Metharbital
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
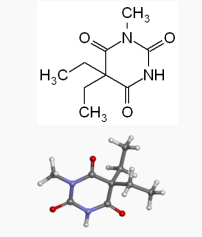
| Formula | C9H14N2O3 |
| Molar mass | 198.219 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
|
WikiDoc Resources for Metharbital |
|
Articles |
|---|
|
Most recent articles on Metharbital Most cited articles on Metharbital |
|
Media |
|
Powerpoint slides on Metharbital |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Metharbital at Clinical Trials.gov Clinical Trials on Metharbital at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Metharbital
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Metharbital Discussion groups on Metharbital Patient Handouts on Metharbital Directions to Hospitals Treating Metharbital Risk calculators and risk factors for Metharbital
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Metharbital |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Metharbital was patented in 1905 by Emil Fischer working for Merck.[1] It was marketed as Gemonil by Abbott Laboratories. It is a barbiturate anticonvulsant, used in the treatment of epilepsy. It has similar properties to phenobarbital.
History
- 1952 Gemonil was introduced by Abbott Laboratories.
- 1990 Abbott stopped marketing.
Synthesis
![700px[2][3] U.S. Patent 782,742](/images/7/76/Metharbital_syn.png)
In an interesting variation on this theme (cf hexobarbital), the bis acid chloride of diethylmalonate (1) is condensed with the O-methyl ether of urea (2, Methyl carbamimidate[4]) to afford the imino ether of the barbituric acid (3). Heating this ether at 200°C results in O to N migration of the methyl group and formation of metharbital 4.
References
- ↑ US Patent 782742
- ↑ A. Halpern, J.W. Jones, J. Am. Pharm. Assoc., 38, 352 (1949).
- ↑ Template:Cite doi
- ↑ http://www.chemspider.com/Chemical-Structure.68069.html?rid=81ad3137-afb0-456a-8c9f-d6c73ada4966
- The Treatment of Epilepsy 2nd Ed by S. D. Shorvon (Editor), David R. Fish (Editor), Emilio Perucca (Editor), W. Edwin Dodson (Editor). Published by Blackwell 2004. ISBN 0-632-06046-8
- The Medical Treatment of Epilepsy by Stanley R Resor. Published by Marcel Dekker (1991). ISBN 0-8247-8549-5.
- The Comparative Toxicogenomics Database: Metharbital
- Pages with script errors
- Template:drugs.com link with non-standard subpage
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Drugs with no legal status
- Drug
- Barbiturates
- Anticonvulsants
- Pyrimidinediones
- Imides
- GABAA receptor positive allosteric modulators
- Anticonvulsant stubs