthesis ( 3' - 5') of polyadenylated, monocistronic mRNAs.
- Translation of the mRNA into viral proteins occurs using the host cell's machinery.
- Post-translational processing of viral proteins occurs. GP0 (glycoprotein precursor) is cleaved to GP1 and GP2, which are heavily glycosylated. These two molecules assemble, first into heterodimers, and then into trimers to give the surface peplomers. SGP (secreted glycoprotein) precursor is cleaved to SGP and delta peptide, both of which are released from the cell.
- As viral protein levels rise, a switch occurs from translation to replication. Using the negative-sense genomic RNA as a template, a complementary +ssRNA is synthesized; this is then used as a template for the synthesis of new genomic (-)ssRNA, which is rapidly encapsidated.
- The newly-formed nucleocapsides and envelope proteins associate at the host cell's plasma membrane; budding occurs, and the virions are released
Ebola hemorrhagic fever (Ebola fever) is caused by a virus belonging to the family called Filoviridae. Scientists have identified four types of the Ebola virus. Three have been reported to cause disease in humans: Ebola-Zaire virus, Ebola-Sudan virus, and Ebola-Ivory Coast virus. The human disease has so far been limited to parts of Africa.
A very small number of people in the United States who were infected with the fourth type of the virus, known as Ebola Reston, did not develop any signs of disease.
The disease can be passed to humans from infected animals and animal materials. Ebola can also be spread between humans by close contact with infected bodily fluids or through infected needles in the hospital.
Tropism
- Ebola virus infects mainly the cells of the mononuclear phagocyte system, but also fibroblasts, hepatocytes, spongiocytes, adrenal cortical cells and endothelial cells.[1]
- The infection of the mononuclear phagocyte system cells plays a key role in the pathogenesis and spread of the disease as they carry the virus from of the initial infection site, through the lymphatic system and blood, to the regional lymph nodes,spleen and liver.[6]
- The next table summarizes the pathogenesis of the disease according to the virus tropism.
| Organ/Tissue | Effect |
|---|---|
| Endothelial cells | Glycoprotein (GP) on the virion envelope allows introduction of its content into the endothelial cells, which induces a cytopathic effect and damage to the endothelial barrier function that, together with effects of TNF-α released by infected mononuclear cells, leads to the loss of vascular integrity and increased leakage. |
| Liver | Causes hepatocellular necrosis which could impair the synthesis of proteins of the coagulation system[6] |
| Adrenal cortex | Affects the synthesis of enzymes responsible for the synthesis of steroids, leading to hypotension, and fluid and electrolytes disturbances.[6] |
| Lymphatic system | [[Necrosis of the spleen, lymph nodes and thymus; Apoptosis of lymphocytes leading to lymphopenia.[6][7] |
Immune response
- The virus activates the macrophages synthesis of interleukins (IL), which leads the Th1/Th2 balance towards a more pronounced Th1-cell mediated response.[8]
- Some inflammatory mediators produced during the ebola virus ]]infection]] include: interferon (IFN)-alpha, IFN-beta, IL-2, IL-6, IL-8, IL-10, interferon-inducible protein 10; monocyte chemoattractant protein 1; regulated upon activation normal T cell expressed and secreted (RANTES); TNF-alpha; and reactive oxygen and nitrogen species.[9][10][11]
- Some viral proteins, such as VP35 and VP24, block the type I interferon response, which plays a key role of the pathogenesis of the disease.[12]
- The reactive oxygen and nitrogen species contribute to the cell and tissue damage, and therefore vascular and organ damage.[13]
- The nitric oxide is known to be an important vasodilator, therefore it plays and important role in the development of hypotension and shock.
Coagulation system
- Alterations of the coagulation system induced by the ebola virus, which are thought to be mediated by the production of tissue factor:[14]
| Alterations of the coagulation system |
|---|
| Consumption of clotting factors |
| Increased concentrations of fibrin degradation products |
| Disseminated intravascular coagulation |
Viral Reservoirs
Despite numerous studies, the wildlife reservoir of Ebolavirus has not been identified. Between 1976 and 1998, from 30,000 mammals, birds, reptiles, amphibians, and arthropods sampled from outbreak regions, no Ebolavirus was detected [15] apart from some genetic material found in six rodents (Mus setulosusand Praomys species) and a shrew (Sylvisorex ollula) collected from the Central African Republic in 1998.[16] Ebolavirus was detected in the carcasses of gorillas, chimpanzees and duikers during outbreaks in 2001 and 2003 (the carcasses were the source of the initial human infections) but the high mortality from infection in these species precludes them from acting as reservoirs.[15] Plants, arthropods, and birds have also been considered as reservoirs, however bats are considered the most likely candidate[17]. Bats were known to reside in the cotton factory in which the index cases for the 1976 and 1979 outbreaks were employed and have also been implicated in Marburg infections in 1975 and 1980.[15] Of 24 plant species and 19 vertebrate species experimentally inoculated with Ebolavirus, only bats became infected.[18] The absence of clinical signs in these bats is characteristic of a reservoir species. In 2002-03, a survey of 1,030 animals from Gabon and the Republic of the Congo including 679 bats found Ebolavirus RNA in 13fruit bats (Hyspignathus monstrosus, Epomops franquetti and Myonycteris torquata).[19] Bats are also known to be the reservoirs for a number of related viruses including Nipah virus, Hendra virus andlyssaviruses.
Bats
Even though rabies and histoplasmosis can be found all over the world, some diseases associated with bats are found exclusively in certain regions of the world. Notably, research suggests that bats might be the source of several hemorrhagic fevers, which affect multiple organ systems in the body and often lead to life-threatening diseases.
One of these diseases is Marburg hemorrhagic fever, which is found exclusively in Africa. Past outbreaks have shown that Marburg Hemorrhagic Fever kills up to 90% of those infected.
While the natural host had for years been unknown, new research suggests that fruit bats are a natural source of this virus, and the virus has been isolated repetitively from fruit bats in Uganda.
The same may be true for Ebola hemorraghic fever. The virus that causes this disease is often referred to as the "cousin" of Marburg virus, since they are the only distinct viruses that belong to a group of viruses known as filoviruses. Like Marburg, Ebola is highly fatal and is found mostly in Africa. Recent studies indicate that, as with Marburg, bats are likely to be a natural source of this virus, although no Ebola virus has been isolated from bats.
Microscopic Pathology
These are that display some of the key features of the ebola virus.
-
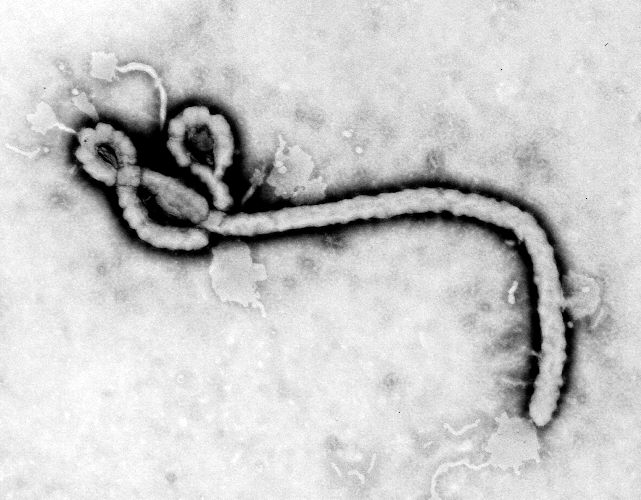
Created by CDC microbiologist Frederick A. Murphy, this transmission electron micrograph (TEM) revealed some of the ultrastructural morphology displayed by an Ebola virus virion.
-
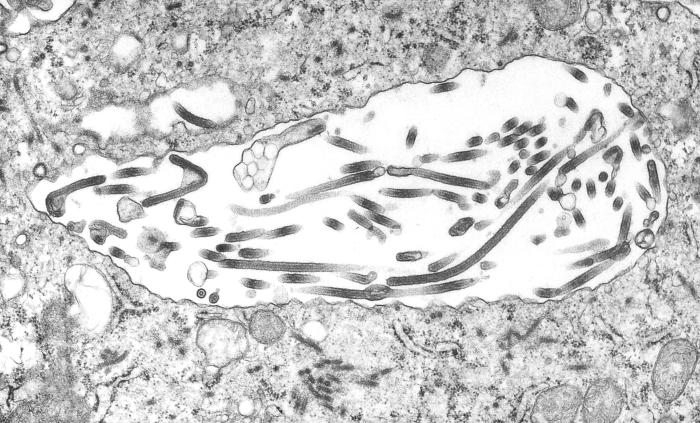
This 1995 transmission electron micrograph (TEM) revealed some of the ultrastructural morphologic changes in this tissue sample isolate brought on due to an Ebola hemorrhagic fever infection, including the presence of numbers of Ebola virions.
-
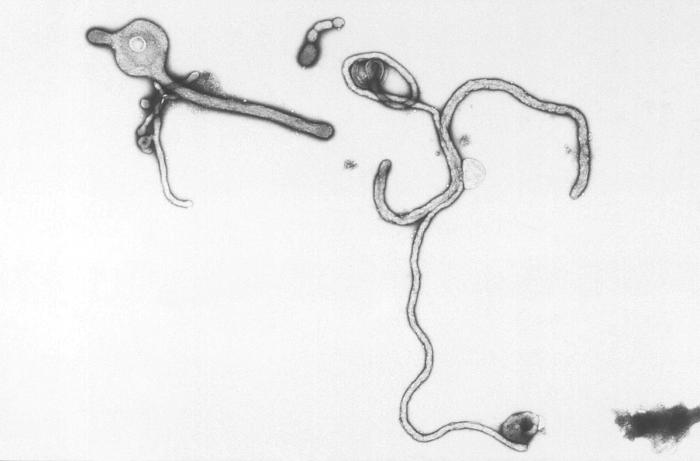
Created by CDC microbiologist Charles Humphrey, this scanning electron micrograph (SEM) revealed some of the ultrastructural morphologic features displayed by the Ebola virus discovered from the Ivory Coast of Africa.
-
Created by CDC microbiologist Charles Humphrey, this negatively-stained transmission electron micrograph (TEM) revealed some of the ultrastructural curvilinear morphologic features displayed by the Ebola virus discovered from the Ivory Coast of Africa.
References
- ↑ 1.0 1.1 Ryabchikova E, Kolesnikova L, Smolina M, Tkachev V, Pereboeva L, Baranova S; et al. (1996). "Ebola virus infection in guinea pigs: presumable role of granulomatous inflammation in pathogenesis". Arch Virol. 141 (5): 909–21. PMID 8678836.
- ↑ Bray M, Davis K, Geisbert T, Schmaljohn C, Huggins J (1998). "A mouse model for evaluation of prophylaxis and therapy of Ebola hemorrhagic fever". J Infect Dis. 178 (3): 651–61. PMID 9728532.
- ↑ Connolly BM, Steele KE, Davis KJ, Geisbert TW, Kell WM, Jaax NK; et al. (1999). "Pathogenesis of experimental Ebola virus infection in guinea pigs". J Infect Dis. 179 Suppl 1: S203–17. doi:10.1086/514305. PMID 9988186.
- ↑ Bray M, Hatfill S, Hensley L, Huggins JW (2001). "Haematological, biochemical and coagulation changes in mice, guinea-pigs and monkeys infected with a mouse-adapted variant of Ebola Zaire virus". J Comp Pathol. 125 (4): 243–53. doi:10.1053/jcpa.2001.0503. PMID 11798241.
- ↑ Harden, Blaine (2001-02-18). "Dr. Matthew's Passion". New York Times Magazine. Retrieved 2008-02-25.
- ↑ 6.0 6.1 6.2 6.3 Geisbert TW, Hensley LE, Larsen T, Young HA, Reed DS, Geisbert JB; et al. (2003). "Pathogenesis of Ebola hemorrhagic fever in cynomolgus macaques: evidence that dendritic cells are early and sustained targets of infection". Am J Pathol. 163 (6): 2347–70. doi:10.1016/S0002-9440(10)63591-2. PMC 1892369. PMID 14633608.
- ↑ Zaki SR, Goldsmith CS (1999). "Pathologic features of filovirus infections in humans". Curr Top Microbiol Immunol. 235: 97–116. PMID 9893381.
- ↑ Qiu X, Audet J, Wong G, Fernando L, Bello A, Pillet S; et al. (2013). "Sustained protection against Ebola virus infection following treatment of infected nonhuman primates with ZMAb". Sci Rep. 3: 3365. doi:10.1038/srep03365. PMC 3842534. PMID 24284388.
- ↑ Villinger F, Rollin PE, Brar SS, Chikkala NF, Winter J, Sundstrom JB; et al. (1999). "Markedly elevated levels of interferon (IFN)-gamma, IFN-alpha, interleukin (IL)-2, IL-10, and tumor necrosis factor-alpha associated with fatal Ebola virus infection". J Infect Dis. 179 Suppl 1: S188–91. doi:10.1086/514283. PMID 9988183.
- ↑ Hensley LE, Young HA, Jahrling PB, Geisbert TW (2002). "Proinflammatory response during Ebola virus infection of primate models: possible involvement of the tumor necrosis factor receptor superfamily". Immunol Lett. 80 (3): 169–79. PMID 11803049.
- ↑ Baize S, Leroy EM, Georges AJ, Georges-Courbot MC, Capron M, Bedjabaga I; et al. (2002). "Inflammatory responses in Ebola virus-infected patients". Clin Exp Immunol. 128 (1): 163–8. PMC 1906357. PMID 11982604.
- ↑ Basler CF, Mikulasova A, Martinez-Sobrido L, Paragas J, Mühlberger E, Bray M; et al. (2003). "The Ebola virus VP35 protein inhibits activation of interferon regulatory factor 3". J Virol. 77 (14): 7945–56. PMC 161945. PMID 12829834.
- ↑ Sanchez A, Lukwiya M, Bausch D, Mahanty S, Sanchez AJ, Wagoner KD; et al. (2004). "Analysis of human peripheral blood samples from fatal and nonfatal cases of Ebola (Sudan) hemorrhagic fever: cellular responses, virus load, and nitric oxide levels". J Virol. 78 (19): 10370–7. doi:10.1128/JVI.78.19.10370-10377.2004. PMC 516433. PMID 15367603.
- ↑ Geisbert TW, Young HA, Jahrling PB, Davis KJ, Kagan E, Hensley LE (2003). "Mechanisms underlying coagulation abnormalities in ebola hemorrhagic fever: overexpression of tissue factor in primate monocytes/macrophages is a key event". J Infect Dis. 188 (11): 1618–29. doi:10.1086/379724. PMID 14639531.
- ↑ 15.0 15.1 15.2 Pourrut, Xavier (2005). "The natural history of Ebola virus in Africa". Microbes and Infection. 7 (7–8): 1005–1014. doi:10.1016/j.micinf.2005.04.006. Unknown parameter
|coauthors=ignored (help) - ↑ Morvan, Jaques (1999). "Identification of Ebola virus sequences present as RNA or DNA in organs of terrestrial small mammals of the Central African Republic". Microbes and Infection. 1 (14): 1193–1201. doi:10.1016/S1286-4579(99)00242-7. Unknown parameter
|coauthors=ignored (help) - ↑ "Fruit bats may carry Ebola virus". BBC News. 2005-12-11. Retrieved 2008-02-25.
- ↑ Swanepoel, R (1996). "Experimental inoculation of plants and animals with Ebola virus". Emerging Infectious Diseases. 2: 321–325. Unknown parameter
|coauthors=ignored (help) - ↑ Leroy, Eric (2005). "Fruit bats as reservoirs of Ebola virus". Nature. 438: 575–576. doi:10.1038/438575a. Unknown parameter
|coauthors=ignored (help)