Primidone
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Deepika Beereddy, MBBS [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Primidone is an anticonvulsant that is FDA approved for the treatment of epilepsy. Common adverse reactions include ataxia, vertigo.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Epilepsy, Adjunct or monotherapy
- Primidone tablets, USP, used alone or concomitantly with other anticonvulsants, is indicated in the control of grand mal, psychomotor, and focal epileptic seizures. It may control grand mal seizures refractory to other anticonvulsant therapy.
- Dosing Information
- The usual oral primidone maintenance dose for adults is 250 milligrams 3 or 4 times daily. When initiating primidone therapy, the drug should be started at 100 to 125 milligrams at bedtime for 3 days, 100 to 125 milligrams twice daily for days 4 to 6, 100 to 125 milligrams 3 times daily for days 7 to 9, and finally a maintenance dose of 250 milligrams 3 times daily.
- Primidone doses greater than 2 grams/day are not recommended.
- When used with or to replace other anticonvulsant therapy, the dosage of primidone should be increased gradually while the dosage of the other medication is maintained or decreased gradually in order to maintain seizure control. When therapy with primidone alone is the objective, the transition should not be completed in less than 2 weeks.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Primidone in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Primidone in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Epilepsy, Adjunct or monotherapy
- Dosing Information
- CHILDREN UNDER 8 YEARS:
- To start primidone therapy in children under 8 years of age, 50 milligrams can be given at bedtime for the first three days. For days 4 through 6, the dose is 50 milligrams twice a day. For days 7 through 9, the dose is 100 milligrams twice a day. The dose from day 10 until maintenance is reached is 125 milligrams to 250 milligrams three times a day, or 10 to 25 milligrams/kilogram/day in divided doses.
- When used with or to replace other anticonvulsant therapy, the dosage of primidone should be increased gradually while the dosage of the other medication is maintained or decreased gradually in order to maintain seizure control. When therapy with primidone alone is the objective, the transition should not be completed in less than 2 weeks.
- CHILDREN OVER 8 YEARS:
- The usual oral primidone maintenance dose for children 8 years of age and older is 250 milligrams 3 or 4 times daily. When initiating primidone therapy, the drug should be started at 100 to 125 milligrams at bedtime for 3 days, 100 to 125 milligrams twice daily for days 4 to 6, 100 to 125 milligrams 3 times daily for days 7 to 9, and finally a maintenance dose of 250 milligrams 3 times daily.
- When used with or to replace other anticonvulsant therapy, the dosage of primidone should be increased gradually while the dosage of the other medication is maintained or decreased gradually in order to maintain seizure control. When therapy with primidone alone is the objective, the transition should not be completed in less than 2 weeks.
- WITHDRAWAL SCHEDULE:
- Withdrawal of anticonvulsant medication in children free of seizures for 2 to 4 years appears to be safe, with the majority of children remaining free of seizures after medication withdrawal. In a prospective study, anticonvulsant medications were discontinued in 88 epileptic children who had not had a seizure for 2 to 4 years. Anticonvulsants were withdrawn gradually over 2 to 3 months. The type of seizure, age at onset and EEG characteristics were considered important in predicting the outcome of anticonvulsant withdrawal. The EEG obtained prior to withdrawal of the anticonvulsant was a useful predictor, with a normal EEG associated with good outcome. A poor outcome was associated with specific abnormalities such as slowing, spikes, and focality. A favorable prognosis was also demonstrated for patients with a history of complex partial seizures that had been controlled for two years on medication. Patients with a history of atypical febrile seizures had a poor prognosis. A better prognosis was observed in patients with a younger age at the onset of seizure activity; however, this was only significant if EEG slowing was present. These data indicate that withdrawal of anticonvulsants in children who have been seizure free for 2 years is relatively safe, and it is possible to predict outcome reasonably well on the basis of EEG, type of seizure and age of onset of seizures.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Primidone in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Primidone in pediatric patients.
Contraindications
- Primidone, USP is contraindicated in:
- patients with porphyria and
- who are hypersensitive to phenobarbital.
Warnings
- The abrupt withdrawal of antiepileptic medication may precipitate status epilepticus. The therapeutic efficacy of a dosage regimen takes several weeks before it can be assessed.
- Suicidal Behavior and Ideation
- Antiepileptic drugs (AEDs), including Primidone, increase the risk of suicidal thoughts or behavior in patients taking these drugs for any indication. Patients treated with any AED for any indication should be monitored for the emergence or worsening of depression, suicidal thoughts or behavior, and/or any unusual changes in mood or behavior.
- Pooled analyses of 199 placebo-controlled clinical trials (mono- and adjunctive therapy) of 11 different AEDs showed that patients randomized to one of the AEDs had approximately twice the risk (adjusted Relative Risk 1.8, 95% CI:1.2, 2.7) of suicidal thinking or behavior compared to patients randomized to placebo. In these trials, which had a median treatment duration of 12 weeks, the estimated incidence rate of suicidal behavior or ideation among 27,863 AED-treated patients was 0.43%, compared to 0.24% among 16,029 placebo-treated patients, representing an increase of approximately one case of suicidal thinking or behavior for every 530 patients treated. There were four suicides in drug-treated patients in the trials and none in placebo-treated patients, but the number is too small to allow any conclusion about drug effect on suicide.
- The increased risk of suicidal thoughts or behavior with AEDs was observed as early as one week after starting drug treatment with AEDs and persisted for the duration of treatment assessed. Because most trials included in the analysis did not extend beyond 24 weeks, the risk of suicidal thoughts or behavior beyond 24 weeks could not be assessed.
- The risk of suicidal thoughts or behavior was generally consistent among drugs in the data analyzed. The finding of increased risk with AEDs of varying mechanisms of action and across a range of indications suggests that the risk applies to all AEDs used for any indication. The risk did not vary substantially by age (5 to 100 years) in the clinical trials analyzed.

- The relative risk for suicidal thoughts or behavior was higher in clinical trials for epilepsy than in clinical trials for psychiatric or other conditions, but the absolute risk differences were similar for the epilepsy and psychiatric indications.
- Anyone considering prescribing primidone or any other AED must balance the risk of suicidal thoughts or behavior with the risk of untreated illness. Epilepsy and many other illnesses for which AEDs are prescribed are themselves associated with morbidity and mortality and an increased risk of suicidal thoughts and behavior. Should suicidal thoughts and behavior emerge during treatment, the prescriber needs to consider whether the emergence of these symptoms in any given patient may be related to the illness being treated.
- Patients, their caregivers, and families should be informed that AEDs increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of the signs and symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
- Usage in Pregnancy
- To provide information regarding the effects of in utero exposure to primidone, physicians are advised to recommend that pregnant patients taking Primidone enroll in the North American Antiepileptic Drug (NAAED) Pregnancy Registry. This can be done by calling the toll free number 1-888-233-2334, and must be done by patients themselves. Information on the registry can also be found at the website
- The effects of primidone in human pregnancy and nursing infants are unknown.
- Recent reports suggest an association between the use of anticonvulsant drugs by women with epilepsy and an elevated incidence of birth defects in children born to these women. Data are more extensive with respect to diphenylhydantoin and phenobarbital, but these are also the most commonly prescribed anticonvulsants; less systematic or anecdotal reports suggest a possible similar association with the use of all known anticonvulsant drugs.
- The reports suggesting an elevated incidence of birth defects in children of drug-treated epileptic women cannot be regarded as adequate to prove a definite cause-and-effect relationship.
- There are intrinsic methodologic problems in obtaining adequate data on drug teratogenicity in humans, the possibility also exists that other factors leading to birth defects, e.g., genetic factors or the epileptic condition itself, may be more important than drug therapy. The great majority of mothers on anticonvulsant medication deliver normal infants. It is important to note that anticonvulsant drugs should not be discontinued in patients in whom the drug is administered to prevent major seizures because of the strong possibility of precipitating status epilepticus with attendant hypoxia and threat to life. In individual cases where the severity and frequency of the seizure disorders are such that the removal of medication does not pose a serious threat to the patient, discontinuation of the drug may be considered prior to and during pregnancy, although it cannot be said with any confidence that even minor seizures do not pose some hazard to the developing embryo or fetus.
- The prescribing physician will wish to weigh these considerations in treating or counseling epileptic women of childbearing potential. Neonatal hemorrhage, with a coagulation defect resembling vitamin K deficiency, has been described in newborns whose mothers were taking primidone and other anticonvulsants. Pregnant women under anticonvulsant therapy should receive prophylactic vitamin K1 therapy for one month prior to, and during, delivery.
Precautions
- The total daily dosage should not exceed 2 g. Since primidone therapy generally extends over prolonged periods, a complete blood count and a sequential multiple analysis-12 (SMA-12) test should be made every six months.
- In Nursing Mothers
- There is evidence that in mothers treated with primidone, the drug appears in the milk in substantial quantities. Since tests for the presence of primidone in biological fluids are too complex to be carried out in the average clinical laboratory, it is suggested that the presence of undue somnolence and drowsiness in nursing newborns of primidone-treated mothers be taken as an indication that nursing should be discontinued.
Adverse Reactions
Clinical Trials Experience
- The most frequently occurring early side effects are ataxia and vertigo. These tend to disappear with continued therapy, or with reduction of initial dosage. Occasionally, the following have been reported: nausea, anorexia, vomiting, fatigue, hyperirritability, emotional disturbances, sexual impotency, diplopia, nystagmus, drowsiness, and morbilliform skin eruptions. Granulocytopenia, agranulocytosis, and red-cell hypoplasia and aplasia, have been reported rarely. These and, occasionally, other persistent or severe side effects may necessitate withdrawal of the drug. Megaloblastic anemia may occur as a rare idiosyncrasy to primidone and to other anticonvulsants. The anemia responds to folic acid without necessity of discontinuing medication.
Postmarketing Experience
- There is limited information regarding Postmarketing Experience of Primidone in the drug label.
Drug Interactions
There is limited information regarding Primidone Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Pregnancy Category
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
- There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Primidone in women who are pregnant.
Labor and Delivery
- There is no FDA guidance on use of Primidone during labor and delivery.
Nursing Mothers
- There is no FDA guidance on the use of Primidone with respect to nursing mothers.
Pediatric Use
- There is no FDA guidance on the use of Primidone with respect to pediatric patients.
Geriatic Use
- There is no FDA guidance on the use of Primidone with respect to geriatric patients.
Gender
- There is no FDA guidance on the use of Primidone with respect to specific gender populations.
Race
- There is no FDA guidance on the use of Primidone with respect to specific racial populations.
Renal Impairment
- There is no FDA guidance on the use of Primidone in patients with renal impairment.
Hepatic Impairment
- There is no FDA guidance on the use of Primidone in patients with hepatic impairment.
Females of Reproductive Potential and Males
- There is no FDA guidance on the use of Primidone in women of reproductive potentials and males.
Immunocompromised Patients
- There is no FDA guidance one the use of Primidone in patients who are immunocompromised.
Administration and Monitoring
Administration
Adult Dosage
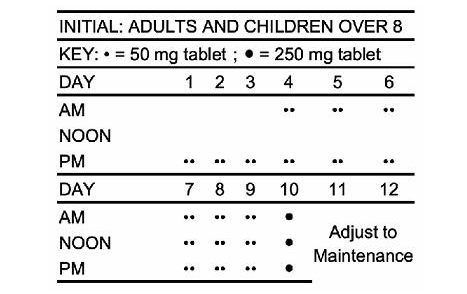
- Patients 8 years of age and older who have received no previous treatment may be started on primidone according to the following regimen using either 50 mg or scored 250 mg primidone tablets, USP:
- Days 1 to 3: 100 to 125 mg at bedtime.
- Days 4 to 6: 100 to 125 mg b.i.d.
- Days 7 to 9: 100 to 125 mg t.i.d.
- Day 10 to maintenance: 250 mg t.i.d.
- For most adults and children 8 years of age and over, the usual maintenance dosage is three to four 250 mg primidone tablets, USP daily in divided doses (250 mg t.i.d. or q.i.d.). If required, an increase to five or six 250 mg tablets daily may be made but daily doses should not exceed 500 mg q.i.d.
- Dosage should be individualized to provide maximum benefit. In some cases, serum blood level determinations of primidone, USP may be necessary for optimal dosage adjustment. The clinically effective serum level for primidone, USP is between 5 to 12 mcg/mL.
- In Patients Already Receiving Other Anticonvulsants
- Primidone tablets, USP should be started at 100 to 125 mg at bedtime and gradually increased to maintenance level as the other drug is gradually decreased. This regimen should be continued until satisfactory dosage level is achieved for the combination, or the other medication is completely withdrawn. When therapy with primidone tablets, USP alone is the objective, the transition from concomitant therapy should not be completed in less than two weeks.
- Pediatric Dosage
- For children under 8 years of age, the following regimen may be used:
- Days 1 to 3: 50 mg at bedtime.
- Days 4 to 6: 50 mg b.i.d.
- Days 7 to 9: 100 mg b.i.d.
- Day 10 to maintenance: 125 mg t.i.d. to 250 mg t.i.d.
- For children under 8 years of age, the usual maintenance dosage is 125 to 250 mg three times daily or 10 to 25 mg/kg/day in divided doses.
Monitoring
- There is limited information regarding Monitoring of Primidone in the drug label.
IV Compatibility
- There is limited information regarding IV Compatibility of Primidone in the drug label.
Overdosage
- There is limited information regarding Chronic Overdose of Primidone in the drug label.
Pharmacology
Mechanism of Action
Structure
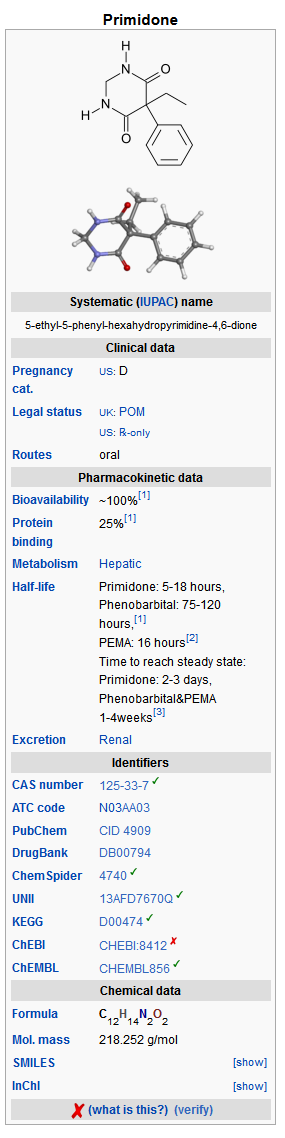
- Primidone, USP is a white, crystalline, highly stable substance, M.P. 279-284° C. It is poorly soluble in water (60 mg per 100 mL at 37° C) and in most organic solvents. It possesses no acidic properties, in contrast to its barbiturate analog.
- Chemical name: 5-ethyldihydro-5-phenyl-4,6 (1H, 5H)-pyrimidinedione. Structural formula:

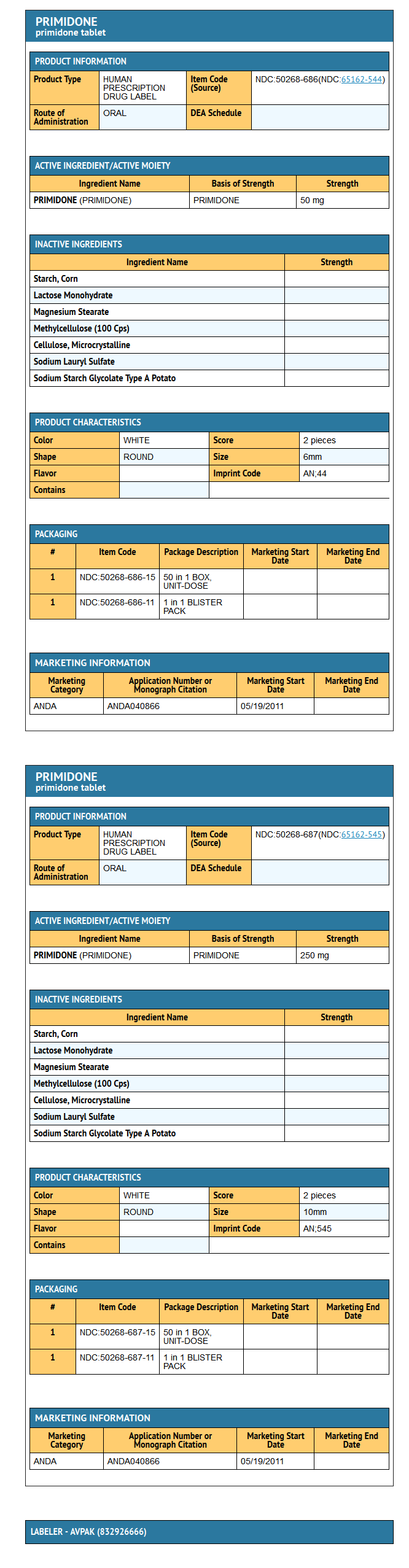
- Primidone tablets, USP 50 mg and 250 mg tablets contain the following inactive ingredients: corn starch, lactose monohydrate, magnesium stearate, methyl cellulose, microcrystalline cellulose, sodium lauryl sulfate, sodium starch glycolate.
Pharmacodynamics
- There is limited information regarding Pharmacodynamics of Primidone in the drug label.
Pharmacokinetics
- There is limited information regarding Pharmacokinetics of Primidone in the drug label.
Nonclinical Toxicology
- There is limited information regarding Nonclinical Toxicology of Primidone in the drug label.
Clinical Studies
- There is limited information regarding Clinical Studies of Primidone in the drug label.
How Supplied
- Primidone Tablets USP 50 mg are supplied as white, round, flat faced tablets, debossed
- AN above 44 on one side and cut-bisected on the other side.
- NDC 50268-686-15 10 Tablets per card, 5 cards per carton
- Primidone Tablets USP 250 mg are supplied as white, round, flat faced tablets, debossed
- AN bisect 545 on one side and plain on the other side.
- NDC 50268-687-15 10 Tablets per card, 5 cards per carton
Storage
- Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15° to 30°C (59° to 86°F).
- [See USP Controlled Room Temperature].
- KEEP THIS AND ALL MEDICATION OUT OF THE REACH OF CHILDREN.
- Dispensed in a blister punch material for Institutional Use Only.
Images
Drug Images
{{#ask: Page Name::Primidone |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel

{{#ask: Label Page::Primidone |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Information for Patients
- Suicidal Thinking and Behavior - Patients, their caregivers, and families should be counseled that AEDs, including Mysoline, may increase the risk of suicidal thoughts and behavior and should be advised of the need to be alert for the emergence or worsening of symptoms of depression, any unusual changes in mood or behavior, or the emergence of suicidal thoughts, behavior, or thoughts about self-harm. Behaviors of concern should be reported immediately to healthcare providers.
- Patients should be encouraged to enroll in the NAAED Pregnancy Registry if they become pregnant. This registry is collecting information about the safety of antiepileptic drugs during pregnancy. To enroll, patients can call the toll free number 1-888-233-2334 (see Usage in Pregnancy section).
- Please refer to the Primidone Medication Guide for more information.

Precautions with Alcohol
- Alcohol-Primidone interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
Mysoline
Look-Alike Drug Names
- A® — B®[1]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Primidone
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Primidone |Label Name=Primidone11.png
}}
{{#subobject:
|Label Page=Primidone |Label Name=Primidone11.png
}}