Posaconazole adverse reactions
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ahmed Zaghw, M.D. [2]
Adverse Reactions
Serious and Otherwise Important Adverse Reactions
The following serious and otherwise important adverse reactions are discussed in detail in another section of the labeling:
- Arrhythmias and QT Prolongation
- Hepatic Toxicity
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in clinical trials of Noxafil cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice. In clinical trials, the type of adverse reactions reported for posaconazole delayed-release tablets were generally similar to that reported in trials of posaconazole oral suspension.
Clinical Trial Experience with Posaconazole Delayed-Release Tablets
The safety of posaconazole delayed-release tablets has been assessed in 230 patients in clinical trials. Patients were enrolled in a non-comparative pharmacokinetic and safety trial of posaconazole delayed-release tablets when given as antifungal prophylaxis (Delayed-Release Tablet Study 1). Patients were immunocompromised with underlying conditions including hematological malignancy, neutropenia post-chemotherapy, GVHD, and post HSCT. This patient population was 62% male, had a mean age of 51 years (range 19-78 years, 17% of patients were ≥65 years of age), and were 93% white and 16% Hispanic. Posaconazole therapy was given for a median duration of 28 days.Twenty patients received 200 mg daily dose and 210 patients received 300 mg daily dose (following BID dosing on Day 1 in each cohort).Table 3 presents treatment-emergent adverse reactions observed in patients treated with 300 mg daily dose at an incidence of ≥10% in posaconazole delayed-release tablet study.
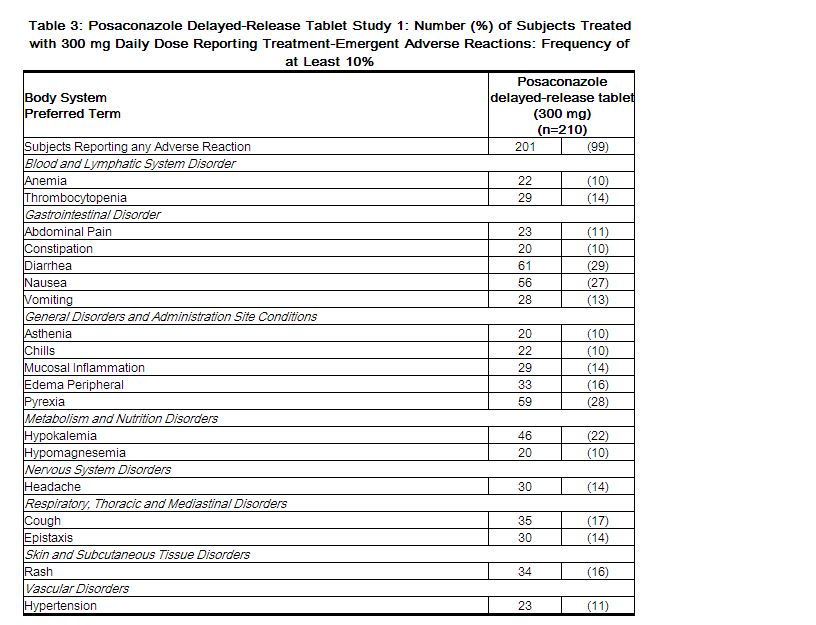 |
The most frequently reported adverse reactions (>25%) with posaconazole delayed-release tablets 300 mg once daily were diarrhea, pyrexia, and nausea.
The most common adverse reaction leading to discontinuation of posaconazole delayed-release tablets 300 mg once daily was nausea (2%).
Clinical Trial Safety Experience with Posaconazole Oral Suspension
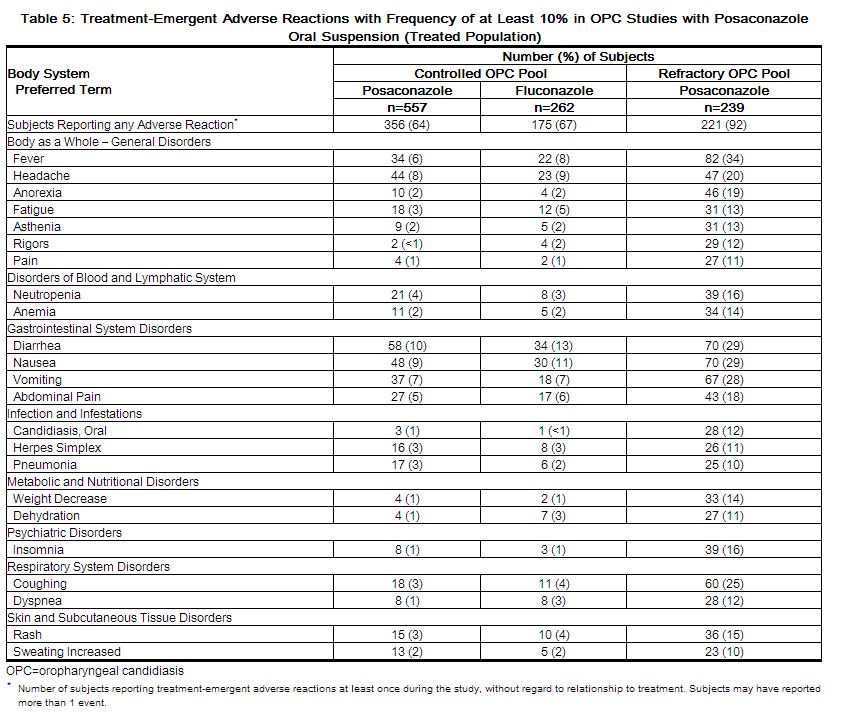
The safety of posaconazole oral suspension has been assessed in 1844 patients. This includes 605 patients in the active-controlled prophylaxis studies, 557 patients in the active-controlled OPC studies, 239 patients in refractory OPC studies, and 443 patients from other indications. This represents a heterogeneous population, including immunocompromised patients, e.g., patients with hematological malignancy, neutropenia post-chemotherapy, GVHD post HSCT, and HIV infection, as well as non-neutropenic patients. This patient population was 71% male, had a mean age of 42 years (range 8-84 years, 6% of patients were ≥65 years of age and 1% was <18 years of age), and were 64% white, 16% Hispanic, and 36% non-white (including 14% black). Posaconazole therapy was given to 171 patients for ≥6 months, with 58 patients receiving posaconazole therapy for ≥12 months. Table 4 presents treatment-emergent adverse reactions observed at an incidence of >10% in posaconazole prophylaxis studies. Table 5 presents treatment-emergent adverse reactions observed at an incidence of at least 10% in the OPC/rOPC studies.
Prophylaxis of Aspergillus and Candida: In the 2 randomized, comparative prophylaxis studies (Oral Suspension Studies 1 and 2), the safety of posaconazole oral suspension 200 mg three times a day was compared to fluconazole 400 mg once daily or itraconazole 200 mg twice a day in severely immunocompromised patients.
The most frequently reported adverse reactions (>30%) in the prophylaxis clinical trials were fever, diarrhea, and nausea.
The most common adverse reactions leading to discontinuation of posaconazole in the prophylaxis studies were associated with GI disorders, specifically, nausea (2%), vomiting (2%), and hepatic enzymes increased (2%).
 |
HIV Infected Subjects with OPC: In 2 randomized comparative studies in OPC, the safety of posaconazole oral suspension at a dose of less than or equal to 400 mg QD in 557 HIV-infected patients was compared to the safety of fluconazole in 262 HIV-infected patients at a dose of 100 mg QD.
An additional 239 HIV-infected patients with refractory OPC received posaconazole oral suspension in 2 non-comparative trials for refractory OPC (rOPC). Of these subjects, 149 received the 800-mg/day dose and the remainder received the less than or equal to 400-mg QD dose.
In the OPC/rOPC studies, the most common adverse reactions were fever, diarrhea, nausea, headache, vomiting, and coughing.
The most common adverse reactions that led to treatment discontinuation of posaconazole in the Controlled OPC Pool included respiratory impairment (1%) and pneumonia (1%). In the refractory OPC pool, the most common adverse reactions that led to treatment discontinuation of posaconazole were AIDS (7%) and respiratory impairment (3%).
 |
Adverse reactions were reported more frequently in the pool of patients with refractory OPC. Among these highly immunocompromised patients with advanced HIV disease, serious adverse reactions (SARs) were reported in 55% (132/239). The most commonly reported SARs were fever (13%) and neutropenia (10%).
Less Common Adverse Reactions: Clinically significant adverse reactions reported during clinical trials in prophylaxis, OPC/rOPC or other trials with posaconazole which occurred in less than 5% of patients are listed below:
Blood and lymphatic system disorders: hemolytic uremic syndrome, thrombotic thrombocytopenic purpura, neutropenia aggravated Endocrine disorders: adrenal insufficiency Nervous system disorders: paresthesia Immune system disorders: allergic reaction Cardiac disorders: Torsades de pointes Vascular disorders: pulmonary embolism Liver and Biliary System Disorders: bilirubinemia, hepatic enzymes increased, hepatic function abnormal, hepatitis, hepatomegaly, jaundice, AST Increased, ALT Increased Metabolic and Nutritional Disorders: hypokalemia Platelet, Bleeding, and Clotting Disorders: thrombocytopenia Renal & Urinary System Disorders: renal failure acute
Clinical Laboratory Values: In healthy volunteers and patients, elevation of liver function test values did not appear to be associated with higher plasma concentrations of posaconazole.
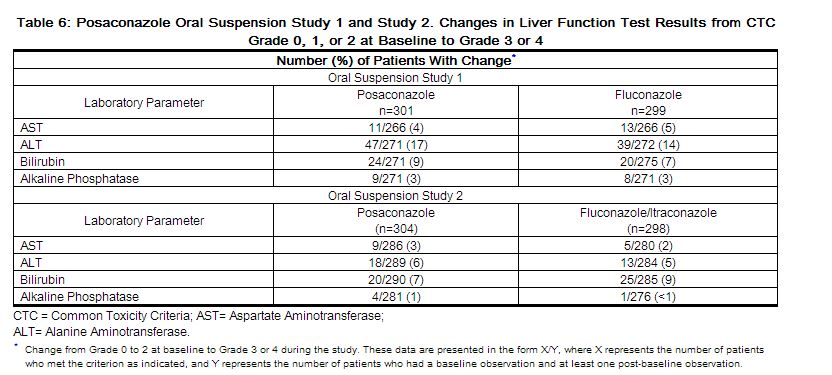
For the prophylaxis studies, the number of patients with changes in liver function tests from Common Toxicity Criteria (CTC) Grade 0, 1, or 2 at baseline to Grade 3 or 4 during the study is presented in Table 6.
 |
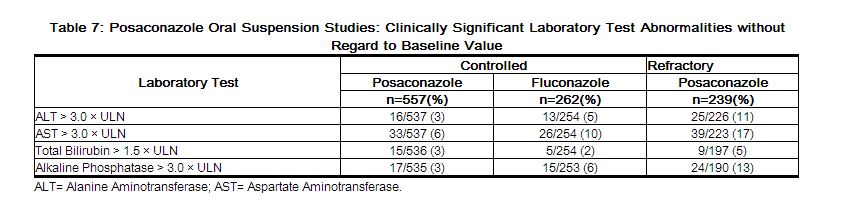
The number of patients treated for OPC with clinically significant liver function test (LFT) abnormalities at any time during the studies is provided in Table 7 (LFT abnormalities were present in some of these patients prior to initiation of the study drug).[1]
 |
References
Adapted from the FDA Package Insert.