Ebola virus
| style="background:#Template:Taxobox colour;"|Ebola virus | ||||||||
|---|---|---|---|---|---|---|---|---|
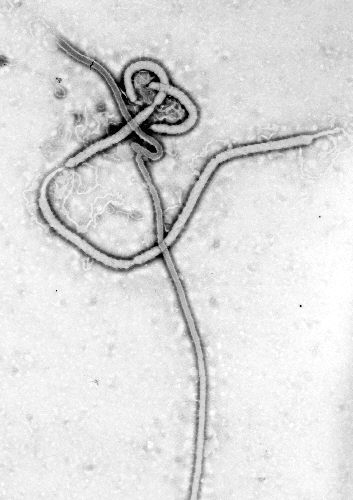 | ||||||||
| style="background:#Template:Taxobox colour;" | Virus classification | ||||||||
| ||||||||
| Type species | ||||||||
| Zaïre Ebolavirus | ||||||||
| Species | ||||||||
|
Reston Ebolavirus |
|
Ebola Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Postmortem Care |
|
Case Studies |
|
Ebola virus On the Web |
|
American Roentgen Ray Society Images of Ebola virus |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Ebola infection is caused by the Ebola virus that belongs to the family Filoviridae. Four viral subtypes have been reported to cause clinical illness in humans: Bundibugyo ebolavirus, Sudan ebolavirus, Tai Forest ebolavirus, and Zaire ebolavirus.
Taxonomy
Viruses; ssRNA viruses; ssRNA negative-strand viruses; Mononegavirales; Filoviridae; Ebolavirus[1]
- Ebolavirus
- Bundibugyo ebolavirus
- Reston ebolavirus
- Reston ebolavirus - Reston
- Reston ebolavirus - Reston (1989)
- Reston ebolavirus - Siena/Philippine-92
- Sudan ebolavirus
- Sudan ebolavirus - Boniface (1976)
- Sudan ebolavirus - Maleo (1979)
- Sudan ebolavirus - Nakisamata
- Sudan ebolavirus - Uganda (2000)
- Tai Forest ebolavirus
- Tai Forest virus - Côte d’Ivoire, Côte d’Ivoire, 1994
- Zaire ebolavirus
- Ebola virus - Mayinga, Zaire, 1976
- Zaire ebolavirus - Eckron (Zaire, 1976)
- Zaire ebolavirus - Gabon (1994-1997)
- Zaire ebolavirus - Zaire (1995)
- Unclassified Ebolavirus
- Ebola virus Yambio0401
- Ebola virus Yambio0402
- Ebola virus Yambio0403
- Ebola virus sp.
Virology
Structure
Electron micrographs of Ebola virus demonstrates the characteristic thread-like structure of a filovirus.[2] EBOV VP30 is around 288 amino acids long.[2] The virions are tubular and variable in shape and may resemble a "U", "6", coiled, circular, or branched shape. It is important to note that laboratory purification techniques, such as centrifugation, may contribute to the various shapes.[2] Virions are generally 80 nm in diameter.[2] They are variable in length, and can be up to 1400 nm long. On average, however, the length of a typical Ebola virus is closer to 1000 nm. In the center of the virion is a structure called nucleocapsid, which is formed of the helically wound viral genomic RNA complexed to the proteins NP, VP35, VP30 and L. The virus has an average diameter of 40-50 nm and contains a central channel of 20–30 nm in diameter. Virally encoded glycoprotein (GP) spikes 10 nm long and 10 nm apart are present on the outer viral envelope of the virion, which is derived from the host cell membrane. Between the envelope and the nucleocapsid exists a matrix space that contains the viral proteins VP40 and VP24.
Genome
Each virion contains one minor molecule of linear, single-stranded, negative-sense RNA, totaling 18959 to 18961 nucleotides in length. The 3′ terminus is not polyadenylated and the 5′ end is not capped. Only 472 nucleotides from the 3' end and 731 nucleotides from the 5' end are sufficient for replication.[2] The genome codes for seven structural proteins and one non-structural protein. The gene order is as follows: 3′ - leader - NP - VP35 - VP40 - GP/sGP - VP30 - VP24 - L - trailer - 5′. Leader and trailer regions are non-transcribed regions that carry important signals to control transcription, replication, and packaging of the viral genomes into new virions. The genomic material by itself is not infectious, because viral proteins such as RNA-dependent RNA polymerase are necessary for viral transcription and replication.
Life Cycle
- The virus attaches to host receptors through the GP (glycoprotein) surface peplomer and is endocytosed into vesicles in the host cell.
- Fusion of virus membrane with the vesicle membrane occurs; nucleocapsid is released into the cytoplasm.
- The encapsidated, negative-sense genomic ssRNA is used as a template for the synthesis (3'-5') of polyadenylated, monocistronic mRNAs using the viral RNA-dependent RNA polymerase.
- Translation of the mRNA into viral proteins occurs using the host cell's machinery.
- Post-translational processing of viral proteins occurs. GP0 (glycoprotein precursor) is cleaved to GP1 and GP2, which are heavily glycosylated. These two molecules assemble, first into heterodimers, and then into trimers to give the surface peplomers. SGP (secreted glycoprotein) precursor is cleaved to SGP and delta peptide, both of which are released from the cell.
- As viral protein levels rise, a switch occurs from translation to replication. Using the negative-sense genomic RNA as a template, a complementary positive-sense ssRNA is synthesized; this is then used as a template for the synthesis of new genomic negative-sense ssRNA, which is rapidly encapsidated.
- The newly-formed nucleocapsides and envelope proteins associate at the host cell's plasma membrane; budding occurs, and the virions are released.
Transmission
- Among humans, the virus is transmitted by direct contact with infected body fluids, or to a lesser extent, skin or mucous membrane contact. The incubation period can be anywhere from 2 to 21 days, but is generally between 5 and 10 days.
- Human-to-human airborne transmission has not been reported in any reported epidemics.
- The infection of human cases with Ebola virus has been documented through the handling of infected chimpanzees, gorillas, and forest antelopes - both dead and alive - as was documented in Côte d'Ivoire, the Republic of Congo and Gabon.
- So far, all epidemics of Ebola have occurred in sub-optimal hospital conditions, where practices of basic hygiene and sanitation are often either luxuries or unknown to caretakers and where disposable needles and autoclaves are unavailable or too expensive. In modern hospitals with disposable needles and knowledge of basic hygiene and barrier nursing techniques, Ebola has never spread on such a large scale.
- In the early stages, particularly when the patient is asymptomatic, Ebola is not generally contagious. Contact with someone in early stages very rarely transmits the disease. As the illness progresses, bodily fluids from diarrhea, vomiting, and bleeding represent an extreme biohazard.
- Other than samples that are grossly contaminated with body fluids, Ebola virus is rarely detected on environmental surfaces and thus transmission by fomites is not very common. However, given that the infectious dose of the virus is low, and that current evaluation techniques such as cell culture and RT-PCR have not been well validated for environmental detection (unknown sensitivity and specificity), the true risk of transmission by contaminated surfaces is unknown.[3]
Transmission in Recovering Patients and Continued Shedding
- Ebola virus has been isolated in the semen of recovering patients 40 days after the onset of illness. This stresses the risk of sexual transmission during the recovery phase. The Zaire Ebola virus in particular has a higher risk of continued shedding in the semen. It has been detected in samples for up to 90 days after the onset of illness.
- Continued shedding may be detected in many bodily fluids with varying durations. The only bodily fluid that does not contain Ebola virus, even during the symptomatic phase of the infection, is urine. This is probably related to the inability of the kidney to filter the Ebola virus particles.
- It is generally recommended to abstain from sex or to use condoms for up to 3 months after the onset of illness. It is also recommended to avoid breastfeeding and contact with the mucous membranes of the eye for the same amount of time.[3]
Viral Reservoirs
Despite numerous studies, the wildlife reservoir of Ebolavirus has not yet been identified. Between 1976 and 1998, 30,000 mammals, birds, reptiles, amphibians, and arthropods were sampled from outbreak regions with no virus detected.[4] Ebolavirus has been detected in the carcasses of gorillas, chimpanzees, and duikers during outbreaks in 2001 and 2003 and these carcasses were deemed to be the source of initial human infection. However given the high mortality in these species, they are unlikely to be able to act as reservoirs.[4] Plants, arthropods, and birds have also been considered as reservoirs, however bats are considered the most likely candidate[5].
Bats
Bats were known to reside in the cotton factory in which the index cases for the 1976 and 1979 outbreaks were employed.[4] Of 24 plant species and 19 vertebrate species experimentally inoculated with Ebolavirus, only bats became infected.[6] The absence of clinical signs in these bats is characteristic of a reservoir species. In 2002-03, a survey of 1,030 animals from Gabon and the Republic of the Congo including 679 bats found Ebolavirus RNA in 13fruit bats (Hyspignathus monstrosus, Epomops franquetti and Myonycteris torquata).[7] Bats are also known to be the reservoirs for a number of related viruses including Nipah virus, Hendra virus andlyssaviruses.
Microscopic Pathology
The images below display key features of the Ebola virus.
-
This transmission electron micrograph (TEM) demonstrates the ultrastructural morphology displayed by an Ebola virus. Source: CDC microbiologist Frederick A. Murphy.Adapted from Public Health Image Library (PHIL), Centers for Disease Control and Prevention.[8]
-
This transmission electron micrograph (TEM) demonstrates the ultrastructural morphologic changes in this tissue sample isolate.Adapted from Public Health Image Library (PHIL), Centers for Disease Control and Prevention.[8]
-
Scanning electron micrograph (SEM) revealing ultrastructural morphologic features of the Ebola virus from the Ivory Coast of Africa.Adapted from Public Health Image Library (PHIL), Centers for Disease Control and Prevention.[8]
-
Negatively-stained transmission electron micrograph (TEM) demonstrating the ultrastructural curvilinear morphologic features displayed by the Ebola virus from the Ivory Coast of Africa.Adapted from Public Health Image Library (PHIL), Centers for Disease Control and Prevention.[8]
Gallery
-
Produced by the National Institute of Allergy and Infectious Diseases (NIAID), this digitally-colorized scanning electron micrograph (SEM) depicts numerous string-like Ebola virus particles as they were in the process of being shed from an infected cell. From Public Health Image Library (PHIL). [8]
-
Produced by the National Institute of Allergy and Infectious Diseases (NIAID), this digitally-colorized scanning electron micrograph (SEM) depicts numerous filamentous Ebola virus particles (blue) budding from a chronically-infected VERO E6 cell (yellow-green). From Public Health Image Library (PHIL). [8]
-
Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a magnification of 25,000X, this digitally-colorized scanning electron micrograph (SEM) depicts numerous filamentous Ebola virus particles (green) budding from a chronically-infected VERO E6 cell (orange). From Public Health Image Library (PHIL). [8]
-
Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a magnification of 15,000X, this scanning electron photomicrograph (SEM) depicts numerous filamentous Ebola virus particles attached and budding from a chronically-infected VERO E6 cell. From Public Health Image Library (PHIL). [8]
-
Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a magnification of 50,000X, this scanning electron micrograph (SEM) depicts numerous filamentous Ebola virus particles replicating from an infected VERO E6 cell. From Public Health Image Library (PHIL). [8]
-
Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a magnification of 50,000X, this scanning electron micrograph (SEM) depicts numerous filamentous Ebola virus particles replicating from an infected VERO E6 cell. From Public Health Image Library (PHIL). [8]
-
Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a very-high magnification, this digitally-colorized scanning electron micrograph (SEM) depicts a single filamentous Ebola virus particle that had budded from the surface of a VERO cell of the African green monkey kidney epithelial cell line. From Public Health Image Library (PHIL). [8]
References
- ↑ "Taxonomy browser (Ebolavirus)".
- ↑ 2.0 2.1 2.2 2.3 2.4 Klenk, Hans-Dieter (2004). Ebola and Marburg Viruses, Molecular and Cellular Biology. Wymondham, Norfolk: Horizon Bioscience. ISBN 0954523237. Unknown parameter
|coauthors=ignored (help) - ↑ 3.0 3.1 Bausch DG, Towner JS, Dowell SF, Kaducu F, Lukwiya M, Sanchez A; et al. (2007). "Assessment of the risk of Ebola virus transmission from bodily fluids and fomites". J Infect Dis. 196 Suppl 2: S142–7. doi:10.1086/520545. PMID 17940942.
- ↑ 4.0 4.1 4.2 Pourrut, Xavier (2005). "The natural history of Ebola virus in Africa". Microbes and Infection. 7 (7–8): 1005–1014. doi:10.1016/j.micinf.2005.04.006. Unknown parameter
|coauthors=ignored (help) - ↑ "Fruit bats may carry Ebola virus". BBC News. 2005-12-11. Retrieved 2008-02-25.
- ↑ Swanepoel, R (1996). "Experimental inoculation of plants and animals with Ebola virus". Emerging Infectious Diseases. 2: 321–325. Unknown parameter
|coauthors=ignored (help) - ↑ Leroy, Eric (2005). "Fruit bats as reservoirs of Ebola virus". Nature. 438: 575–576. doi:10.1038/438575a. Unknown parameter
|coauthors=ignored (help) - ↑ 8.00 8.01 8.02 8.03 8.04 8.05 8.06 8.07 8.08 8.09 8.10 "Public Health Image Library (PHIL), Centers for Disease Control and Prevention Lassa fever history".
![This transmission electron micrograph (TEM) demonstrates the ultrastructural morphology displayed by an Ebola virus. Source: CDC microbiologist Frederick A. Murphy.Adapted from Public Health Image Library (PHIL), Centers for Disease Control and Prevention.[8]](/images/9/9d/Ebola_virus2.png)
![This transmission electron micrograph (TEM) demonstrates the ultrastructural morphologic changes in this tissue sample isolate.Adapted from Public Health Image Library (PHIL), Centers for Disease Control and Prevention.[8]](/images/7/78/Ebola_virus1.png)
![Scanning electron micrograph (SEM) revealing ultrastructural morphologic features of the Ebola virus from the Ivory Coast of Africa.Adapted from Public Health Image Library (PHIL), Centers for Disease Control and Prevention.[8]](/images/c/c4/Ebola_virus3.png)
![Negatively-stained transmission electron micrograph (TEM) demonstrating the ultrastructural curvilinear morphologic features displayed by the Ebola virus from the Ivory Coast of Africa.Adapted from Public Health Image Library (PHIL), Centers for Disease Control and Prevention.[8]](/images/2/2e/Ebola_virus4.png)
![Produced by the National Institute of Allergy and Infectious Diseases (NIAID), this digitally-colorized scanning electron micrograph (SEM) depicts numerous string-like Ebola virus particles as they were in the process of being shed from an infected cell. From Public Health Image Library (PHIL). [8]](/images/3/3e/Ebolavirus10.jpeg)
![Produced by the National Institute of Allergy and Infectious Diseases (NIAID), this digitally-colorized scanning electron micrograph (SEM) depicts numerous filamentous Ebola virus particles (blue) budding from a chronically-infected VERO E6 cell (yellow-green). From Public Health Image Library (PHIL). [8]](/images/1/1d/Ebolavirus09.jpeg)
![Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a magnification of 25,000X, this digitally-colorized scanning electron micrograph (SEM) depicts numerous filamentous Ebola virus particles (green) budding from a chronically-infected VERO E6 cell (orange). From Public Health Image Library (PHIL). [8]](/images/6/68/Ebolavirus08.jpeg)
![Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a magnification of 15,000X, this scanning electron photomicrograph (SEM) depicts numerous filamentous Ebola virus particles attached and budding from a chronically-infected VERO E6 cell. From Public Health Image Library (PHIL). [8]](/images/1/1d/Ebolavirus07.jpeg)
![Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a magnification of 50,000X, this scanning electron micrograph (SEM) depicts numerous filamentous Ebola virus particles replicating from an infected VERO E6 cell. From Public Health Image Library (PHIL). [8]](/images/8/8c/Ebolavirus06.jpeg)
![Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a magnification of 50,000X, this scanning electron micrograph (SEM) depicts numerous filamentous Ebola virus particles replicating from an infected VERO E6 cell. From Public Health Image Library (PHIL). [8]](/images/f/f4/Ebolavirus05.jpeg)
![Produced by the National Institute of Allergy and Infectious Diseases (NIAID), under a very-high magnification, this digitally-colorized scanning electron micrograph (SEM) depicts a single filamentous Ebola virus particle that had budded from the surface of a VERO cell of the African green monkey kidney epithelial cell line. From Public Health Image Library (PHIL). [8]](/images/1/13/Ebolavirus04.jpeg)